Have you ever thought about the super chill elements on the periodic table? We're talking about the Noble Gases. They're a special group, kind of like the cool kids who hang out on their own. You've probably heard of Helium, the stuff that makes your voice squeaky at parties. Or maybe Neon, lighting up those super bright signs. But there's a whole bunch more, and they're all pretty interesting.
These guys are called "noble" because they're not very reactive. They like to keep to themselves, which is pretty unique in the wild world of chemistry. Most elements are always looking to bond with others, like buddies who can't be without each other. Noble gases? Not so much. They're perfectly happy being single atoms. It's like they have their own little bubble.
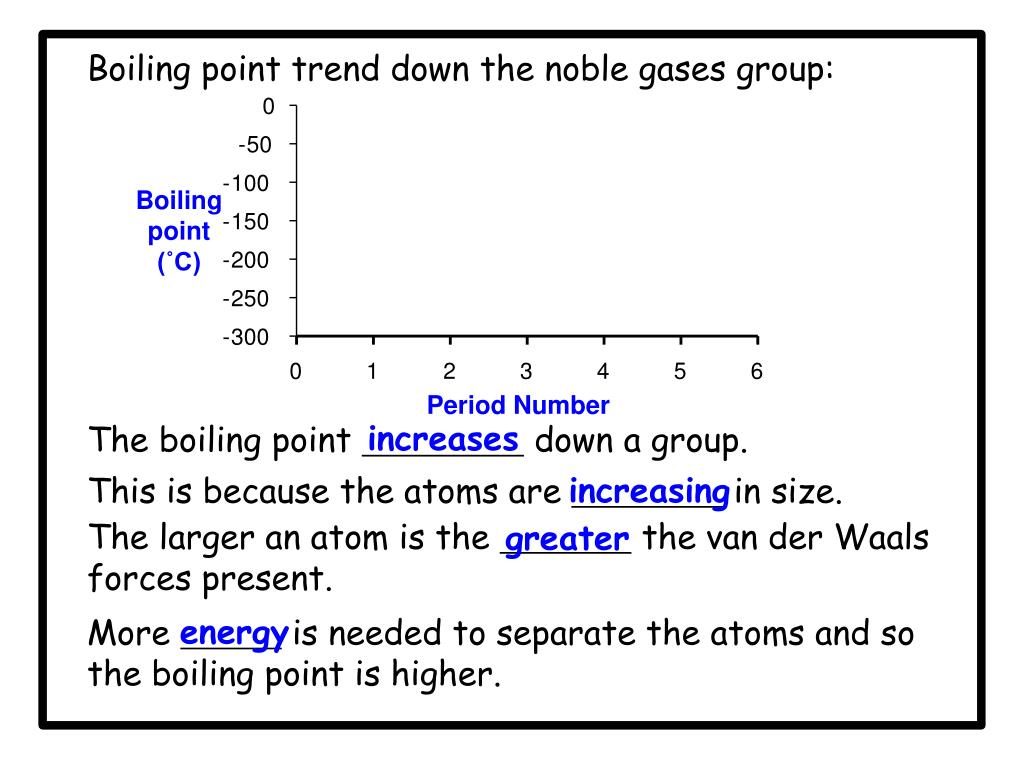
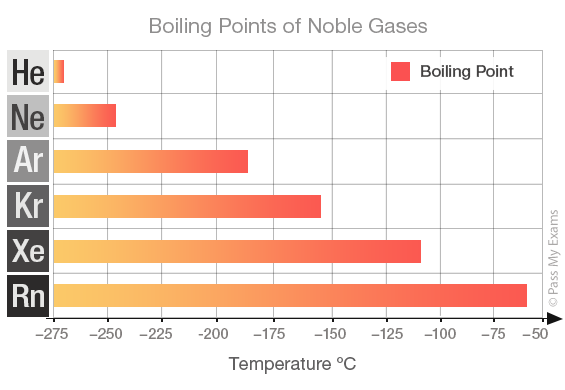
But here's where things get really cool. Even though they're all noble and independent, they have their own personalities. And one of the most fun ways to see their differences is by looking at their boiling points. Think of boiling point as the temperature at which something goes from being a liquid to a gas. For these gases, it's how cold they have to get to actually become a liquid in the first place. Pretty mind-bending, right?
So, the big question is: which of these cool cats has the highest boiling point? Which one needs the most effort to get it to stop being a gas and start being a liquid? It's like a competition to see who can stay a gas the longest, or, the opposite, who's the easiest to get to chill out and become a liquid.
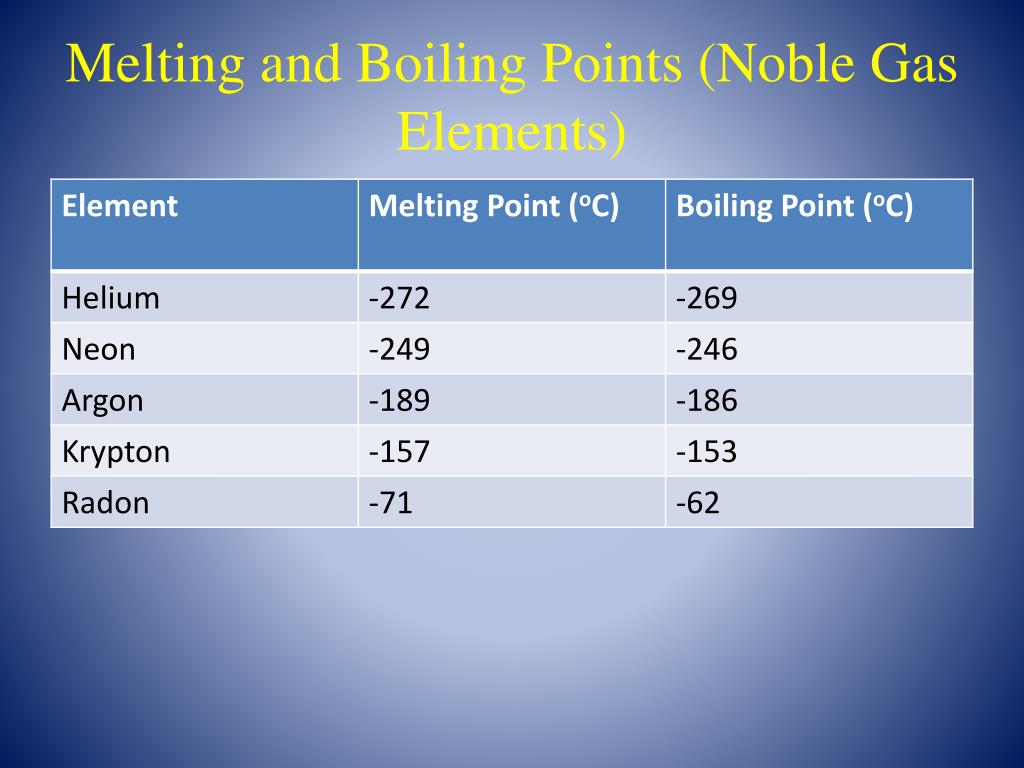
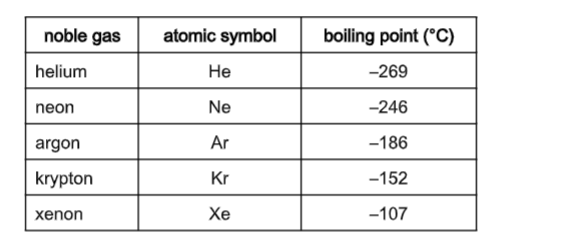
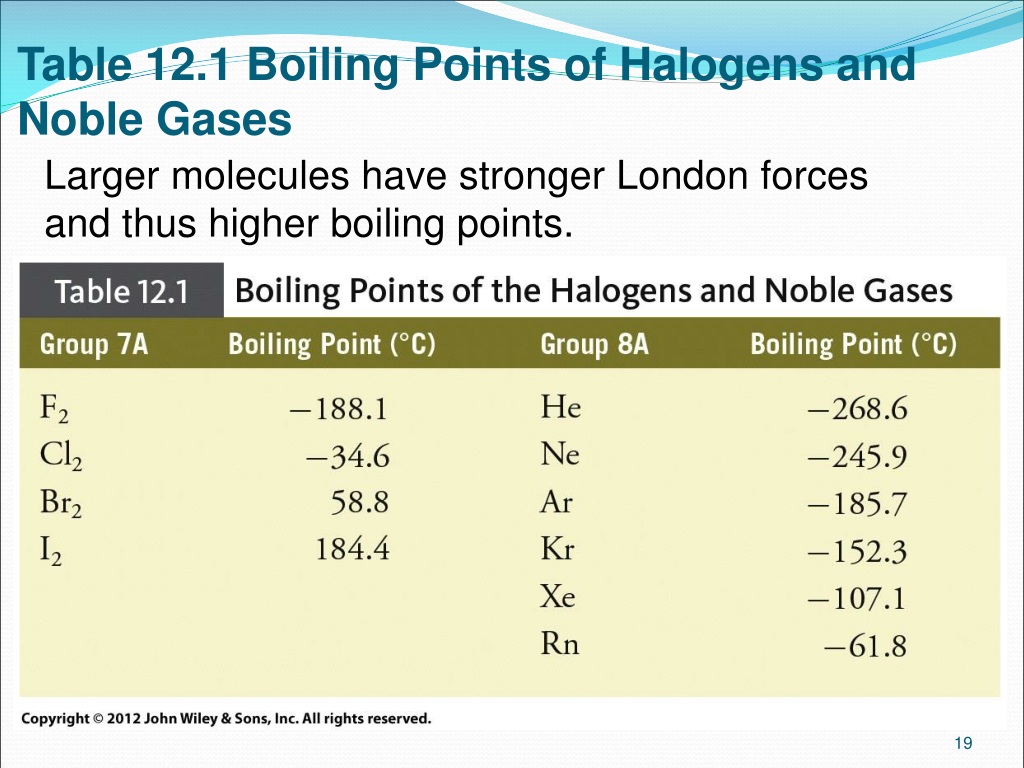
Let's go down the list, shall we? We start with Helium. This little guy is so laid-back, its boiling point is incredibly low. We're talking about -268.9 degrees Celsius! That's colder than a polar bear's toenails in January. You need some serious chill to even get Helium to be a liquid. It's the ultimate free spirit of the noble gas family.
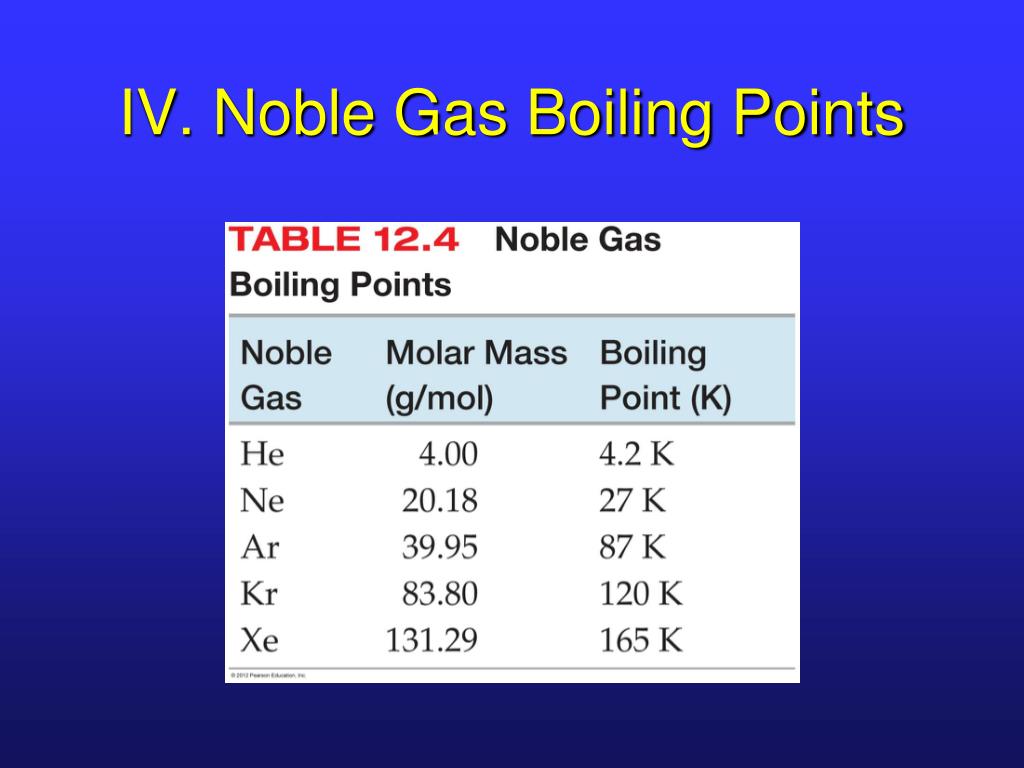
Then we have Neon. This one is a bit more of a homebody than Helium. Its boiling point is a tad higher, around -246.1 degrees Celsius. Still super, super cold, but just a little bit easier to manage than Helium. It's like Neon is willing to hang out in a slightly less frigid environment.
Next up is Argon. You might have heard of this one. It's actually the third most abundant gas in our atmosphere! Argon's boiling point is a bit warmer, about -185.9 degrees Celsius. It's like it's starting to feel a little more comfortable in the temperature range we humans can actually, you know, imagine. It's still freezing, but compared to Helium, it's practically a tropical vacation.
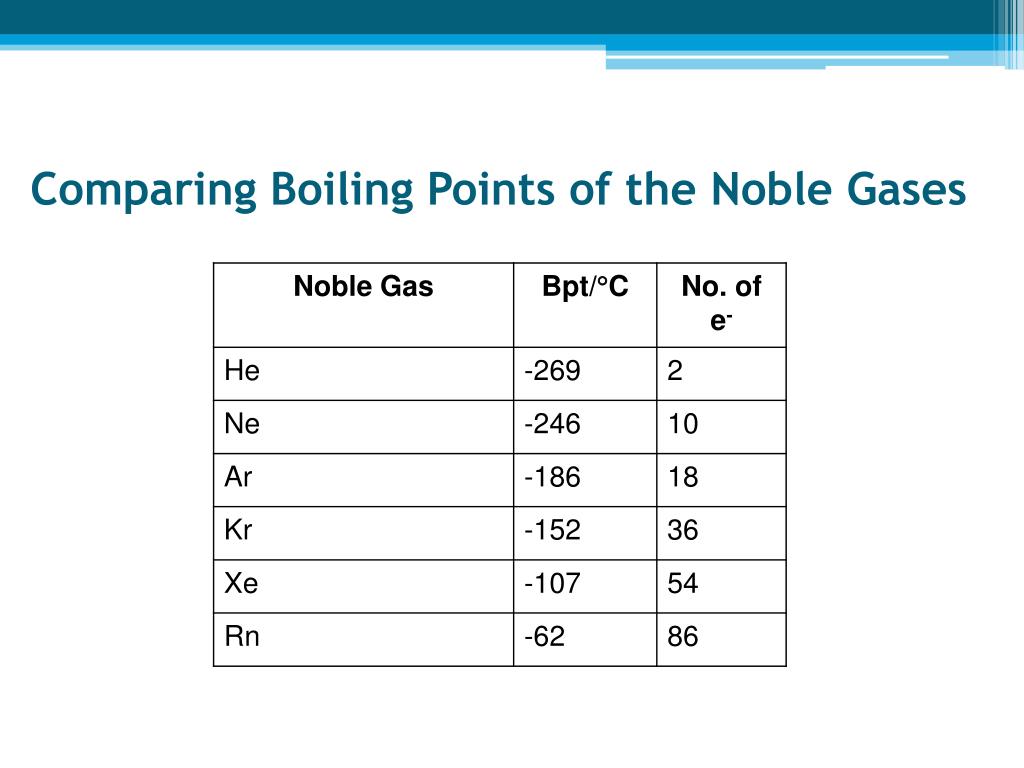
After Argon comes Krypton. Now, this one is getting serious. Krypton has a boiling point of -153.4 degrees Celsius. This is where things start to feel... well, not exactly warm, but definitely less extreme. Krypton is a bit more willing to join the liquid party. It's like it's saying, "Okay, fine, I'll come in from the absolute freezing cold."
And then there's Xenon. Ah, Xenon! This noble gas is really starting to show its personality. Its boiling point is -108.1 degrees Celsius. This is a significant jump up from the others. Imagine how much easier it is to get something to become a liquid at -108 degrees compared to -268 degrees. Xenon is like the friend who's always up for a slightly less extreme adventure.
But the champion, the ultimate king of the chill, the noble gas with the highest boiling point is... drumroll please... Radon!

Yes, Radon! This invisible, odorless gas has a boiling point of -61.7 degrees Celsius. That might still sound incredibly cold to us, but in the world of noble gases, it's practically balmy!
Think about it. While Helium needs to be colder than the surface of Pluto to even think about being a liquid, Radon is perfectly happy to chill out and become a liquid at a temperature that, while still freezing, is much more "manageable" on the cosmic scale of cold. It's like Radon is saying, "You know what? I can handle a little bit of this temperature. Let's do this!"

What makes Radon so special? Well, it's also radioactive, which adds a whole other layer of intrigue. But focusing on its boiling point, it's this slight difference in how the atoms interact with each other that makes Radon the highest achiever in the boiling point department. Even though they're all noble gases, they aren't exactly the same. There are these tiny, tiny differences in how their electrons are arranged and how they "feel" each other's presence.
It's these subtle interactions, like a gentle nudge or a fleeting glance between atoms, that determine how much energy (in the form of heat) it takes to make them change from a liquid to a gas. For Radon, these "nudges" are just a little bit stronger, meaning you need more heat to get it to break free and become a gas. It's like it has a slightly tighter grip on its liquid state.
So, the next time you think about noble gases, remember Radon. It's the one that's a bit more willing to take a chill pill. It's the one that surprises us with its ability to be a liquid at "higher" temperatures. It’s a testament to how even in the most seemingly unchanging families, there are always fascinating differences waiting to be discovered. Isn't science just the coolest? You might even find yourself curious enough to look up some pictures of what these elements look like as liquids. It's a whole different world out there!