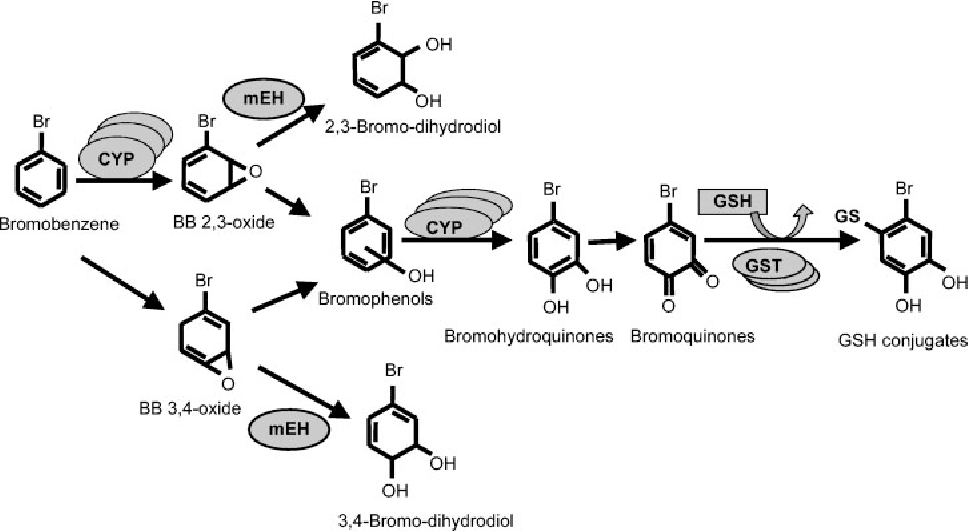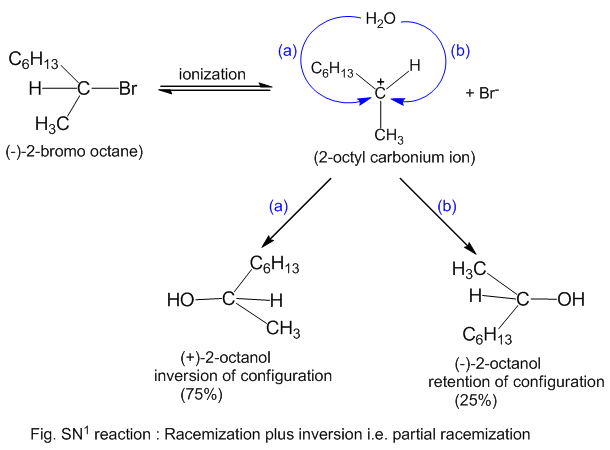
Ever wondered why some molecules are like shy wallflowers at a party, refusing to join the dance of chemical reactions? Today, we're going to pull back the curtain on one such molecule, bromobenzene, and explore why it’s surprisingly unreactive in two common types of reactions: SN1 and SN2. This might sound a bit technical, but understanding this is like unlocking a little secret about how chemistry works, and it’s actually quite fascinating!
So, why should you care about bromobenzene's quiet nature? For aspiring chemists, whether you're a student just dipping your toes into the world of organic chemistry or a hobbyist experimenting in your home lab, this knowledge is super useful. It helps you predict how molecules will behave, saving you time and preventing potentially messy (or just plain unsuccessful!) experiments. For families, imagine this as a fun science tidbit to share at the dinner table – a little peek into the invisible world of molecules that make up everything around us.
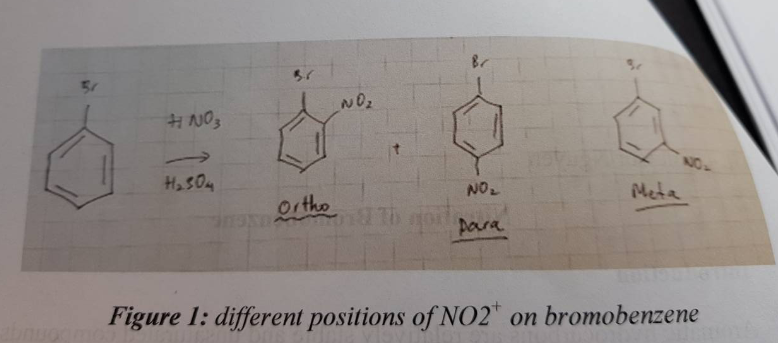
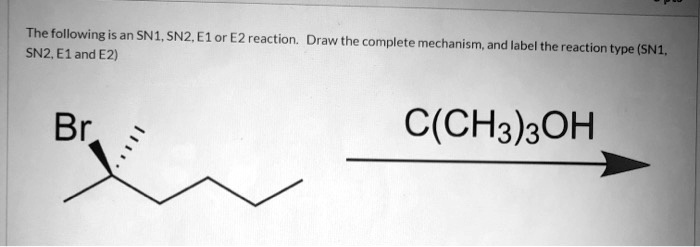
Let's break down SN1 and SN2 reactions briefly. Think of them as two different ways for a molecule to swap out a part of itself. In SN2, it’s a quick, one-step process where one group leaves as another arrives. In SN1, it's a two-step dance, where one group leaves first, creating a temporary "ion," and then another group comes in. Now, bromobenzene, with its benzene ring (that's a six-carbon ring with alternating double bonds, looking like a little hexagon) attached to a bromine atom, is generally a bit of a tough nut to crack for these reactions.
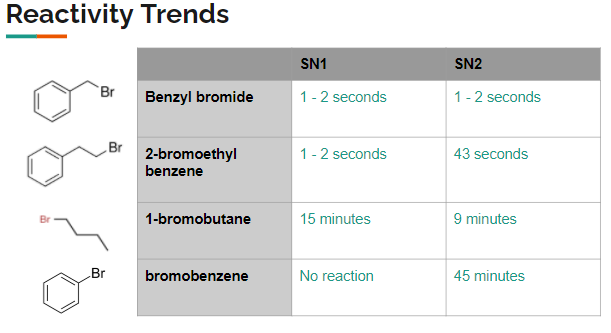
The main culprit is the strong bond between the carbon on the benzene ring and the bromine atom. This bond is particularly stable due to something called "resonance," where the electrons in the benzene ring are spread out, making that carbon-bromine connection much harder to break than in, say, a simple alkane. For SN2, this strong bond means it's very difficult for a new group to come in and kick the bromine out. Think of trying to push something out of a really snug-fitting hole!
For SN1, the situation is also tricky. If the bromine were to leave, it would form a positively charged ion on the benzene ring. This type of ion is quite unstable and doesn't like to stick around for long. The benzene ring's structure actually prefers to stay neutral and stable, so it resists forming this unstable intermediate. So, even though the bond might weaken a bit, the instability of the resulting ion means the SN1 pathway is also a no-go for bromobenzene under normal conditions.
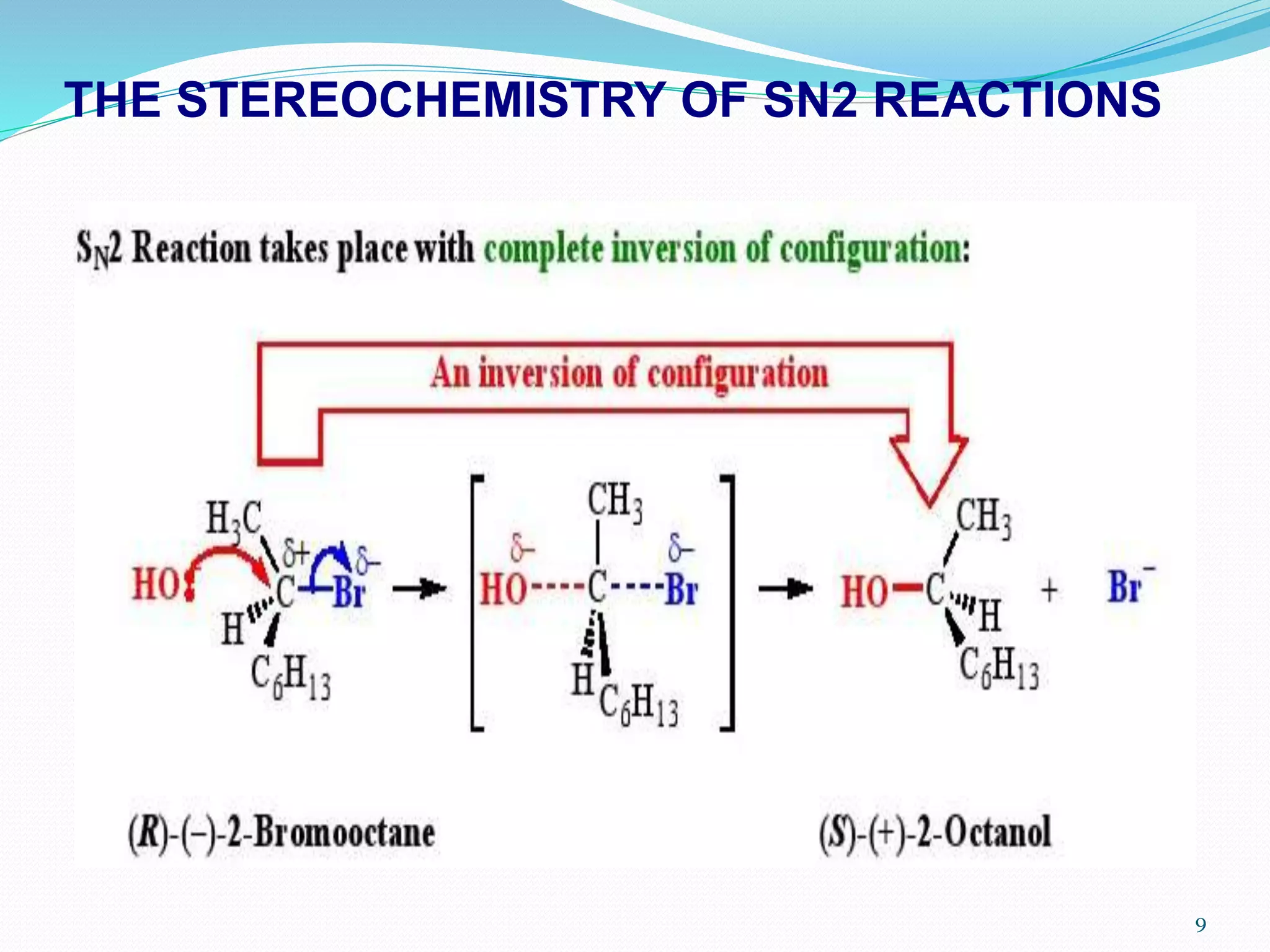
If you're keen to explore this further, a simple way to get started is to look at some visualizations of the bromobenzene molecule. You can find many online that show the electron distribution, helping you see why that carbon-bromine bond is so special. You could also compare its structure to molecules that do readily undergo SN1 or SN2 reactions, like simple alkyl halides (think methane with a bromine attached), to see the stark differences.
Understanding why bromobenzene is so unreactive in SN1 and SN2 reactions isn't just about memorizing facts; it's about appreciating the elegant stability of molecules. It’s a small piece of the vast and wonderful puzzle of chemistry, and recognizing these molecular personalities can make your journey of discovery even more rewarding and fun!