Hey there, science curious folks! Ever found yourself staring at a chemistry problem, or maybe just a list of chemicals, and wondered, "What's the deal with all these acids?" Today, we're gonna take a little dive into the fascinating world of acids, and specifically, we're going to tackle a question that might pop up: "Which one of the following is a diprotic acid?" Sounds a bit fancy, right? But don't worry, we're going to break it down in a way that's as chill as a lazy Sunday afternoon.
So, what even is an acid in the first place? Think of it like this: acids are those molecules that are a bit like generous donors. They love to hand out something called a proton. Now, a proton, in this context, is basically a positively charged particle that lives in the nucleus of an atom. When we talk about acids in water, they often release these protons, and that's what makes solutions acidic. Think of that sour taste in lemons or vinegar – that's your friendly neighborhood acid at work, donating protons!
But here's where it gets interesting. Not all acids are created equal, and they don't all donate protons in the same way. Some are like those friends who give you a single, really useful piece of advice. Others are a bit more… generous. And that's where our diprotic heroes come in.
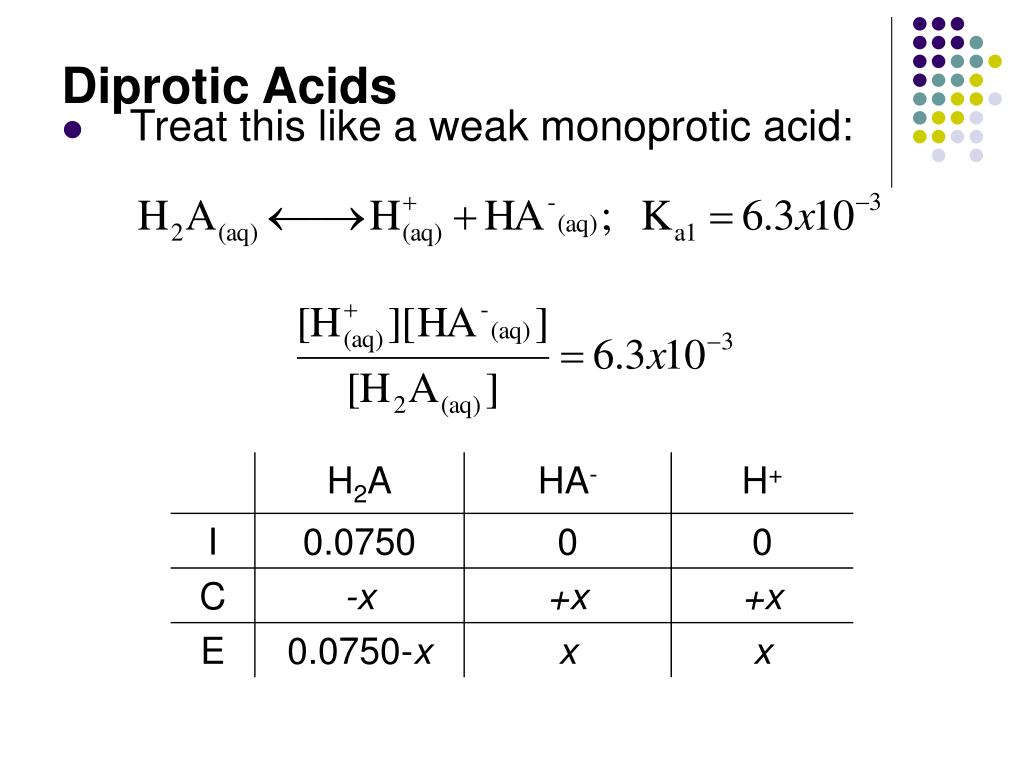
The word "di" means two, right? Like a bicycle has two wheels. So, a diprotic acid is an acid that can donate two protons. Isn't that neat? It’s like a two-for-one deal in the world of chemistry!
Imagine you have a molecule of a diprotic acid. It's got two little proton passengers it's willing to let go of. It doesn't just give them both up at once, though. It's usually a bit of a process. It'll donate one proton, and then, if the conditions are right, it can donate its second proton. It's like having two chances to get that proton donation!

Why is this even a big deal? Well, it affects how these acids behave in reactions. A diprotic acid can react differently depending on whether it's giving up one proton or both. This makes them super useful in a whole bunch of chemical processes. Think of them as versatile tools in a chemist's toolbox.
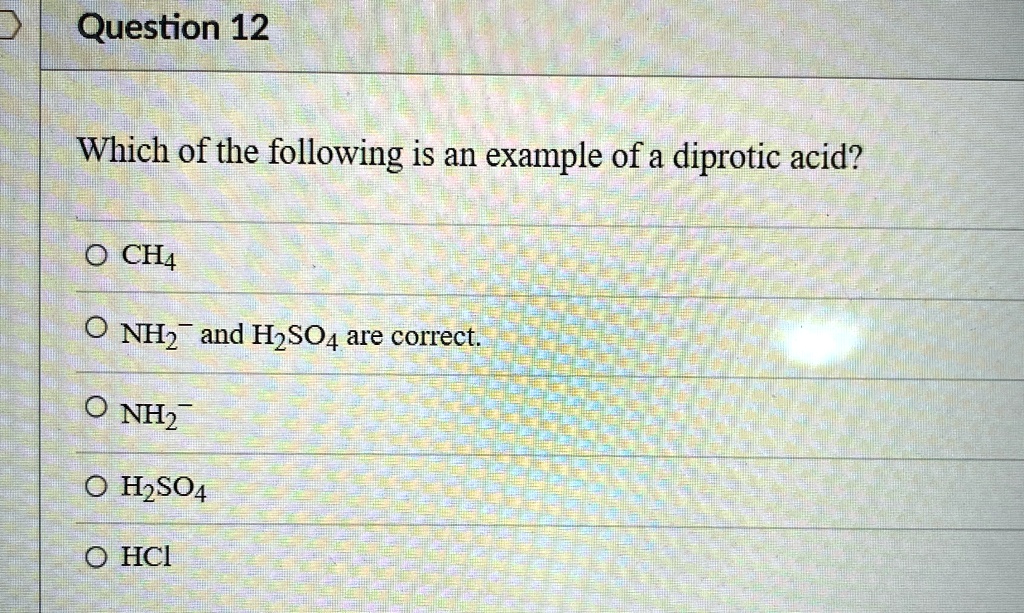
Now, you might be asking, "Okay, I get the concept, but what are some actual examples?" That's the fun part! Chemistry is full of these cool, everyday-ish substances that have these fascinating properties. The question we're playing with today is asking us to identify one of these diprotic wonders from a list. So, let's think about some common diprotic acids.
One of the most classic examples you'll often see is sulfuric acid. You might have heard of it. It's a pretty strong acid, and its chemical formula is H₂SO₄. See those two little 'H's at the beginning? Those are the protons ready to be donated. Sulfuric acid can donate one proton to become HSO₄⁻ (hydrogen sulfate ion), and then, under the right circumstances, it can donate its second proton to become SO₄²⁻ (sulfate ion). Pretty cool, huh?

Another really common and important diprotic acid is carbonic acid. This one is super relevant to our everyday lives, even if we don't always think about it. Carbonic acid is formed when carbon dioxide dissolves in water. You know how when you open a fizzy drink, you hear that hiss? That's the release of carbon dioxide, which then forms a little bit of carbonic acid in the liquid. Its formula is H₂CO₃. Again, look at those two 'H's! It can donate one proton to become HCO₃⁻ (bicarbonate ion) and then another to become CO₃²⁻ (carbonate ion).
Think about how important carbonic acid is. It plays a role in our blood, helping to maintain the right pH balance. It's also crucial for many natural processes, like weathering of rocks. It’s a quiet achiever, this carbonic acid!
So, if you were presented with a list, and you saw something like HCl (hydrochloric acid) or HNO₃ (nitric acid), those would be monoprotic acids. "Mono" means one, so they only have one proton to give. But if you saw H₂SO₄ or H₂CO₃, you'd be looking at our diprotic friends.

What about something like phosphoric acid (H₃PO₄)? That one is actually triprotic, meaning it can donate three protons. So, it's like the ultimate proton donor in this comparison! It’s a step up in generosity from our diprotic pals.
The question "Which one of the following is a diprotic acid?" is a great way to test your understanding of acid structure and how many acidic hydrogens a molecule possesses. It's not just about memorizing formulas; it's about understanding the logic behind them.
Why is this concept useful? For chemists, understanding if an acid is mono-, di-, or triprotic is key to predicting how it will react. It affects things like the buffer solutions they can create, the titrations they perform, and the overall chemistry of a system.
For us, as curious observers of the world, it’s just plain interesting to know that the substances around us have these subtle but significant differences. That fizzy drink isn't just bubbles; it's a temporary home for carbonic acid, a diprotic acid with a story to tell.
So, when you encounter that question, just remember: look for the acid that's ready to give up two protons. It’s the one with the "di" in its name, or at least, the "di" in its proton-donating potential. It’s a little bit of chemical superpower, hidden in plain sight!
Keep asking questions, keep exploring, and don't be afraid of the fancy-sounding terms. Most of the time, they're just describing something really cool and understandable. Happy learning!