Ever wondered what's really going on inside a liquid? It's not just plain old water, you know! Sometimes, when you mix things in, it's like a tiny, invisible party is happening.
Today, we're going to peek at a solution of K2CO3. That might sound like a secret code, but it's actually pretty fun to unpack. Think of it like a mysterious treasure chest of tiny bits!
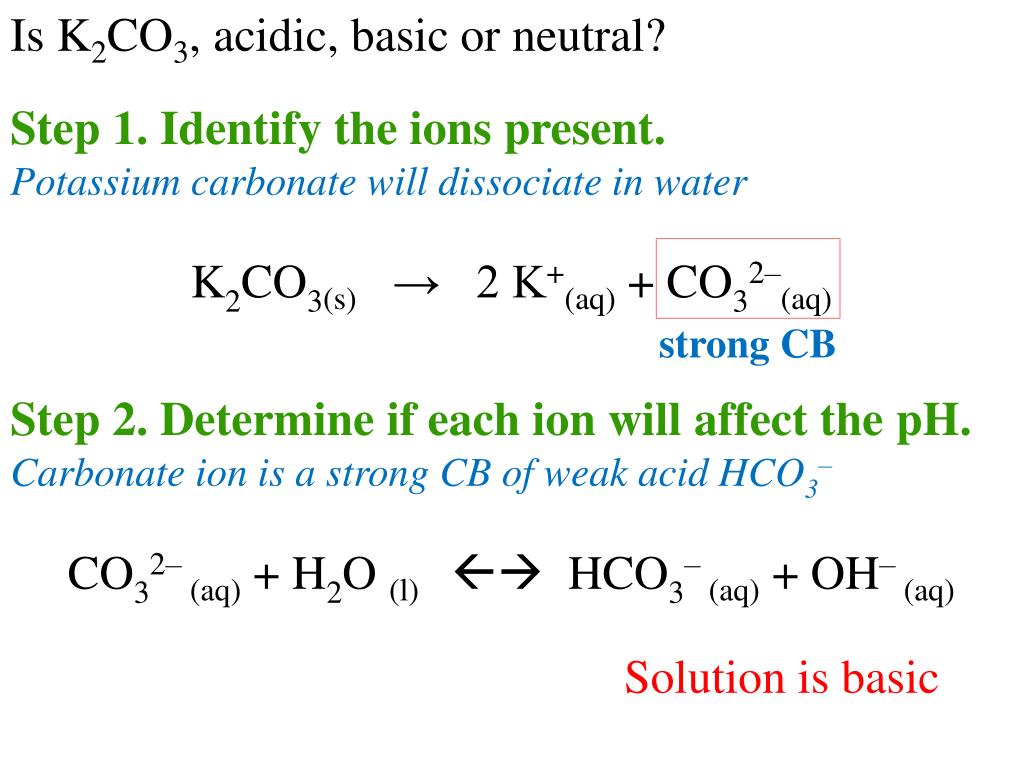
So, what happens when we dissolve K2CO3 in water? It's not like the whole thing just disappears. Instead, it breaks apart into its special pieces. These pieces are called ions.
Imagine you have a LEGO castle. When you break it down, you get individual LEGO bricks. These ions are like the LEGO bricks of K2CO3. They're the fundamental building blocks in the water!
The first superstar ion we meet is Potassium. It usually comes in pairs, so we have two of these guys. They're like the friendly twins of the solution.
These Potassium ions are represented by the symbol K+. The little plus sign is super important! It tells us they have a slight positive charge. It's like they're a little bit excited and eager to mingle.
Then, there's the other star of the show: Carbonate. This one is a bit more complex. It's not a single atom like potassium.
The Carbonate ion has the formula CO3^2-. See that little minus sign and the '2'? That means it has a double negative charge. It's like it's a bit shy and tends to attract positive things.
So, in our K2CO3 solution, we have these two types of ions dancing around. We have our energetic Potassium ions (K+) and our more reserved Carbonate ions (CO3^2-).
It's a constant swirl of activity. The Potassium ions are zipping around, and the Carbonate ions are doing their thing. They're all floating freely in the water.

Think of it like a busy swimming pool. You've got lots of swimmers (the ions) moving around. Some are doing laps (the potassium) and others are just chilling in a float (the carbonate).
And here's the really cool part: these ions aren't just passively floating. They're actually electrically charged! This is what makes them so interesting.
The positive Potassium ions and the negative Carbonate ions have a natural attraction. It's like magnets! Opposite charges love to be near each other.
However, because they're dissolved in water, they have plenty of space to move. Water molecules are great at separating these charged particles. They act like little buffers.
So, even though they're attracted, they don't clump back together permanently. They just sort of sway and interact. It's a dynamic balance.
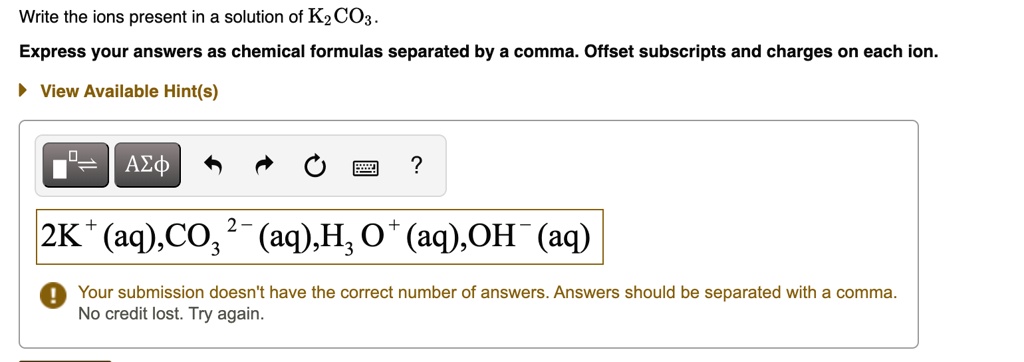
The number of each ion is also important. For every one CO3^2- ion, we have two K+ ions. This is to keep the whole solution neutral overall. It's like a perfectly balanced team.
If you had too many of one type of charge, the whole solution would be unbalanced. Chemistry likes things to be neat and tidy!
The presence of these ions is what gives the solution its unique properties. It's not just flavored water; it's a charged environment.
For example, these ions can react with other things you might add to the solution. This is where the real magic happens! It's like a chemistry playground.
When you add an acid to this solution, for instance, the Carbonate ions get really excited. They're like, "Oh, a positive charge! Let's react!"
This reaction can produce carbon dioxide gas. You might even see bubbles! That's the Carbonate ion and the acid having a little chat and creating something new.
The Potassium ions are also present and can participate in reactions, though sometimes they are more like spectators, cheering from the sidelines. They still play a vital role in maintaining the electrical balance.
So, the next time you hear about a solution of K2CO3, remember it's not just a boring chemical name. It's a whole world of tiny, charged particles having a lively time.
You've got your two friendly Potassium ions, K+, zipping around. And you've got your sophisticated Carbonate ion, CO3^2-, ready to engage.
It's like a miniature universe in a beaker. Each ion has its own personality and role to play. They interact and create a dynamic environment.
The beauty of it is that these ions are invisible to our naked eyes. We can't see them dancing or interacting, but their presence is undeniable. It's a hidden world of chemistry.
This concept of ions is fundamental to understanding how many substances behave in water. It explains why solutions conduct electricity or why they react in certain ways.
So, when you think about K2CO3, picture this: two happy K+ characters and one more thoughtful CO3^2- character, all hanging out together in a watery disco.
It’s a simple setup, but the possibilities for interaction are endless. This is what makes chemistry so fascinating. Even the most common substances have hidden depths.
The fact that these tiny particles can carry a charge and influence the entire solution is pretty amazing, don't you think? It's a testament to the intricate nature of matter.
So, to recap our little adventure, the key players in a solution of K2CO3 are the Potassium ions (K+) and the Carbonate ions (CO3^2-).
They're the charged bits that make the solution behave the way it does. They're the unsung heroes of the chemical world.

It’s like a dance floor, with the water molecules providing the music and the ions showing off their moves. Some are fast and energetic, others are a bit more graceful.
And it all comes back to that initial formula, K2CO3. It elegantly describes the exact ratio of these ions that will be present. It's a perfect blueprint.
So, the next time you encounter K2CO3, remember this invisible party. It’s a world of charged particles, a testament to the fun and fascinating nature of chemistry.
It's these little details, like the charges on ions, that make the whole chemical world so engaging and sometimes, wonderfully surprising. Keep an eye out for more invisible parties!
The invisible dance of ions is where the real excitement lies!
It’s like a secret code that unlocks the behavior of solutions. Understanding these ions opens up a whole new way of looking at the world around us.
So, the solution of K2CO3 is a vibrant mix. It’s not just a liquid; it’s a dynamic community of charged particles, each contributing to the overall character of the solution.
And that's the charm of it all. The seemingly simple can be incredibly complex and captivating when you look a little closer.
So, there you have it: the ions present in a solution of K2CO3. They're the stars of our miniature chemical show!