Hey there, science enthusiasts and curious minds! Ever wondered about those super-powered tiny things that make up, well, almost everything? Today, we're diving into the sparkly, sometimes shocking, world of ionic compounds! These are like the dynamic duos of the chemical world, and understanding them is easier than you think. Get ready for some fun facts and maybe a giggle or two!
Imagine a couple of friends. One is a bit of a giver, always happy to share their stuff. The other is a bit of a taker, always looking for a good deal. That's kind of what happens in an ionic compound! We have atoms that are basically looking to be complete, to have a full set of outer electrons, like collecting a full set of trading cards.
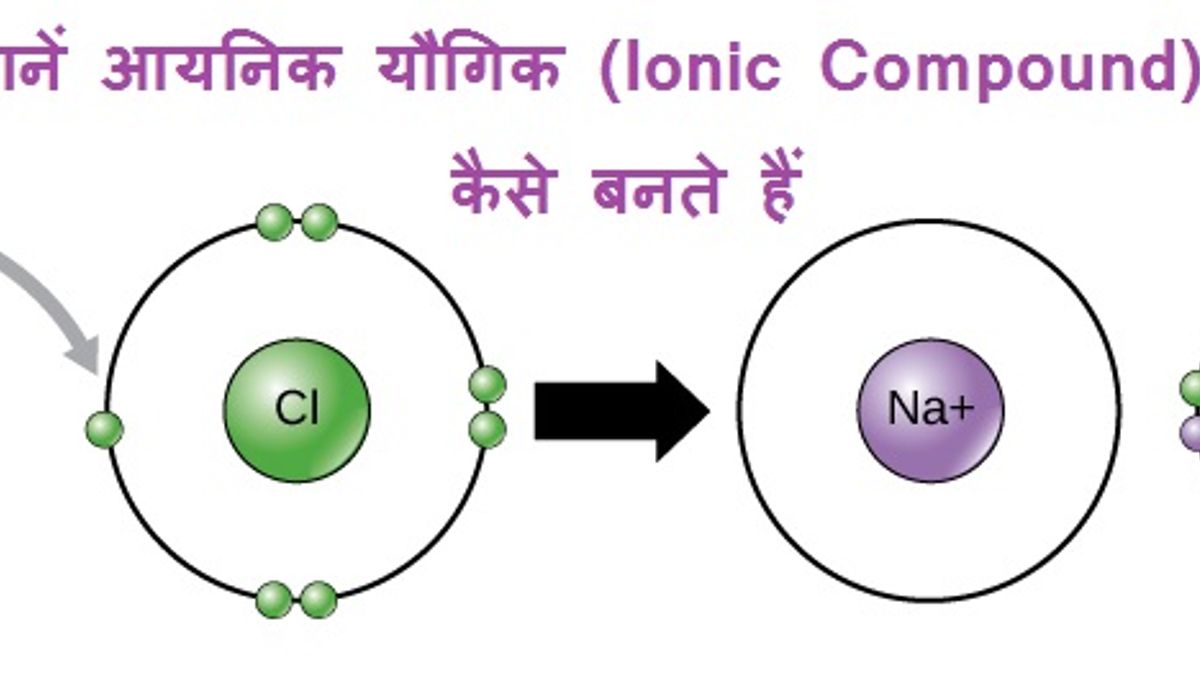
So, we have our generous atom, let's call him Sodium (Na). He's got one extra electron in his outer shell that he's just itching to get rid of. And then we have our eager atom, say Chlorine (Cl). Chlorine is desperately looking for just one electron to feel perfectly happy. It's a match made in chemical heaven!
When these two meet, Sodium, with a flourish, hands over that extra electron to Chlorine. Poof! Sodium is now a positively charged ion (it lost a negative electron, so it's got more positive protons hanging around). Chlorine, having happily accepted that electron, is now a negatively charged ion (it gained a negative electron).
And here's the magic: opposite charges attract! It's like a cosmic handshake, a magnetic pull that draws these newly formed ions together. They cling to each other with all their might, forming a solid, beautiful structure. This strong attraction is what we call an ionic bond, and the resulting creation is our fabulous ionic compound!
Think of it like little magnets sticking together. They don't just float around willy-nilly; they form an organized, repeating pattern. This is why many ionic compounds tend to form crystals. You know those sparkly, geometric shapes you see in geodes or fancy salt shakers? Many of those are the work of ionic compounds!
Let's talk about one of the most famous ionic compounds ever: table salt! Yep, that stuff you sprinkle on your popcorn or use to season your dinner is actually sodium chloride (NaCl). It's formed by that electron-donating Sodium and electron-accepting Chlorine we were just talking about.

Now, here's a super cool property of ionic compounds: they tend to have really high melting and boiling points. Imagine trying to pull apart those super-sticky magnets! It takes a lot of energy, a lot of heat, to break those strong ionic bonds. So, while your butter melts on a warm day, salt just sits there, stubbornly solid.
Which Statement Is True About Ionic Compounds?
Let's get down to brass tacks, shall we? When you see a statement about ionic compounds, what should you be looking for? What are their defining characteristics?
One big clue is their behavior in water. Remember those charged ions we created? Well, water is a fantastic solvent for many ionic compounds. When you dissolve salt in water, those individual ions get surrounded by water molecules, effectively breaking apart the crystal structure.

This is why solutions of ionic compounds can often conduct electricity. The charged ions are free to move around, carrying the electrical charge from one place to another. It's like they become tiny, mobile little batteries! So, if you see a statement about them conducting electricity when dissolved, that's a strong contender for truth!
However, in their solid, crystalline form, ionic compounds are generally poor conductors of electricity. The ions are locked in place within the crystal lattice, unable to move freely. They're like a tightly packed crowd at a concert – everyone's there, but they can't really go anywhere.
Another common characteristic you might encounter is their brittle nature. Remember how they form those organized crystal structures? If you hit one of these crystals, instead of bending, it tends to shatter. The layers of ions get pushed out of alignment, and suddenly, you have positive ions next to positive ions and negative next to negative. Like repels like, so the whole thing breaks apart!
So, to sum it up, when you're trying to identify a true statement about ionic compounds, keep these key features in mind:
![[ANSWERED] Which of the following statements about ionic compounds is](https://media.kunduz.com/media/sug-question/raw/50735035-1659119201.3454733.jpeg?h=512)
- They are formed by the transfer of electrons between atoms.
- They are held together by strong electrostatic attractions (ionic bonds).
- They often form crystalline solids.
- They typically have high melting and boiling points.
- They conduct electricity when melted or dissolved in water, but not in their solid state.
- They are often brittle.
Let's put this to the test with some hypothetical statements. Imagine someone says: "Ionic compounds are always soft and squishy, like a marshmallow." Nope! Remember those strong bonds and crystal structures? Marshmallows are definitely not made of ionic compounds.
Or what about: "Ionic compounds love to melt into a puddle at room temperature." Again, a big no-no! Their high melting points mean they prefer to stay solid unless you really crank up the heat.
But if you hear: "When you dissolve table salt in water, the solution can light up a tiny light bulb." Now we're talking! That's a classic example of an ionic compound solution conducting electricity.
It's all about those electron transfers and the resulting magnetic-like attraction between oppositely charged particles. They're not just random collections of atoms; they are beautifully organized structures with some pretty impressive properties.

Think about how common they are! From the minerals in the Earth's crust to the electrolytes in your body, ionic compounds are everywhere, playing crucial roles. They might seem simple, but their impact is colossal. It's like the humble brick – seemingly basic, but essential for building mighty castles!
So, the next time you're looking at a chemical statement, just picture those electron-swapping, attraction-loving, crystal-forming friends. Does the statement fit their personality? Does it align with their tendency to be strong, solid, and electrically conductive when given the chance?
Understanding ionic compounds is like unlocking a secret code to the material world. They're not just abstract scientific concepts; they're the building blocks of so much around us, and learning about them is a journey filled with fascinating discoveries. Keep exploring, keep questioning, and have fun with it!
Remember, the truth about ionic compounds often lies in their ability to form strong attractions, create ordered structures, and conduct electricity when their ions are on the move. It's a world of charged-up relationships, and it's absolutely amazing!