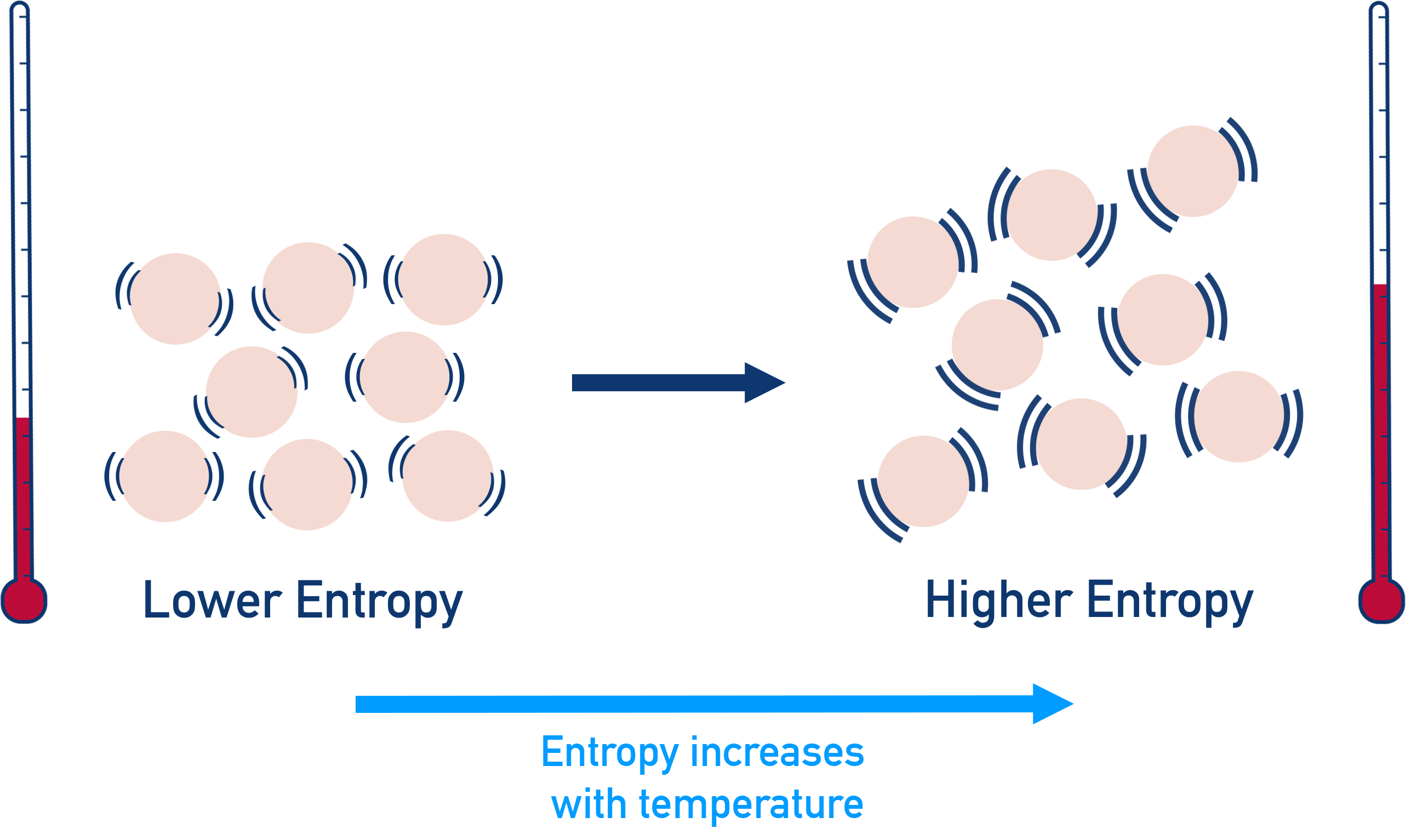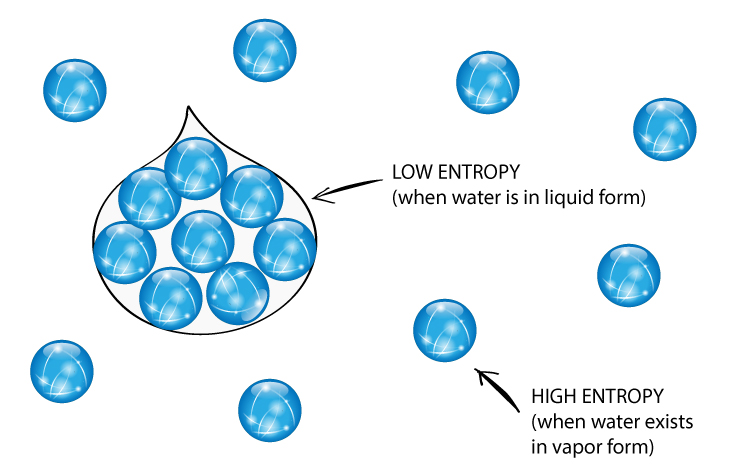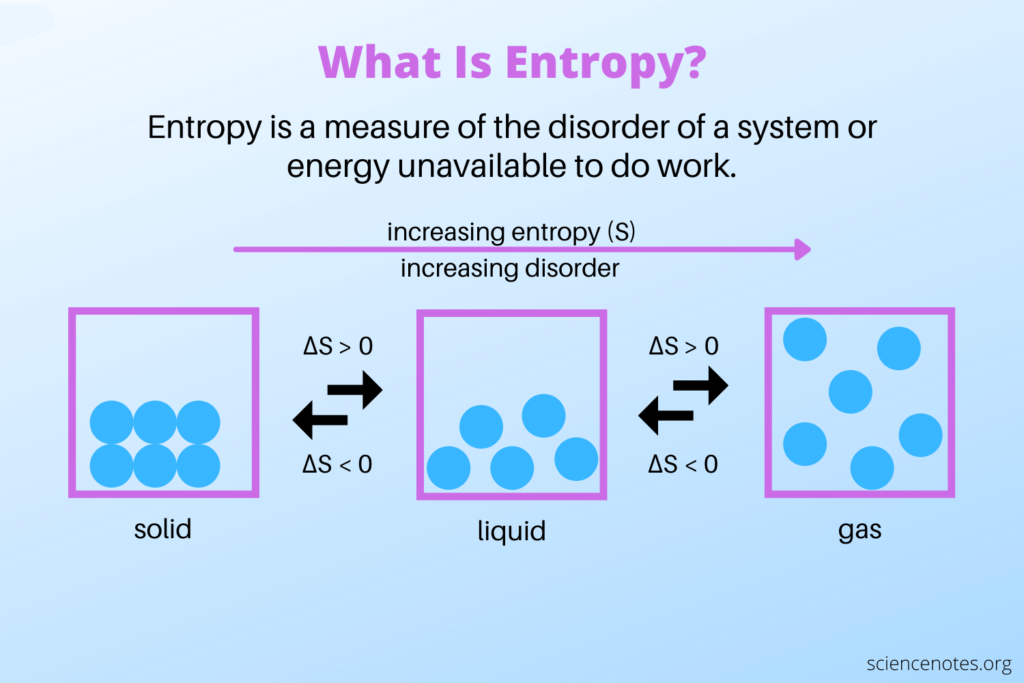
Ever wonder about the quietest corners of the universe? Or maybe you've pondered the ultimate tidiness? Well, buckle up, because we're diving into a super cool concept that sounds a bit science-y but is actually, like, totally fascinating. We're talking about entropy. Don't let the big word scare you! Think of it like this: entropy is basically a measure of messiness or randomness. The more spread out and jumbled up something is, the higher its entropy. Conversely, if things are super organized, neat, and predictable, they’ve got low entropy.
Now, imagine a cosmic game of "Which of the Following Has the Lowest Entropy?" It's like a fun quiz for your brain, making you think about how orderly things can get. We're looking for the ultimate champion of tidiness. It's not about who's the strongest or the fastest, but who's the most… well, put together! Think of your bedroom after a massive clean-up versus a hurricane hitting a toy store. Big difference, right? The cleaned bedroom has super low entropy, while the toy store aftermath? Maximum entropy!
So, what kind of contenders are we looking at in this "Lowest Entropy" showdown? We're not just talking about everyday stuff. We're venturing into the mind-bending realms of physics and the universe. It's like choosing the neatest superhero in a lineup. Is it the one who always has their cape perfectly ironed, or the one whose powers are so precise they never miss a beat?
Let's consider some potential players. We've got the perfect crystal. Picture a gemstone, all those atoms lined up in a beautiful, repeating pattern. It’s like a perfectly stacked deck of cards, where every card is in its exact spot. That’s pretty darn orderly, don't you think? There’s very little room for randomness there. Every atom knows its place. It’s the ultimate organized structure.
Then, we might have something like a supercooled liquid. This is a bit more advanced, but imagine a liquid that's been cooled way, way down, below its usual freezing point, but it hasn't actually frozen yet. It's kind of in a weird, in-between state. It's not as rigid as a solid, but it’s not as free-flowing as a normal liquid either. It's trying really hard to be organized, but it's still got a little wiggle room. So, maybe not the absolute lowest, but getting there!

And what about a black hole? Now, this one is a curveball! Black holes are known for being incredibly dense and powerful. But here's the wild part: at the heart of a black hole, all the matter that falls in is thought to be compressed into an unimaginably small point called a singularity. If everything is squeezed into one single point, where’s the messiness? Where’s the randomness? It’s like all the ingredients for a messy room are crammed into a tiny, tiny box. Some scientists argue that this extreme concentration could mean incredibly low entropy. It’s a mind-boggling idea!
To really get a grip on this, think about a perfectly shuffled deck of cards versus a brand-new, ordered deck. The brand-new deck, with all the suits and numbers in perfect sequence, has very low entropy. It's predictable. You know exactly what card is coming next. A shuffled deck, on the other hand, is a chaotic mess. Any card could be next. That’s high entropy!
So, when we ask, "Which of the following has the lowest entropy?" we're essentially asking, "Which one is the most perfectly, unbelievably organized?" It’s a quest for ultimate order. It’s like finding the most minimalist, clutter-free space in the entire universe. Imagine a perfectly silent, perfectly still room where every single molecule is in its designated spot. That’s the dream of low entropy!

Why is this so fun to think about? Because it challenges our everyday ideas of order and mess. We’re used to things getting messy over time. Your desk doesn't magically clean itself, right? But the universe, in certain extreme cases, can achieve incredible levels of order. It’s like discovering that sometimes, things can actually get neater on their own, under the right, bizarre conditions.
It’s also about appreciating the incredible forces at play in the cosmos. The ideas of perfect crystals and the mysteries of black holes push the boundaries of our imagination. They make us pause and think, "Wow, the universe is way weirder and more wonderful than I ever thought!"

This whole entropy concept, especially when we're looking for the lowest, is like a cosmic puzzle. It makes you want to learn more. You start thinking about how scientists even measure this stuff. Do they have a tiny, cosmic measuring tape for messiness? It’s the kind of question that sparks curiosity and makes you want to dig a little deeper. You might find yourself watching a documentary or reading a fun article about the universe, all because of this intriguing question about who’s the tidiest!
So, next time you see something perfectly organized – a stack of perfectly aligned books, a beautifully crafted snowflake, or even just your socks neatly paired – you can think, "Hey, that’s low entropy!" And when you hear about the extreme environments of the universe, like the intense gravity of a black hole, you can wonder about the incredible order that might exist there. It’s a fascinating peek into the fundamental workings of everything. And who doesn't love a good, orderly mystery?