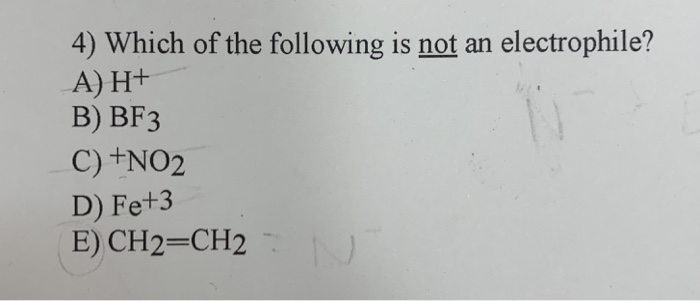
Imagine a world where certain molecules are like super-eager puppies, always sniffing around for a good time. They’re the life of the party, the ones who can’t resist a good hug. These are our electrophiles, and they’re a big deal in the invisible world of chemistry. Think of them as the folks who just have to be in the middle of the action, looking for something to latch onto. They’re generally a bit needy, with a craving for electrons – those tiny, negatively charged particles that make up the outer shells of atoms.
Now, in this bustling, electron-seeking world, there are also molecules that are the opposite. They’re like the calm, generous friends who have plenty to share. They’re not desperately looking for a handout; instead, they’re happily offering up their own electrons. These are the nucleophiles, and they’re just as important, often playing the role of the rescuer or the generous host.
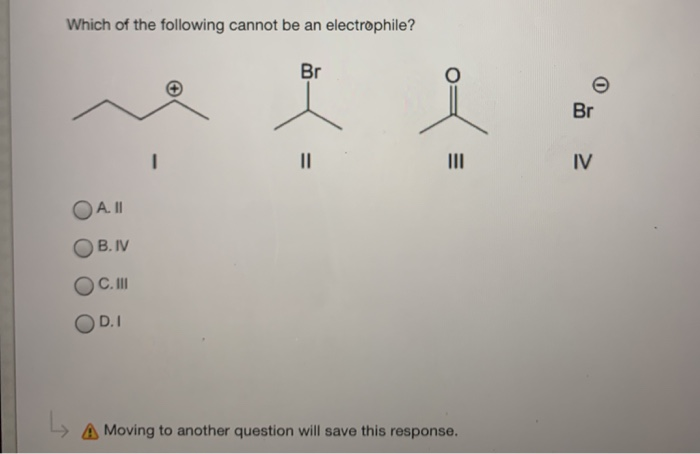
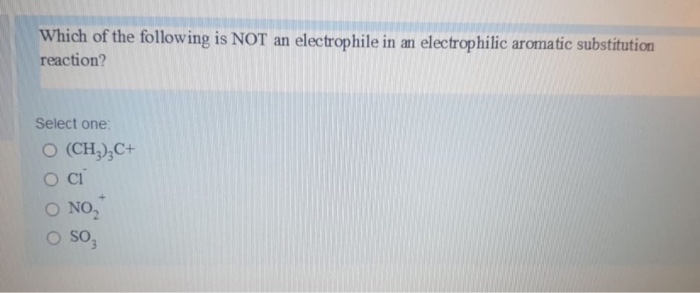
The question of the day, in this simplified chemical drama, is which of our contenders is not an electrophile. It’s like a “who’s who” of chemical personalities, and one of them is definitely not playing by the puppy-like, electron-grabbing rules. It’s a bit like trying to figure out who brought the vegan snacks to a cheese-tasting party – you know someone’s not quite fitting in, but it’s not necessarily a bad thing!
Let’s meet some of our potential players. We have BF₃, which stands for Boron Trifluoride. Now, Boron, the central atom here, is a bit of a loner in terms of electrons. It’s like it’s missing a crucial dance partner. It has these empty spots, these electron voids, that just scream, “Come hither, electron lovers!” So, BF₃ is definitely one of our eager puppies, always on the lookout for something to fill those empty spaces. It’s a classic electrophile, a real go-getter.
Then there’s NH₃, Ammonia. This one is the friendly neighbor with a big heart. The Nitrogen atom in Ammonia has a lone pair of electrons – a whole extra set that it’s not really using to bond with anything else. Imagine having two perfectly good cookies in your pocket when you’re already full. You’d probably offer one to a friend, right? That’s NH₃. It’s positively bursting with electron-rich generosity, making it a prime candidate for being a nucleophile, the electron-giver, rather than an electron-seeker.

Next up, we have AlCl₃, Aluminum Chloride. Similar to Boron Trifluoride, Aluminum here is in a bit of an electron-deficient situation. It’s got those same empty spaces, that same yearning for a good electron cuddle. So, AlCl₃ is another one of our enthusiastic electrophiles, always ready to jump into a situation where it can grab some much-needed electrons. It’s like the child who always volunteers for the task that involves collecting something, anything!
Finally, let’s consider H₂O, Water. Ah, water! The universal solvent, the essential ingredient for life, the stuff that makes up most of us! Water is pretty special. The Oxygen atom in water also has lone pairs of electrons, much like Ammonia. It’s got those extra electrons just waiting to be shared. Because of these electron-rich spots, water can act as a nucleophile, happily donating its electrons to an electron-hungry molecule. It’s like a kind librarian, always ready to lend a book (or in this case, electrons).

So, when we look at our lineup – BF₃, NH₃, AlCl₃, and H₂O – and ask, "Which of the following is not an electrophile?" – we’re looking for the one that doesn’t desperately want electrons. We’re looking for the one that has electrons to give.
BF₃ and AlCl₃ are our electron-craving champions. They’re the ones with the empty pockets, always looking to fill them. They are, without a doubt, electrophiles. They have a strong desire, an affinity, for electrons.
Now, NH₃ and H₂O are the generous souls. They have those extra electron pairs, ready to be shared. They are nucleophiles. They are electron-rich and love to donate. This electron-donating ability is what defines them as nucleophiles.
Therefore, the one that is not an electrophile is the one that is rich in electrons and ready to share. It's the molecule that acts as a giver, not a taker, in the world of chemical interactions. It’s not about being good or bad, just about chemical personalities! And in this particular lineup, the role of the electron-giver, the non-electrophile, is clearly played by those molecules with those happy, surplus electrons ready for a friendly exchange. They’re the ones who bring the party to the puppy, so to speak!