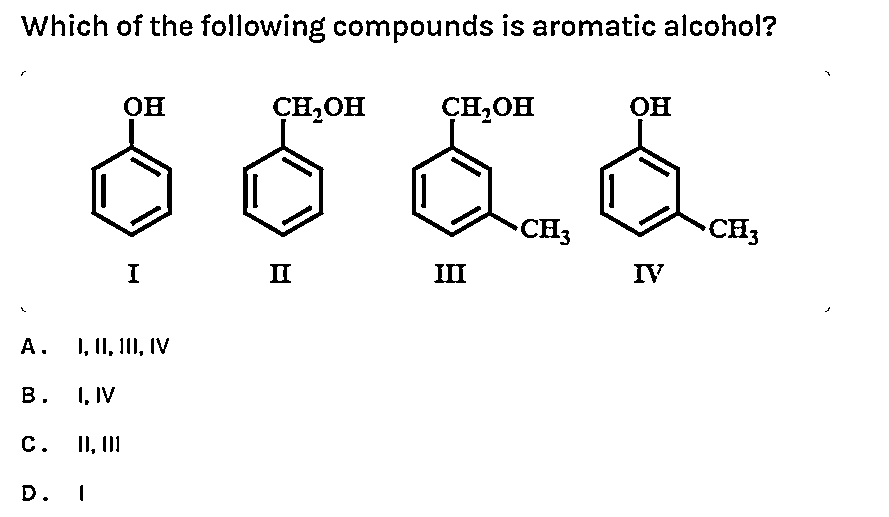
Imagine a secret club, a super exclusive clique of molecules that have all the right moves. They're not just any old compounds; these are the rockstars of the chemical world, the ones that sparkle a little brighter and smell a little sweeter. We're talking about aromatic compounds. Now, you might be thinking, "Aromatic? Like perfume?" Well, yes and no! While many aromatic compounds do have lovely smells, that's just a bonus. The real magic is in their unique structure and how they behave.
So, what’s the secret handshake to get into this elite club? It's a bit like a cosmic dance, a perfectly choreographed ballet happening at the atomic level. For a molecule to be considered aromatic, it needs to tick a few very specific boxes. Think of it as a cosmic checklist. First, it’s got to be flat, like a perfectly ironed shirt. No crumples allowed! This flatness is crucial because it allows something really special to happen with its electrons.
The second box on the checklist is a bit trickier, but let's try to make it fun. Imagine a group of friends passing a ball around in a circle. In an aromatic molecule, it's like the electrons are constantly being passed around a ring of atoms. This isn't just a casual game of catch; it’s a continuous, delocalized flow. These electrons aren’t tied down to any single atom; they’re free spirits, dancing around the entire ring. This “electron party” is what gives aromatic compounds their special stability. It’s like they’ve found the ultimate way to share and be happy!
The third, and perhaps most famous, rule of being aromatic is about the number of these free-spirited electrons. It's a very specific number: 4n + 2. Now, don't let the math scare you! Think of 'n' as a secret code. If 'n' is 0, you get 2 electrons. If 'n' is 1, you get 6 electrons. If 'n' is 2, you get 10 electrons, and so on. The key is that the number of circulating electrons must fit this formula. It's like having the perfect number of guests for a party – not too few, not too many, just right!
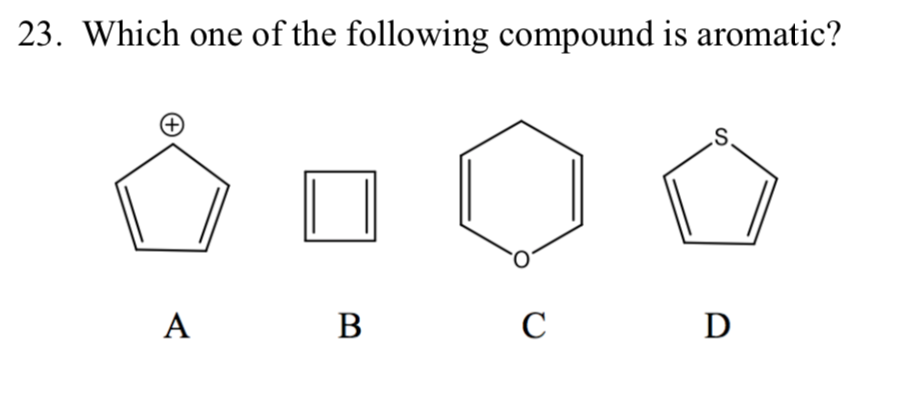
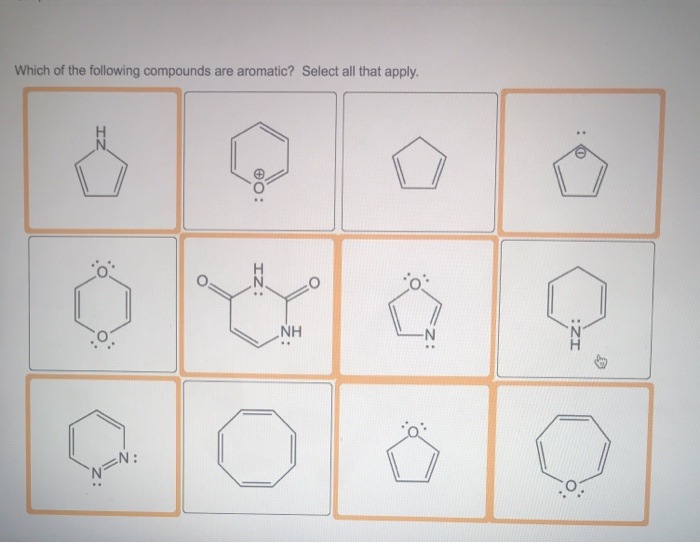
Now, let's look at a few molecules and see if they pass the test. Have you ever heard of benzene? It’s probably the most famous aromatic compound, the celebrity of the bunch. Benzene looks like a little hexagon, six carbon atoms connected in a ring, with a hydrogen atom attached to each carbon. It's as flat as a pancake and has that beautiful electron dance happening. And guess what? It has 6 electrons circulating – and 6 is exactly 4n + 2 when n=1! So, benzene is a definite yes, a founding member of our aromatic club.
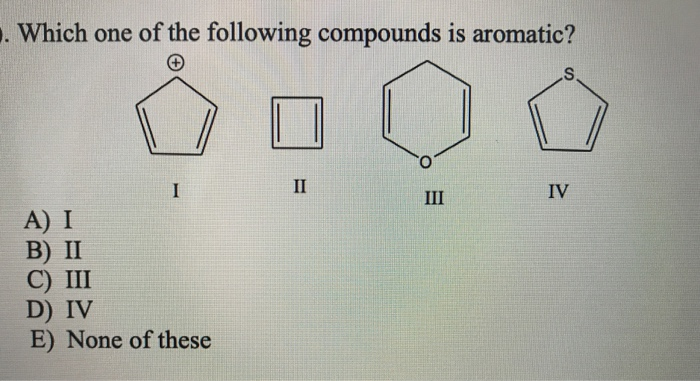
What about something a bit more complex, like naphthalene? You might know naphthalene as the stuff that used to be in mothballs. It's essentially two benzene rings stuck together. It’s still flat, and it still has that magical electron circulation. Count up the circulating electrons, and you’ll find it has 10 electrons. And lo and behold, 10 is also a number that fits the 4n + 2 rule (when n=2). So, naphthalene gets a VIP pass!
But not every ring-shaped molecule is automatically aromatic. Imagine a molecule that’s trying to look the part, but it’s a bit wobbly. It might be flat, it might even have a few extra electrons, but if they’re not circulating properly in that ring, or if the number of electrons just doesn’t fit the 4n + 2 magic number, it’s out. It's like trying to join a dance troupe without knowing the steps – you might be in the studio, but you're not part of the performance.

Let's think about cyclobutadiene. This molecule is a four-membered ring. It’s flat, and it looks like it could be aromatic. It has 4 circulating electrons. Now, if we try to fit 4 into our 4n + 2 formula, we run into trouble. There’s no whole number 'n' that works. So, despite its ring structure and circulating electrons, cyclobutadiene isn't aromatic. It’s more like the awkward cousin at the party who tries to join the dance but just can't quite get the rhythm. In fact, cyclobutadiene is notoriously unstable, a bit like a house of cards.
The beauty of aromatic compounds is that this special stability makes them incredibly important in our everyday lives. Think about the vibrant colors of your favorite dyes, the medicinal properties of many drugs, and even the delicious flavors and aromas in your food. Many of these amazing qualities come from molecules that are aromatic. They are the backbone of so many things we use and love. So, the next time you smell a beautiful flower or enjoy a tasty treat, you can thank these incredible, rule-following, electron-dancing aromatic molecules for making it all possible!