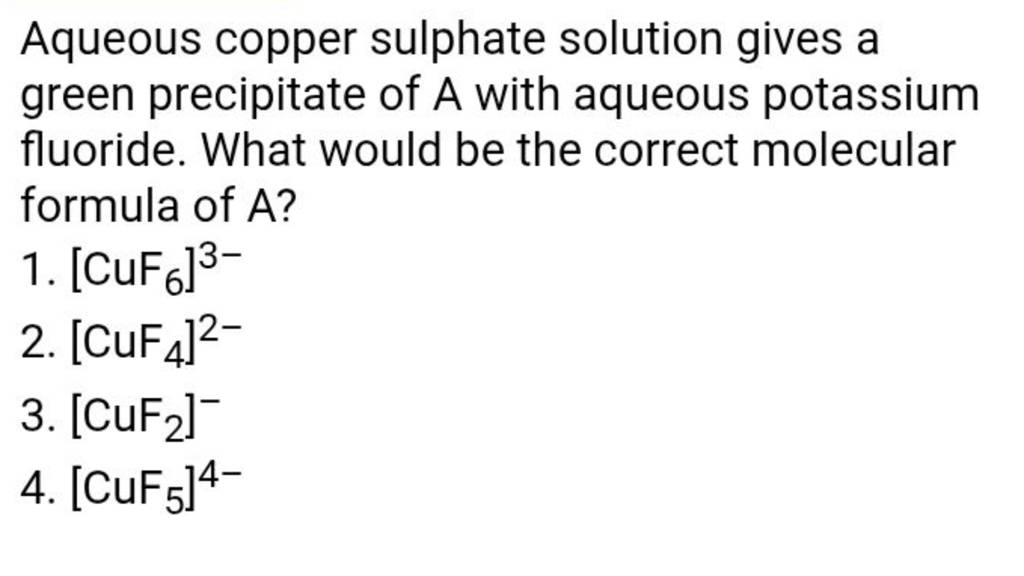Ah, who doesn't love a touch of magic in their everyday lives? Whether it's whipping up a vibrant smoothie, creating a mesmerizing science experiment for the kids, or even just adding a splash of color to your crafting projects, the world of chemistry and the transformations it offers are endlessly fascinating. There's a certain delight in seeing the ordinary turn extraordinary, and one of the most visually appealing of these transformations involves a compound that, when dissolved in water, paints a beautiful and distinct green aqueous solution. It’s a color that evokes nature, tranquility, and even a hint of mystery, and its presence can signal some really interesting chemical happenings.
So, what exactly is this alchemist's secret? When we talk about a compound forming a green aqueous solution, one of the most common and recognizable culprits is copper(II) sulfate. You might know it by other names, like bluestone or blue vitriol, but when it's properly prepared and dissolved, the characteristic green hue emerges. Now, before you imagine complicated lab equipment, let's talk about why this might matter to you in your daily life. Copper(II) sulfate isn't just for show; it has some practical applications that are more relevant than you might think. For instance, it's widely used as a fungicide and algaecide. This means it can be found in solutions to treat swimming pools, keeping them clear and free from slimy green growth. It's also used in agriculture to protect crops from fungal diseases, contributing to the food we eat.
Beyond its practical uses, the visual appeal of a green copper sulfate solution is undeniable. For budding scientists, it's a fantastic way to explore the principles of solubility and chemical change. Imagine setting up a simple science experiment at home. All you need is some copper(II) sulfate powder (available from some garden supply stores or online) and water. Dissolving it can be a captivating experience. You can observe how the color intensifies as you add more powder, or how it might change if you introduce other substances. It’s a hands-on lesson that sticks, making learning fun and engaging for all ages. For crafters, that lovely green can be a unique dye for fabrics or a component in homemade bath bombs, adding a touch of sophisticated color to your creations.
To get the most out of your experience with a green aqueous solution, especially if you're experimenting, a few tips can help. Firstly, safety first! Copper(II) sulfate is a chemical, so always handle it with care. Wear gloves and avoid ingestion or prolonged skin contact. Secondly, the concentration matters. A dilute solution will be a paler green, while a more concentrated one will be a richer, deeper emerald. Experimenting with different amounts can show you this effect beautifully. Thirdly, consider the container. A clear glass or plastic container will best showcase the vibrant green. If you're doing a science project, a beaker or a clear jar works wonders. Finally, observe and document. What do you notice? How does the light reflect off it? Does it change over time? Taking notes or pictures can enhance your learning and appreciation.
So, the next time you see a beautiful green liquid, whether it’s in a swimming pool, a garden spray, or a science demonstration, you’ll have a better idea of the fascinating compound responsible for that lovely hue. It’s a simple yet elegant display of chemistry that brings a splash of color and a hint of usefulness into our world.