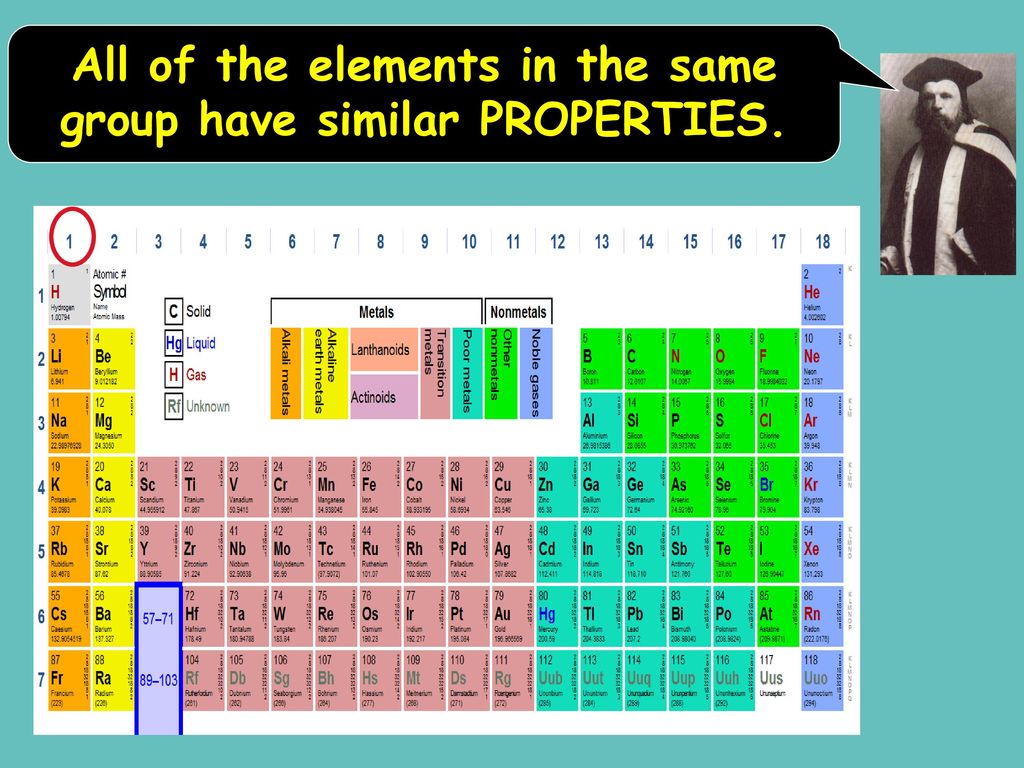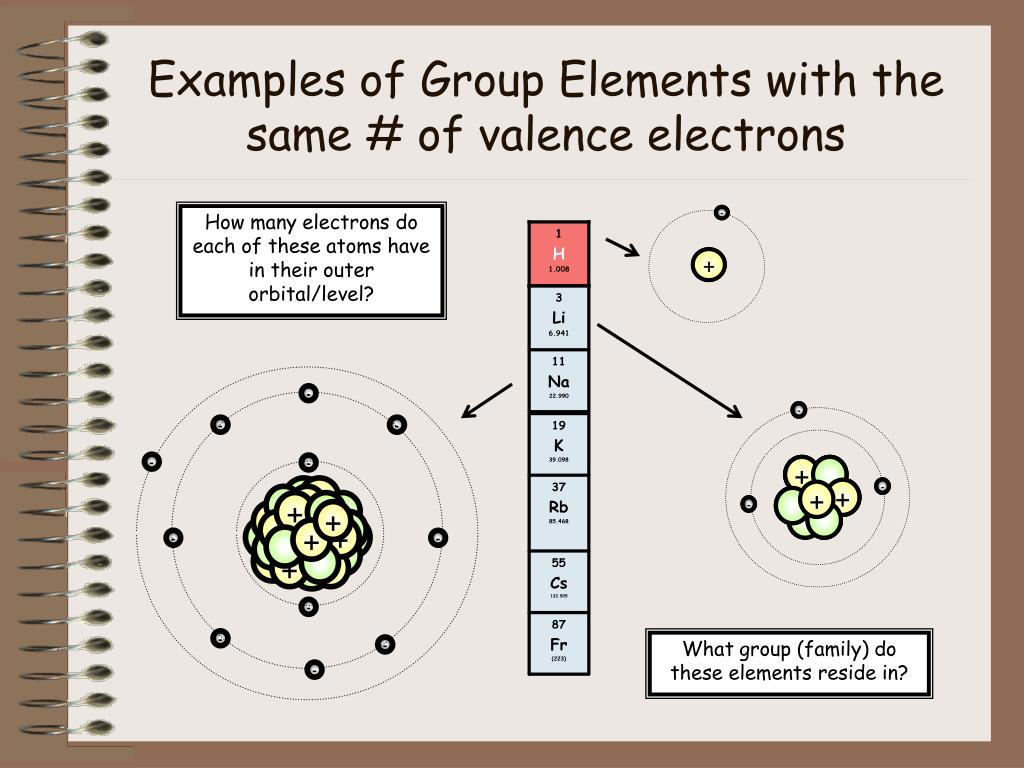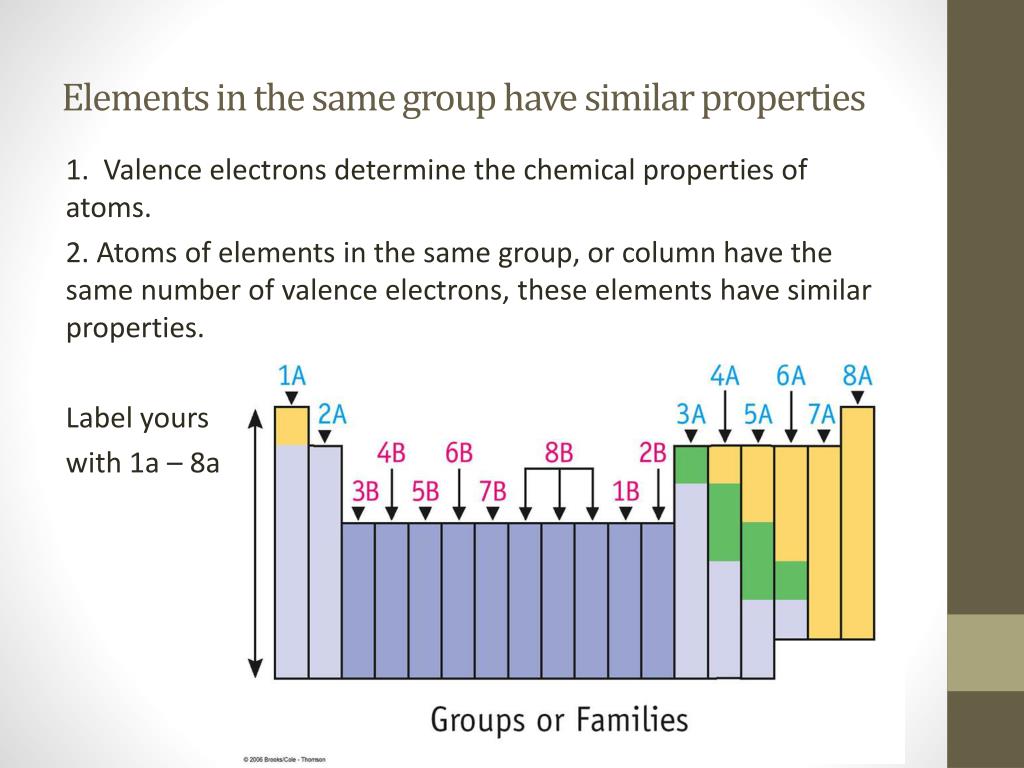
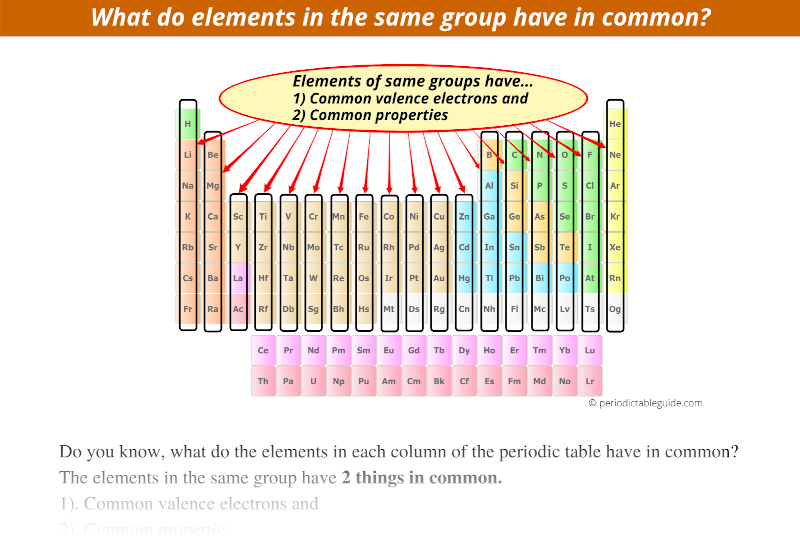
Hey there, curious minds! Ever looked at a science documentary or, dare I say it, a chemistry textbook (don't run away!) and felt a little... overwhelmed? All those symbols, numbers, and those weirdly organized charts? Well, guess what? There's a secret handshake in the world of elements, a little wink and a nod that makes them way less mysterious and, dare I say it again, a lot more fun. We're talking about the magical world of the periodic table, and specifically, about elements that hang out together in the same group. Think of it like a really cool family reunion, but with atoms!
So, what's the big deal about elements in the same group? It's all about their outer electrons. Now, before your eyes glaze over, stick with me! These outer electrons are like the element's personality traits. They're the ones that do all the interacting, all the bonding, all the dancing with other elements. And guess what? Elements in the same group have a very, very similar number of these important outer electrons. It's like they're all wearing the same uniform, or perhaps have been blessed with the same super-power!
Because they have this fundamental similarity in their electron "outfits," elements in the same group tend to behave in remarkably similar ways. It's like discovering that all your cousins, no matter how far apart you live, share your weird obsession with collecting novelty socks. They might have different hair colors or preferred pizza toppings, but that core similarity is there. And this, my friends, is where the real magic and fun begin!
A Family Affair: The Noble Gases!
Let's take a peek at one of the coolest families on the block: the Noble Gases. These guys are in Group 18, and they're famous for being, well, noble! They're incredibly unreactive. Think of them as the chill, laid-back members of the family who just don't get into any drama. Helium, neon, argon – these are the elements that make your party balloons float (helium, obviously!) and light up those flashy neon signs.
Why are they so chill? Because their outer electron shells are completely full! They've got all the electrons they need, and they're perfectly content. They don't need to borrow, steal, or share. They're the ultimate introverts of the elemental world, and that makes them uniquely useful. Imagine trying to build a stable structure if all your building blocks were constantly trying to hook up with each other! The noble gases provide that essential stability. Pretty neat, right?
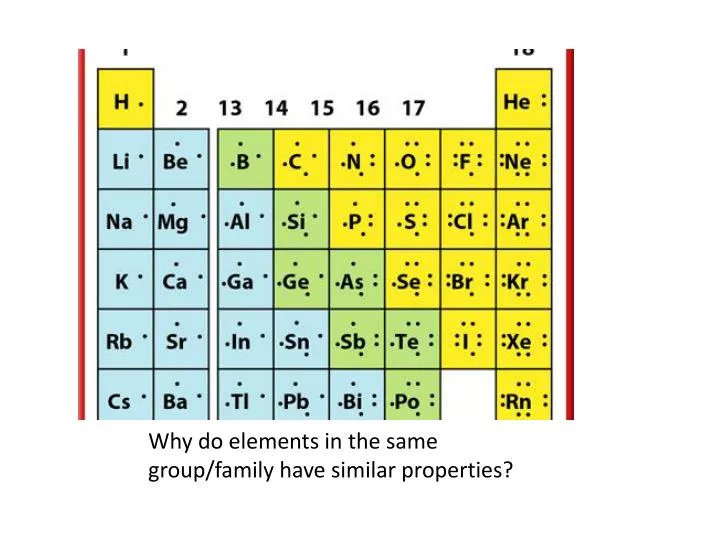
The Alkali Metals: Ready to Party!
Now, let's flip the script and meet the polar opposite: the Alkali Metals! These are in Group 1, and these guys are the life of the party! Lithium, sodium, potassium – you might know sodium as table salt. These elements are highly reactive. They have just one outer electron, and they're practically begging to get rid of it. They're like the generous friend who always offers you the last slice of pizza, even when they're hungry.
This eagerness to share their electron makes them incredibly useful in a whole bunch of ways. Sodium, for instance, is essential for nerve function in our bodies. Potassium is too! Lithium is the superstar behind your phone's battery. See? These reactive elements, with their single, eager outer electron, are powering our lives in so many ways, often without us even realizing it. It’s a testament to how powerful simple "giving" can be.

The Halogens: The Social Butterflies
Let's hop over to Group 17 and meet the Halogens. Fluorine, chlorine, bromine, iodine – these are the social butterflies of the elemental world. They have *seven outer electrons and are just one electron away from a full, happy shell. They're like that friend who's always looking for someone to complete their outfit.
Because they're so good at grabbing that extra electron, they readily react with the alkali metals (remember those guys?). This forms things like sodium chloride – your everyday table salt! Chlorine is also a vital disinfectant, keeping our water safe. And iodine? Crucial for your thyroid health! These elements, with their magnetic pull for an electron, are essential for life and well-being. It’s a beautiful demonstration of how opposites attract and create something wonderful.
Why This Makes Life More Fun
So, you might be thinking, "Okay, that's cool, but how does this make *my life more fun?" Well, think about it! The periodic table isn't just a bunch of abstract concepts. It's the blueprint for everything around you. The phone in your hand, the food you eat, the air you breathe – it's all made of elements!
Understanding that elements in the same group have similar properties allows you to start predicting things. If you learn about sodium's reactivity, you have a good idea that potassium will behave similarly, just perhaps a bit more intensely (it's the bigger sibling effect, maybe!). This makes learning about new elements less about memorizing a massive list and more about understanding families and their tendencies. It’s like learning the rules of a game, and suddenly, you can play!
.jpg)
It also adds a layer of appreciation for the world. The vibrant colors in fireworks? Often due to alkali metals or alkaline earth metals in different groups! The sparkling crystals in your jewelry? Many are compounds formed from elements that reacted because of their group tendencies. Suddenly, the mundane becomes magical, the ordinary becomes extraordinary. You start seeing the hidden chemistry in everything.
It’s a constant reminder that even though things appear very different on the surface, there can be profound underlying similarities that govern their behavior. It teaches us about connection, about shared characteristics, and about how these fundamental building blocks interact to create the universe we inhabit. It’s a beautiful dance of attraction and repulsion, stability and reactivity, all governed by the simple arrangement of electrons.
So, the next time you see the periodic table, don't just see a chart. See a vibrant community of elements, each with its own personality, all connected by the invisible threads of their outer electrons. Explore the groups, discover their quirks, and see how these simple similarities lead to the incredible complexity and beauty of our world. It’s a journey of discovery that’s waiting for you, and who knows what amazing things you’ll uncover!