Hey there, science nerds and curious cats! Let's dive into something that sounds super serious but is actually kinda cool. We're talking about atoms, those tiny building blocks of everything. And specifically, we're going to figure out which neutral atom is playing dress-up with a grumpy, positively charged manganese ion. Mind. Blown. (Okay, maybe not blown, but definitely tickled.)
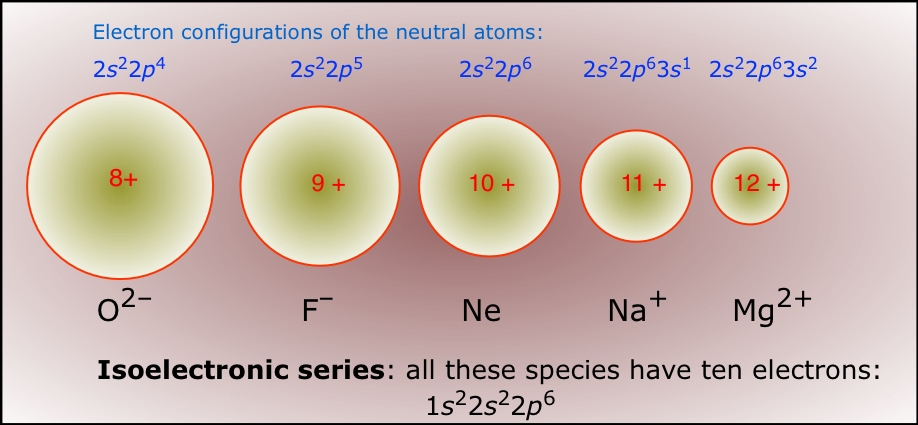
So, the question is: which neutral atom is isoelectronic with Mn+4? Sounds like a mouthful, right? Don't sweat it. Isoelectronic just means they have the same number of electrons. Think of it like two people wearing the exact same outfit. They look different on the outside, but underneath all the bling (or in this case, the protons and neutrons), they're rocking the same electron swag.
Let's Break Down the Mystery!
First up, we've got Mn. That's manganese, folks! It's a metal, kinda important for your health (ever heard of supplements?), and it's pretty reactive. In its neutral state, a manganese atom has 25 protons. And because atoms are usually chill and balanced, a neutral atom has the same number of electrons as protons. So, neutral manganese = 25 electrons.
Now, the plot thickens. We're not dealing with chill, neutral manganese. We're dealing with Mn+4. That little +4 superscript is a big deal. It means this manganese atom has lost 4 electrons. Yep, it's gotten rid of some of its electron buddies. So, if it started with 25 electrons and lost 4, how many does it have now? Do the math: 25 - 4 = 21 electrons.
So, our goal is to find a neutral atom – meaning it has no charge, and therefore, its number of electrons equals its number of protons – that also has exactly 21 electrons. Easy peasy, lemon squeezy!
The Electron Counting Game
We're looking for an atom with 21 electrons. Since it's neutral, it must also have 21 protons. Now, who's rocking 21 protons on the periodic table? Time for a little peek. The periodic table is basically a superhero roster of elements, each with its own atomic number (which is just the proton count). We just need to find the element with atomic number 21.

Drumroll, please... it's Scandium! Yep, Sc. Scandium has 21 protons. And as a neutral atom, it also has 21 electrons. Boom! We found our match. So, Scandium (Sc) is isoelectronic with Mn+4.
Pretty neat, huh? It's like finding out your favorite celebrity secretly has the same favorite ice cream flavor as you. It’s a small world, even at the atomic level!
Quirky Facts and Fun Details
Let's talk about manganese for a sec. Did you know it's named after a district in Greece called Magnesia? And guess what else is named after Magnesia? Magnesium! Coincidence? Or maybe they were just really into that area back in the day. The ancient Greeks probably were like, "This place is so full of cool stuff, let's name some elements after it!"
Manganese is also a pretty important element for life. It helps with metabolism and bone health. So, next time you eat something healthy, give a little nod to manganese. It's working hard for you!
Now, what about our hero, Scandium? It's not as famous as manganese, but it's still got some cool stuff going on. It's a silvery-white metal, and it’s often used in alloys, especially with aluminum, for things like airplane parts. Imagine your plane flying high because of a metal that shares its electron count with a grumpy manganese ion. That's some serious intergalactic (or at least, inter-elemental) teamwork!
One particularly quirky thing about Scandium is that it's relatively rare in the Earth's crust. It's like the introvert of the periodic table – not everywhere, but when you find it, it's pretty special. And it's often found in the same ore deposits as rare earth elements, so it hangs out with some pretty exclusive company.

Why Is This Stuff Fun?
Okay, so why should you care about isoelectronic atoms? Because it’s a little puzzle! It’s like a secret code that scientists use to understand how atoms behave. When atoms have the same electron configuration, they often share similar chemical properties. This means they might react in similar ways, or form similar types of bonds.
Think of it like this: if two cars have the exact same engine, they're probably going to drive and perform pretty similarly, even if they have different paint jobs and fancy spoilers. In the same way, if two atoms (or ions) have the same electron arrangement, they'll behave in similar chemical ways. This is super important for chemists and material scientists who are trying to create new materials or understand how existing ones work.
It’s also a great way to appreciate the order and patterns in the universe. The periodic table isn’t just a random collection of elements; it’s a beautifully organized system. Understanding concepts like isoelectronic species helps us see those deeper connections and appreciate the elegance of chemistry.

Plus, let's be honest, it sounds smart. Dropping "isoelectronic" into a conversation might just impress your friends. Just be ready to explain it (or just smile mysteriously). You can even make up your own isoelectronic pairings and pretend you're a chemistry genius. "Oh yeah, that muffin looks isoelectronic with a unicorn horn. Totally the same number of sprinkles." (Okay, maybe don't do that one.)
The Takeaway
So, to recap: Mn+4 has lost 4 electrons, leaving it with 21 electrons. We’re looking for a neutral atom with 21 electrons. That atom has 21 protons. And the element with 21 protons is Scandium (Sc). Therefore, neutral Scandium is isoelectronic with manganese that has lost 4 electrons. They're basically electron twins, separated at formation!
It’s a simple concept, really, but it unlocks a whole world of understanding about how atoms interact. It’s a peek behind the curtain of the atomic world, where things are much more organized and interconnected than you might think.
So, the next time you see manganese or scandium mentioned, you’ll know they have a secret electron-based connection. Science is full of these fun little surprises, and uncovering them is what makes learning so darn enjoyable. Keep asking questions, keep exploring, and who knows what other isoelectronic friendships you might discover!