Ever wondered what makes different substances behave the way they do? Why is water a liquid at room temperature while oxygen is a gas? Or why does oil and water refuse to mix, no matter how hard you shake them? The answer, my friends, lies in the fascinating world of intermolecular forces! These invisible tug-of-wars between molecules are the unsung heroes of chemistry, dictating everything from boiling points to solubility. And today, we're diving into the specific case of carbon tetrachloride, or CCl4, to uncover which of these molecular attractions are at play. It might sound a bit technical, but trust us, it's a peek behind the curtain of the everyday!
Understanding intermolecular forces isn't just for scientists in lab coats; it's surprisingly useful and can explain a lot about the world around us. For instance, knowing these forces helps us design new materials, understand how drugs dissolve in our bodies, and even develop better cleaning agents. It’s like having a secret decoder ring for the physical properties of matter!
The Molecular Tango of CCl4
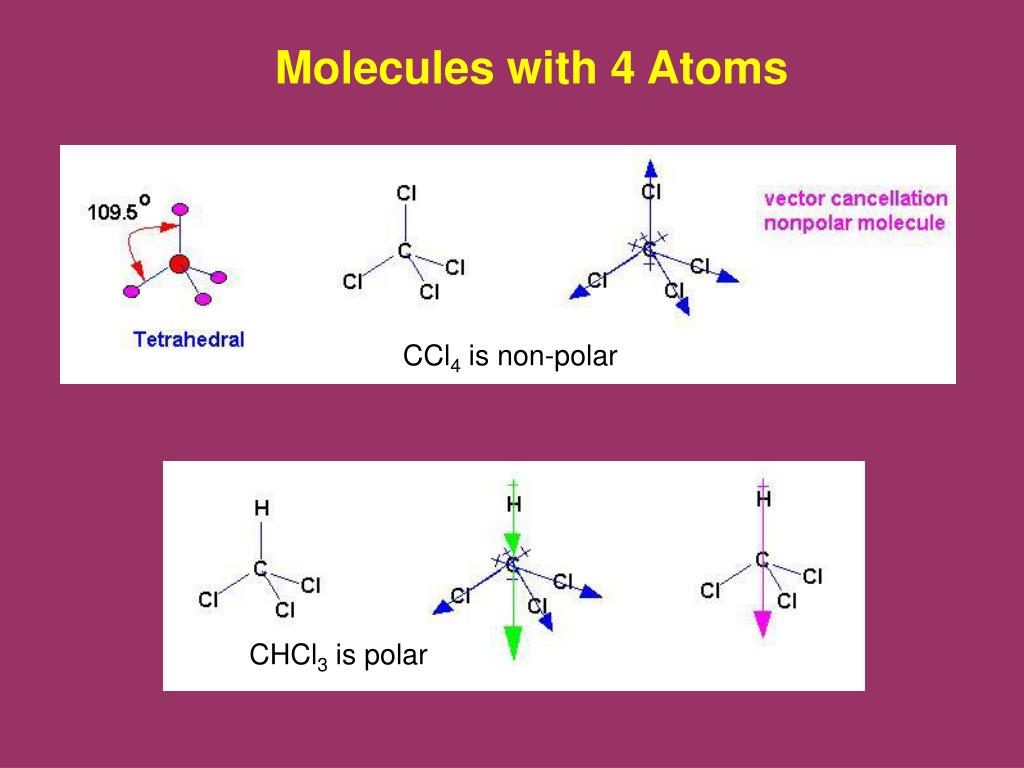
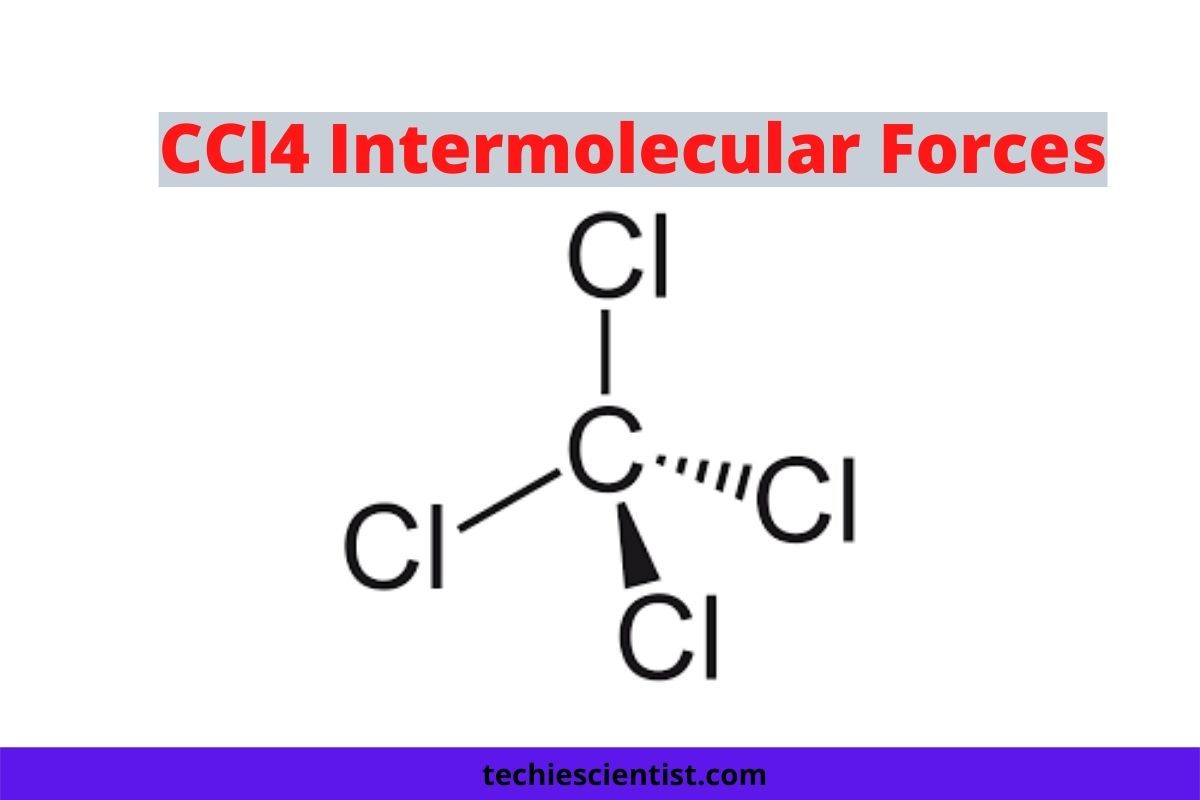
So, what exactly is CCl4? It’s a molecule made of one central carbon atom bonded to four chlorine atoms. On its own, this molecule is pretty symmetrical. Imagine a carbon atom at the center of a tetrahedron, with a chlorine atom at each corner. This neat, balanced arrangement is key to understanding its intermolecular forces. The carbon-chlorine bonds themselves are polar, meaning the electrons in the bond are pulled slightly more towards the chlorine atoms (because chlorine is more "electronegative"). This gives the chlorine atoms a slight negative charge and the carbon atom a slight positive charge.
However, because the molecule is so symmetrical, these individual bond polarities cancel each other out. It’s like having four people pulling in perfectly balanced directions; the net result is no movement. This means that CCl4 as a whole molecule is nonpolar. This is a crucial piece of information!
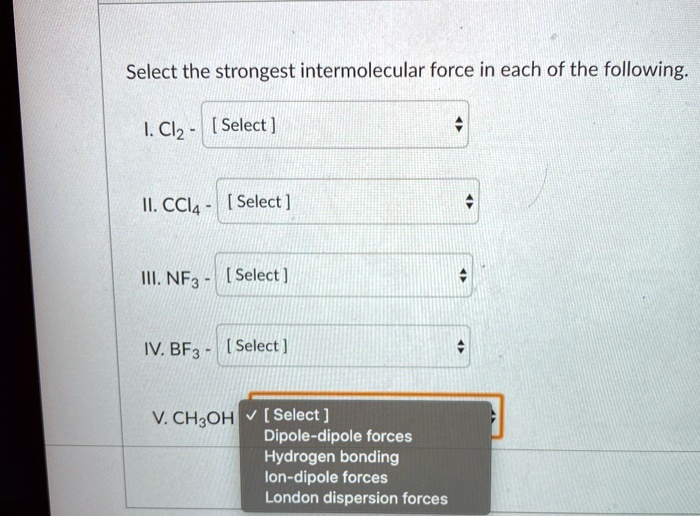
Now, let's talk about the stars of our show: the intermolecular forces. For CCl4, the primary, and in fact, the only significant intermolecular force present is the London dispersion force, also known as van der Waals forces or induced dipole-induced dipole forces. Let’s break down why this is the case and what it means.
London Dispersion Forces: The Universal Charmers
London dispersion forces are the weakest type of intermolecular force, but they are present in all molecules, polar or nonpolar. They arise from temporary, fleeting fluctuations in electron distribution within a molecule. Even in a nonpolar molecule like CCl4, the electrons are constantly moving. At any given moment, there might be a slightly higher concentration of electrons on one side of the molecule, creating a temporary, instantaneous dipole. This temporary dipole can then induce a similar, temporary dipole in a neighboring molecule. Think of it like a ripple effect. These temporary attractions, while weak individually, can add up, especially in larger molecules.
For CCl4, with its symmetrical structure and lack of permanent dipoles, the London dispersion forces are the only game in town. There are no permanent positive and negative ends to attract other molecules (like in polar molecules with dipole-dipole interactions), and it doesn't involve hydrogen bonded to a highly electronegative atom like oxygen, nitrogen, or fluorine (which would lead to hydrogen bonding).
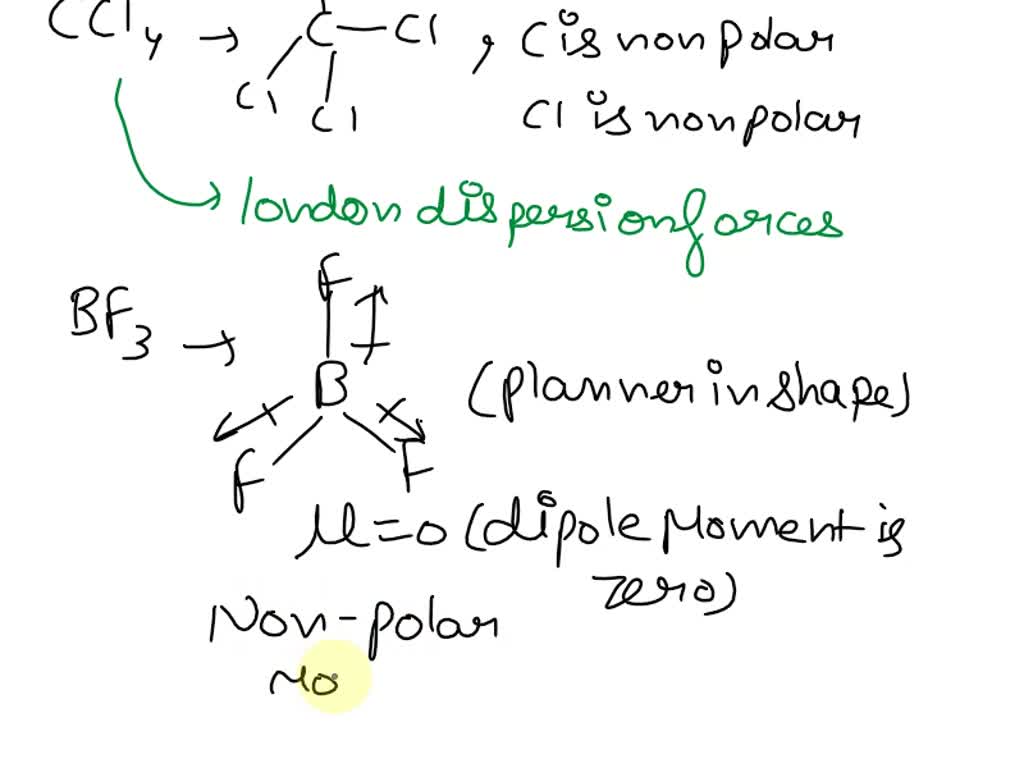
The key takeaway is that because CCl4 is a symmetrical, nonpolar molecule, the primary intermolecular forces holding its molecules together are London dispersion forces.
Why This Matters (Beyond Just Chemistry Class!)
So, what are the practical implications of CCl4 being held together solely by London dispersion forces? Well, these forces are generally quite weak, which means molecules that only have these forces tend to have lower melting and boiling points compared to similar-sized molecules with stronger intermolecular forces (like dipole-dipole or hydrogen bonding). CCl4 is a liquid at room temperature with a boiling point of about 76.7°C, which is relatively low when you consider other compounds of similar molecular weight.
Furthermore, the "like dissolves like" rule comes into play here. Because CCl4 is nonpolar, it will tend to dissolve well in other nonpolar solvents (like hexane or benzene) but will not mix well with polar solvents (like water). This is why oil and water don't mix – oil is nonpolar, and water is very polar. CCl4 exhibits similar behavior.
Historically, CCl4 was used as a cleaning solvent and a fire extinguisher due to its nonpolar nature and relatively low reactivity. However, it’s now known to be toxic and harmful to the environment, so its use has been significantly restricted. This is another example of how understanding molecular behavior, driven by intermolecular forces, impacts our decisions about chemical safety and sustainability.
In essence, by looking at the structure of CCl4 and recognizing its symmetry, we can predict and understand its physical properties. The seemingly simple concept of electron movement leads to the ubiquitous London dispersion forces, which are the sole architects of the attractions between CCl4 molecules. It’s a beautiful illustration of how the microscopic world of molecules dictates the macroscopic properties we observe every day!