We've all been there, haven't we? Staring at a steaming cup of coffee, a perfectly sculpted ice sculpture, or a mesmerizing cloud formation. These aren't just pretty sights; they're everyday encounters with the fascinating world of states of matter. And when we talk about these states – solid, liquid, and gas – there's one concept that truly unlocks their secrets: entropy. It sounds a bit scientific, but trust me, understanding it can make observing the world around you infinitely more engaging and even a little bit mind-blowing.
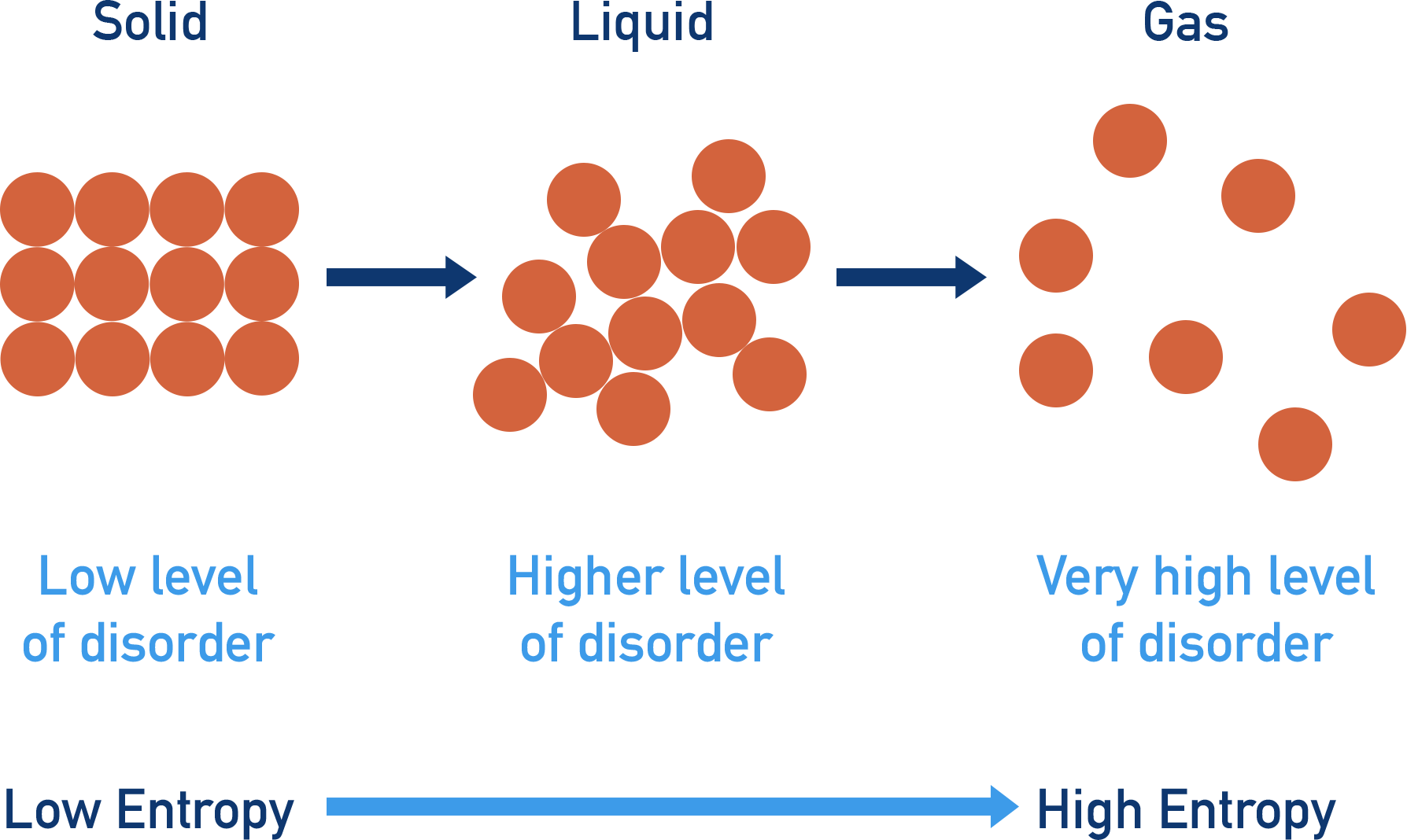
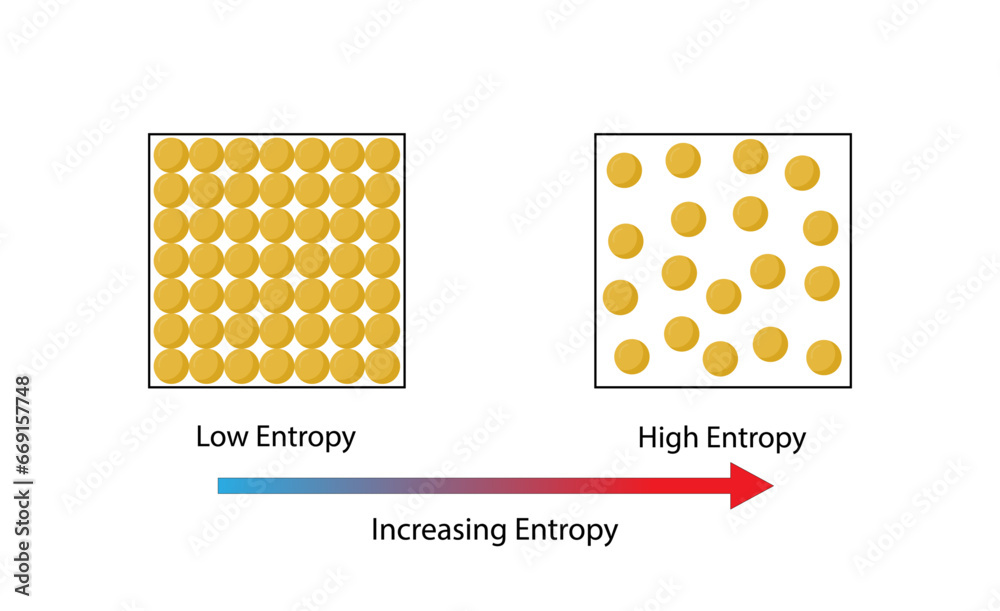
So, what exactly is entropy, and why should you care? In simple terms, entropy is a measure of disorder or randomness within a system. Think of it as how spread out and jumbled up the particles within something are. The higher the entropy, the more chaotic and less structured the arrangement of those particles. This concept is fundamental to how the universe works. It explains why your room tends to get messy if you don't actively tidy it, why heat flows from hot to cold, and why, ultimately, everything tends towards a state of greater disorder over time.
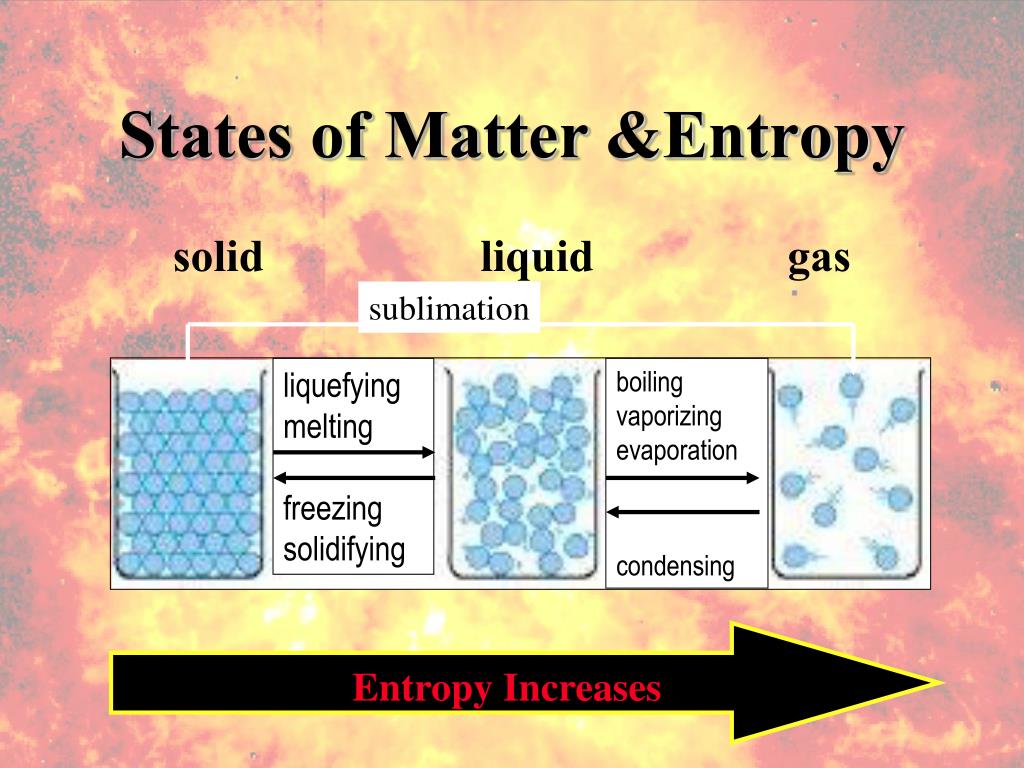
Now, let's get to the juicy part: which state of matter reigns supreme in the entropy department? While solids have their molecules locked in a rigid, orderly structure (low entropy), and liquids have molecules that can move around but are still relatively close together (medium entropy), it's the gas state that truly takes the crown for highest entropy. Imagine a gas: its particles are flying around with incredible speed and in complete disarray, bouncing off each other and anything else in their path. They spread out to fill any available space, exhibiting maximum freedom and minimum order. This is why a tiny puff of perfume can fill an entire room, or why a deflated balloon suddenly expands to take up so much more space when filled with air.
The practical applications of understanding entropy in states of matter are all around us. From the way engines work (converting heat energy into mechanical work often involves the expansion of gases) to weather patterns (the chaotic movement of air masses), entropy is at play. Even cooking involves entropy! Boiling water, for instance, transforms liquid water into gaseous steam, dramatically increasing its entropy and making it a powerful tool for cooking and cleaning.
Want to enjoy this concept more? Next time you’re out and about, make it a game! Observe the steam rising from a hot meal – marvel at the high entropy of the water vapor. Watch a balloon float away – think about the dispersed gas molecules. Or simply make a cup of tea and witness the sugar crystals dissolving, their ordered structure breaking down into a more disordered solution. You can even try a simple experiment at home: open a bottle of soda and watch the bubbles (dissolved gas escaping, increasing entropy) rise to the surface. It’s a fantastic way to connect with the fundamental forces shaping our universe, one state of matter at a time.