Okay, imagine you're playing with LEGOs. You've got all these tiny little bricks, right? Some are red, some are blue, some are long, some are short. You can snap them together to build anything you can dream up – a castle, a spaceship, even a whole tiny town!
Well, the universe is kind of like that, but instead of LEGOs, it's built with something even tinier and way more fundamental. These are called atoms. And honestly, thinking about them is like unlocking a secret code to how everything works. It's super cool!
So, let's dive into this awesome world of the super-small. When we ask, "Which of the following best describes an atom?", we're basically asking, "What is this fundamental building block made of?" It's like asking what makes up a LEGO brick. Is it just one solid piece of plastic? Nope!
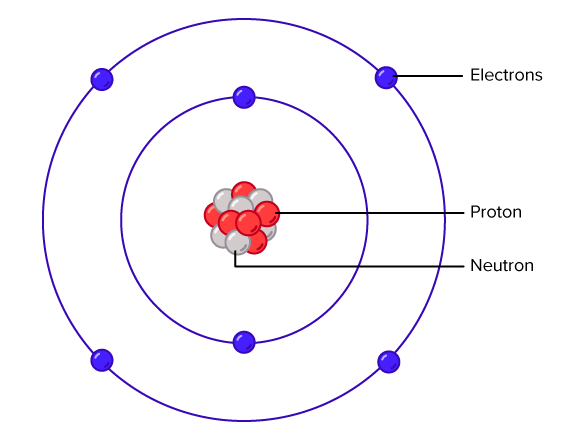
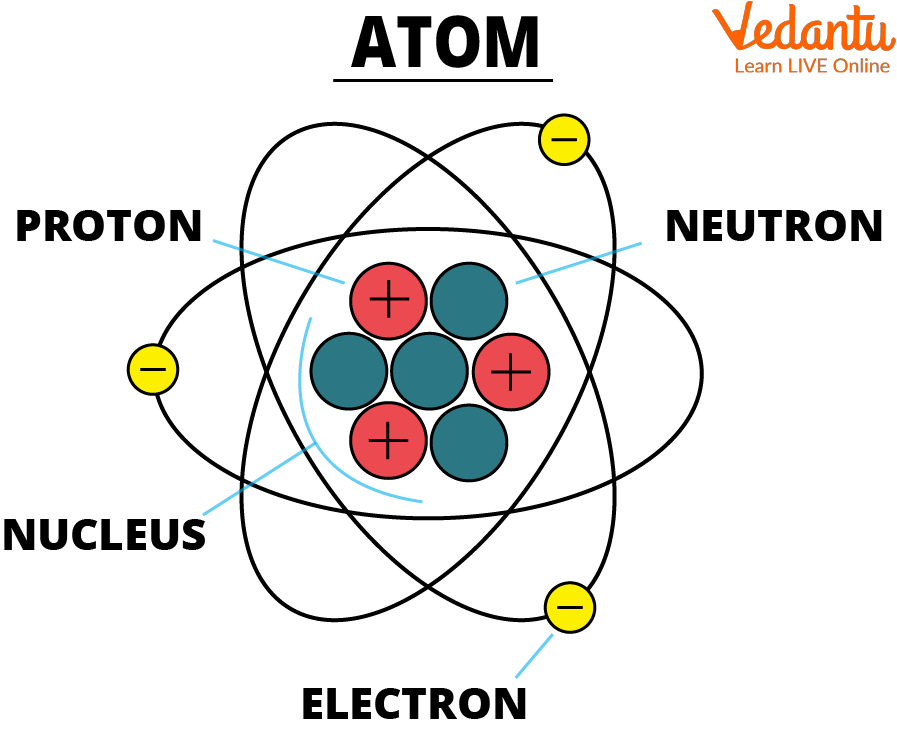
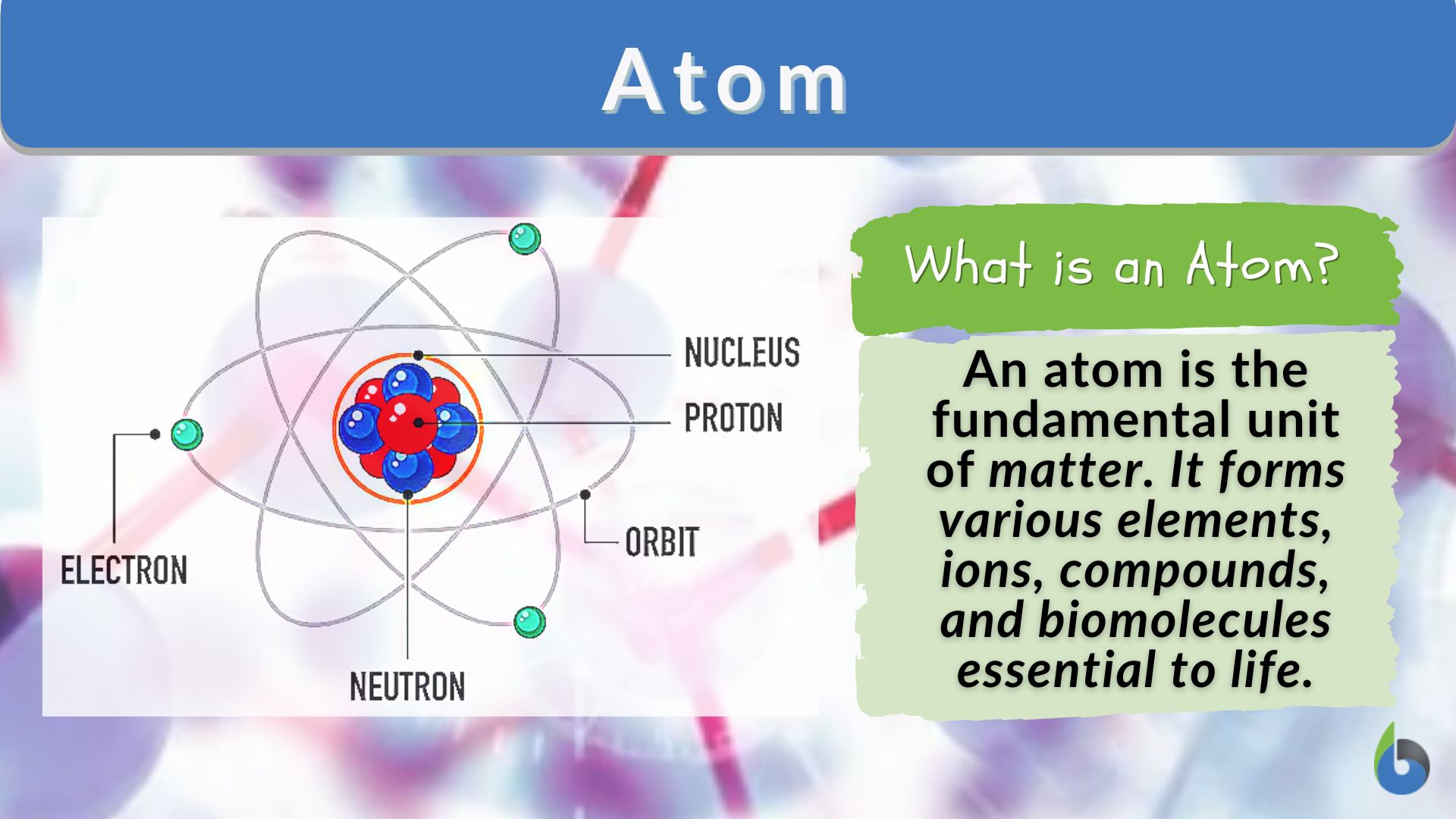
An atom isn't just a single, solid little dot. That would be way too boring! Atoms are actually made of even smaller bits. Think of it like a tiny solar system, but instead of planets orbiting a sun, you have even tinier particles zipping around a central part.
That central part is called the nucleus. It's like the atom's heart. And inside this nucleus, you find two main types of "mini-LEGOs": protons and neutrons. Protons are like the positively charged bouncy balls, and neutrons are like the neutral, chill ones. They just hang out together in the middle.

Now, orbiting around this nucleus, at what feels like lightning speed, are even tinier things called electrons. Electrons are super important. They have a negative charge, and they're constantly on the move. They're like tiny, energetic cheerleaders for the atom, buzzing around and making things happen.
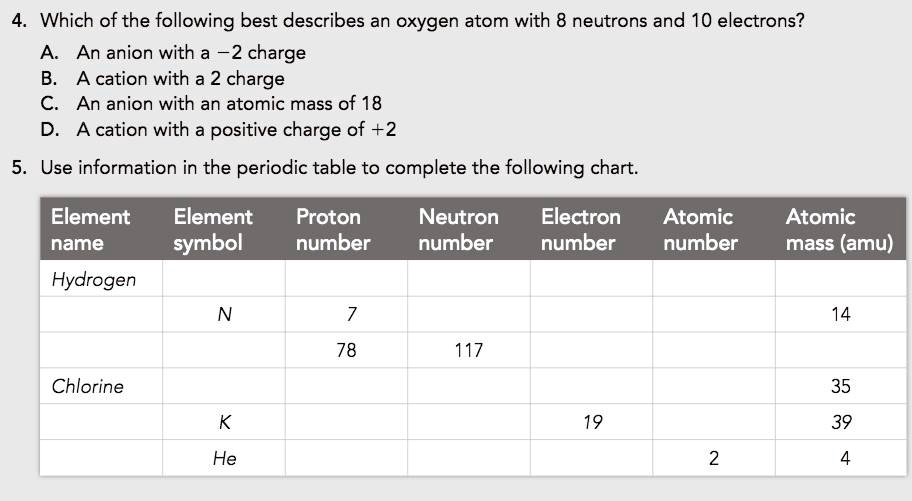
So, when you hear about different types of atoms, like hydrogen or oxygen, what's really different? It's usually the number of protons in that central nucleus! That's their secret identity. A hydrogen atom has one proton. An oxygen atom has eight. It's like having a LEGO set with a specific number of red bricks – that's what makes it "red."
The amazing part is, these simple combinations of protons, neutrons, and electrons can create everything we see. From the air we breathe to the phone you're reading this on, it's all just different arrangements of these atomic building blocks. Isn't that mind-blowing? It’s like a universe-sized LEGO set!

Think about it: You can have a bunch of oxygen atoms join up with some hydrogen atoms, and poof – you get water! It’s like snapping LEGO bricks together in a specific pattern to build a car. The same basic pieces, just arranged differently, create completely new things.
And the way these atoms interact is what makes the world so dynamic and interesting. Those electrons? They're the life of the party. They can jump from one atom to another, or share themselves, forming these amazing connections called bonds. These bonds are what hold everything together.

When atoms bond, they can create bigger structures called molecules. So, a water molecule is basically two hydrogen atoms holding hands (electronically speaking!) with one oxygen atom. It’s a tiny, stable team.
This whole concept of atoms being made of protons, neutrons, and electrons, with electrons zipping around a nucleus, is the most widely accepted and best description. It’s the foundation of chemistry and physics, and it’s surprisingly fun to think about.
It’s like realizing that the entire world, from the stars in the sky to your own two feet, is built from these incredibly tiny, buzzing, dancing particles. It makes you feel like a cosmic detective, uncovering the secrets of the universe, one atom at a time!
SOLVED: Question 1 Which of the following best describes an orbital
So, when you see a question about what an atom is, remember the tiny solar system, the LEGO bricks, and the energetic electrons. It’s not just a boring science word; it's the key to understanding the magic of existence itself. It’s the building block of everything, and that’s pretty spectacular, don’t you think? It's a little bit of everyday magic, happening all around you, all the time. And once you start noticing it, you'll see it everywhere!
This idea that everything is made of these fundamental units, that they have this internal structure, is what truly sets atoms apart. They aren't just random bits; they have a specific, organized way of being. And that organization is what leads to all the diversity and wonder we experience. It's a bit like a perfectly crafted recipe – the right ingredients, in the right amounts, with the right preparation, create something delicious. Atoms are the universe's ultimate recipe ingredients!
The beauty of the atom lies in its simplicity and its complexity all at once. It's a simple concept – a nucleus with electrons – but the sheer number of ways these can combine and interact is mind-boggling. It’s an endless playground for discovery, and it all starts with understanding these fundamental particles. So, next time you look at anything, just remember it's all a grand, intricate dance of these tiny, fundamental dancers.