
Okay, confession time. I have a secret pet peeve. It’s something totally harmless, completely niche, and probably makes zero sense to anyone who isn't me. But here we are.
We’re talking about hydrocarbons. Yeah, those things from chemistry class. You know, the ones that are basically chains of carbon and hydrogen atoms. Super simple, right?
Most of the time, they’re straight lines. Like a perfectly aligned row of dominoes. Or a string of tiny beads on a thread. Very orderly. Very predictable.
But then, things get… interesting. What happens when that perfectly straight chain decides to get a little… wobbly? When it decides to take a detour?
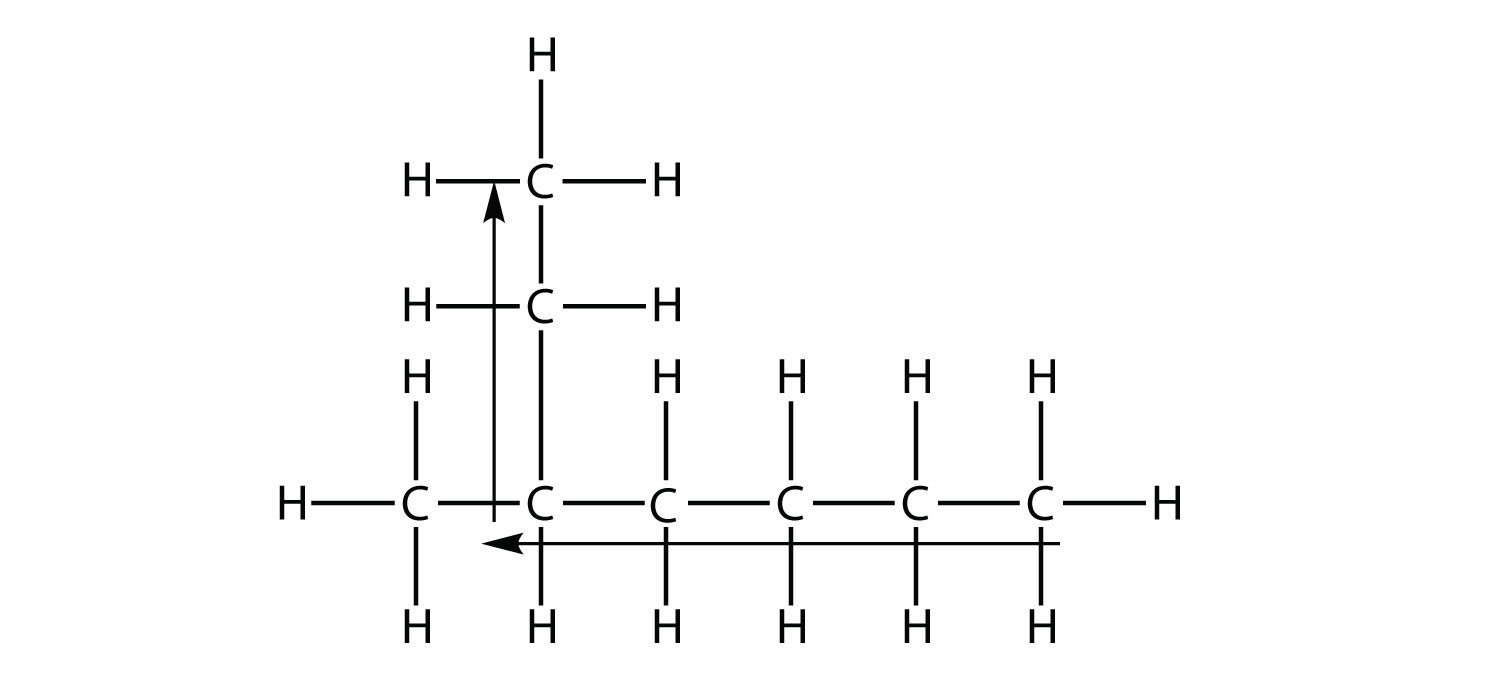
When a hydrocarbon chain is bent, it is called… well, it’s called something. And that something is where my quiet, internal debate lives.
The Straight and Narrow (and Boring)
Let's appreciate the beauty of a straight hydrocarbon for a moment. We’ve got methane, ethane, propane, butane. They just keep going, carbon after carbon, hydrogen clinging on like happy little passengers. It’s elegant in its simplicity.
Think of it like a perfectly straight road stretching out before you. No curves, no surprises. Just pure, unadulterated linearity. It gets the job done, no doubt.
But honestly? It’s a bit… plain. Like vanilla ice cream. Good, reliable, but not exactly setting your taste buds on fire.
Enter the Bendy Boys
Now, imagine that straight chain gets a bit restless. It’s been staring at the same straight path for too long. It wants to explore. It wants to see what’s around the corner.

So, it does what any sensible, adventurous chain would do. It bends. It loops. It gets a little… coiled.
And that, my friends, is where the magic, and my mild obsession, begins.
The Name Game: A Subjective Saga
So, when a hydrocarbon chain is bent, it is called… a cycloalkane.
Yes, I know. It sounds very official. Very scientific. Very… like something a professor would say with a stern look.
But here’s my unpopular opinion: it feels a little… underwhelming?
I mean, think about it. We have these perfectly straight chains, diligently marching along. And then, one of them gets a bit cheeky, takes a little bend, and suddenly it’s burdened with this long, somewhat intimidating word.
It’s like calling a dog that wags its tail a “posterior caudal appendage agitator.” It’s technically correct, but where’s the fun in that?
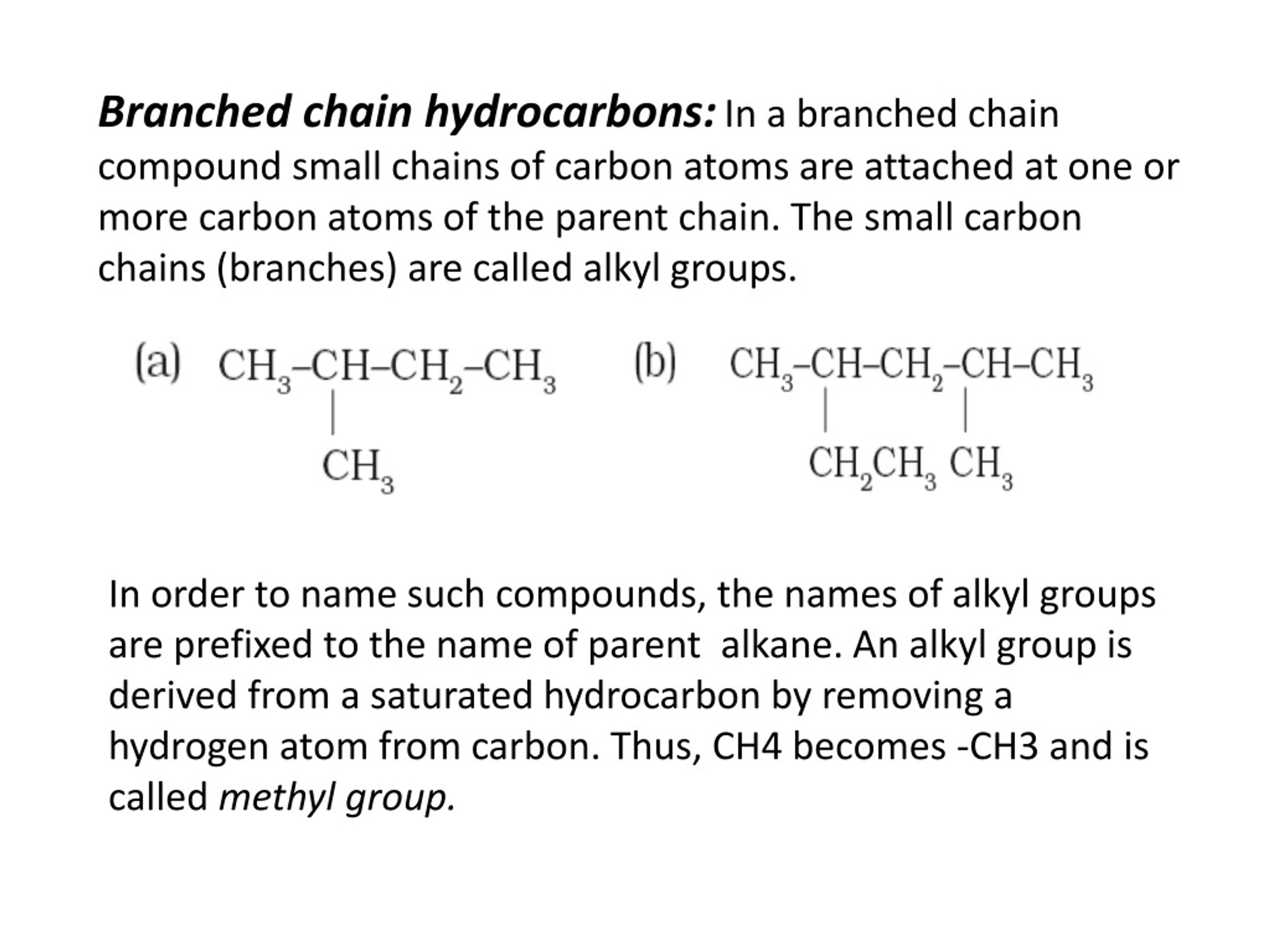
My Wishful Thinking
In my ideal world, a bent hydrocarbon chain would have a much more… spirited name.
Perhaps something like a “Curve-bon.” Or a “Loop-ane.” Maybe even a “Twisty-tine.”
Wouldn’t that just be more… entertaining? It paints a picture, doesn't it? You can almost see the little carbon atoms having a huddle and deciding to link up in a circle.
It sounds less like a chemical compound and more like a character from a whimsical children’s book. And frankly, I think chemistry could use a little more whimsy.
The Cycloalkane Reality Check
But alas, the universe of chemistry is not built on my whimsical naming conventions. The term is indeed cycloalkane.
It’s derived from “cyclo,” meaning ring or circle, and “alkane,” referring to the saturated hydrocarbon. So, it’s a ring of alkanes. Straightforward, I suppose.
The most famous example is cyclopropane. Imagine three carbon atoms linked together in a triangle. Pretty cute, if you ask me.

Then you have cyclobutane (four carbons in a square), cyclopentane (five in a pentagon), and cyclohexane (six in a hexagon, which is quite popular).
The Practicality of the Bend
Now, I’m not saying the bend is bad. Oh no. It’s actually quite important. These bent structures have different properties than their straight-chain cousins.
For instance, they can be more stable or less stable depending on the ring size. They react differently. They have different uses.
It’s like the difference between a straight road and a scenic route. The straight road gets you there faster. The scenic route offers more to see. Both have their purpose.
But still. The name. When a hydrocarbon chain is bent, it is called… cycloalkane.
My Lingering Thoughts
I try to embrace it. I really do. I tell myself, “Yes, it’s a cycloalkane. It’s formed a ring.” And in my head, I picture a tiny, energetic loop.
But sometimes, when I’m reading a textbook or a scientific paper, and I see that word pop up, a little part of me whispers, “Couldn’t we have come up with something a bit more… bouncy?”
It’s like when you see someone wearing a perfectly tailored suit, and you think, “Sharp! But imagine if they had a little sequin on their lapel. Just one. To add a touch of mischief.”
The Humor in the Nomenclature
Maybe it’s just the inherent humor in scientific naming. We take something that sounds so complex and try to assign it a label that’s supposed to be universally understood.
And sometimes, those labels are just a little bit… dry. Like a cracker with no cheese.
So, the next time you hear about a bent hydrocarbon chain, think of the cycloalkane. And maybe, just maybe, you’ll smile a little, picturing those carbon atoms deciding to hold hands and form a circle.
And if you want to call it a “Loop-ane” in your own private thoughts, who am I to judge? We all have our little quirks.
After all, when a hydrocarbon chain is bent, it’s not just a chemical structure; it’s a tiny, circular adventure. And that, in its own way, is rather delightful.
So, cycloalkane it is. But in my heart? It’s forever the “Twisty-tine.”

%2C+long+hydrocarbon+chain%2C+and+a+methyl+(CH3-).jpg)

