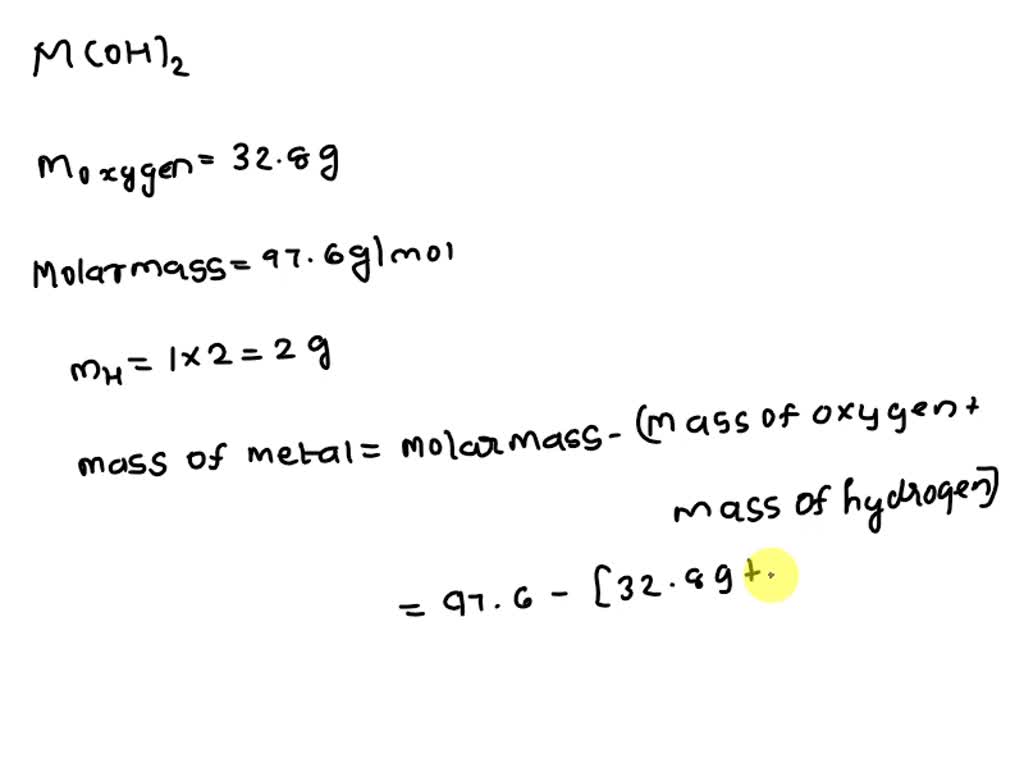
So, you wanna know about molar mass? Specifically, the molar mass of metal hydroxides? Sounds… science-y, right? But trust me, it’s actually pretty neat. Think of it as a super detailed shopping list for atoms. We're talking about how much a bunch of certain tiny things weighs. And metal hydroxides? They’re like the rock stars of this particular atomic party.
Let's break it down. What even is a metal hydroxide? Easy peasy. You take a metal – like iron, or copper, or even that stuff in your deodorant – and you stick it onto a hydroxide group. A hydroxide group is just one oxygen atom and one hydrogen atom chilling together. Like a tiny, inseparable duo.
So, we have our metal friend, and our OH buddy. Put them together, and BAM! You've got a metal hydroxide. Think of it like a partnership. The metal is the big shot, and the hydroxide is its trusty sidekick. It’s a pretty common combo in the world of chemistry. You find these guys everywhere, from your kitchen sink to the earth's crust.
Now, the molar mass. This is where things get a little more… precise. Imagine you're baking. You need exact amounts of flour, sugar, and eggs. Molar mass is chemistry's version of that. It tells you the mass of one mole of a substance. A mole? Don't get bogged down in that for now. Just know it's a ridiculously huge number of particles. Like, more zeros than you can count on a calculator.
So, when we talk about the molar mass of a metal hydroxide, we’re figuring out the weight of that specific combination of atoms. It’s like weighing a whole bunch of tiny Lego bricks, but for chemistry.
Why is this even fun? Because it’s like a puzzle! Each element has its own weight, its own atomic weight. We find those numbers, and then we just add them up. It's like solving a secret code. You look up the weight of the metal, you look up the weight of oxygen, you look up the weight of hydrogen, and then you do some math. Simple, right?

Let’s take a classic. Sodium hydroxide. You probably know it as lye. Yep, the stuff that makes soap! Super important, and a little bit… fiery. You don't want to mess with pure sodium hydroxide. But its molar mass? Totally chill to calculate.
Sodium (Na) has a molar mass of about 22.99 grams per mole. Oxygen (O) is around 16.00 grams per mole. And hydrogen (H) is about 1.01 grams per mole. So, sodium hydroxide (NaOH) is just 22.99 + 16.00 + 1.01. That gives you a molar mass of roughly 40.00 grams per mole. Easy!
See? No magic involved. Just a bit of looking things up and adding.
What about something a bit more… dramatic? How about iron(III) hydroxide? That's Fe(OH)₃. This stuff is the rusty red gunk you see on old bikes or neglected garden furniture. It’s like the metal's way of saying, "Oops, I got a little too friendly with the air and water!"
Iron (Fe) has a molar mass of about 55.85 grams per mole. Now, remember that little '3' next to the hydroxide? That means we have three hydroxide groups. So, we need to count the oxygen and hydrogen three times over. Three oxygens: 3 * 16.00 = 48.00 grams per mole. Three hydrogens: 3 * 1.01 = 3.03 grams per mole.
Add it all up: 55.85 (iron) + 48.00 (oxygens) + 3.03 (hydrogens) = a grand total of about 106.88 grams per mole for iron(III) hydroxide. Whoa. That’s a lot of atoms weighing in!

The cool thing is, this applies to any metal hydroxide. We’re talking calcium hydroxide (the stuff they use in cement!), magnesium hydroxide (that's in antacids – your tummy might thank you for this one!), aluminum hydroxide (found in antiperspirants, helping you stay dry and smell nice!). Each one has its own unique molar mass, a signature weight determined by its atomic makeup.
Why is this even useful, you ask? Well, scientists use molar masses for everything. It helps them figure out how much of a substance they need for a reaction. It's like measuring ingredients for a chemical recipe. They can predict how much product they’ll get, or how much of one thing they need to react with another. It’s fundamental to making new materials, understanding how things work, and even developing medicines.
Think of it this way: if you want to build a Lego castle, you need to know how many red bricks you have and how many blue bricks. Molar mass is the chemistry equivalent of counting your bricks. It's about having the right proportions to make something happen.

And here’s a little quirky fact for you: the molar mass is basically the average weight of all the different versions (isotopes) of an element. So, even though a single atom might be a tiny bit heavier or lighter, the molar mass is like the average joe weight of that element.
Another fun detail? Sometimes, the number you get for molar mass is surprisingly close to the atomic mass number you see on the periodic table. It’s not exactly the same, but it’s a good ballpark figure. It’s like the molar mass is the atomic mass's big, enthusiastic cousin who’s always bringing a crowd.
So, the next time you hear about "molar mass of a metal hydroxide," don't run away screaming. It’s just a fun little chemical calculation. It’s about understanding the building blocks of the world around us. It's about knowing that even the tiniest things have a weight, a presence. And when those tiny things team up into metal hydroxides, they create compounds that are essential to our lives, from the soap in your shower to the concrete holding up your house.
It’s a simple concept, but it unlocks a whole universe of chemical possibilities. It's the foundation for so much of what we understand about the physical world. So, go forth, ponder the molar masses of metal hydroxides, and impress your friends with your newfound scientific swagger. You’ve got this!


![Aluminum hydroxide [Al(OH)3] Molar Mass (With Calculations)](https://knordslearning.com/wp-content/uploads/2022/08/aloh3-molar-mass-aluminum-hydroxide-molar-mass.jpg)

