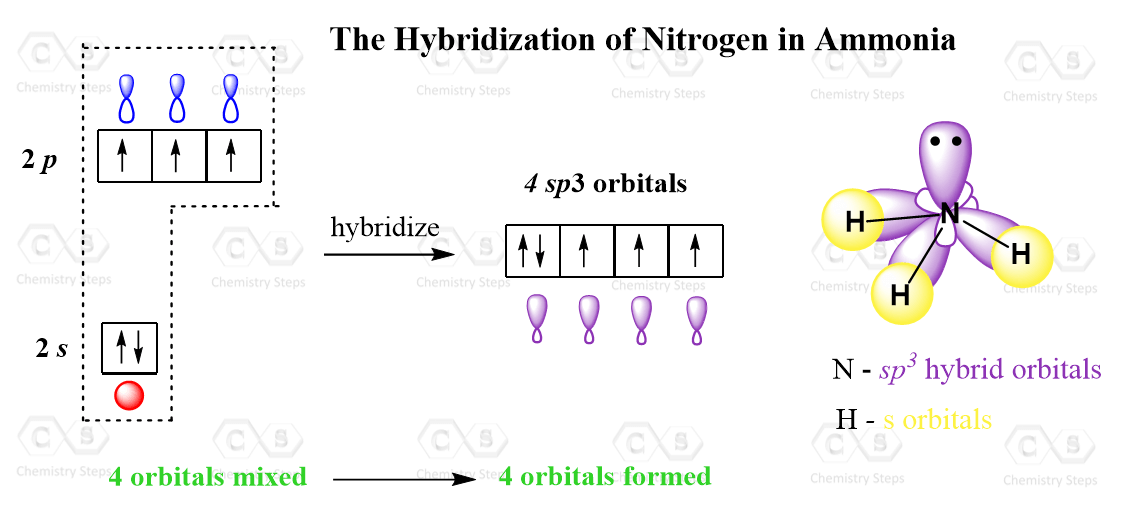
Hey there, fellow science enthusiasts and curious minds! Ever wondered about the secret lives of atoms, especially when they team up to form molecules? Today, we’re diving headfirst into the super cool world of Nitrogen and its electrifying persona in the NO₂⁺ ion. Get ready for a journey that’s as exciting as a roller coaster and as easy to understand as your favorite recipe!
So, what’s this fuss about hybridization? Think of it like atoms putting on a costume. Sometimes, to get the perfect molecular shape, atoms need to blend their “personality traits” – their electron orbitals – to form new, super-powered orbitals. It’s like a superhero team-up where individual powers are combined to create something even more awesome!
Now, let’s talk about our star, Nitrogen. In the wild, Nitrogen is usually a bit of a lone wolf, preferring to hang out with other Nitrogen atoms in its comfy N₂ form. But when Nitrogen gets a bit… well, charged up, it can become part of something really special. Enter the NO₂⁺ ion. This isn't just any old molecule; it's got a serious positive vibe, hence the “plus sign”!
Imagine Nitrogen as a talented chef. In its everyday life, it has a few different cooking styles (its electron orbitals: s and p). But when Nitrogen needs to bond with two Oxygens to create that super-stable NO₂⁺ ion, it’s got to get its act together and present a perfectly formed dish. It needs a plan, a strategy, and a way to connect with those Oxygens in the most efficient and pleasing way possible.
This is where the magic of hybridization comes in! For NO₂⁺, our Nitrogen chef decides to blend one of its s orbitals with one of its p orbitals. Poof! Abracadabra! It creates two brand new, equally awesome orbitals called sp orbitals. These aren’t just any old orbitals; they're like perfectly blended ingredients, ready to be used for maximum deliciousness – or in this case, maximum bonding!

It's like taking your basic flour and sugar and creaming them together with a splash of vanilla to make a cake batter that's ready for anything!
With these two shiny new sp orbitals, Nitrogen is now perfectly equipped to make its connections. It uses one sp orbital to form a strong bond with one Oxygen atom, and the other sp orbital for a strong bond with the second Oxygen atom. These bonds are like the sturdy pillars of a magnificent building, holding everything together with impressive strength and stability.
But wait, there’s more! Remember how Nitrogen started with more than just s and p orbitals? It had two leftover p orbitals that it didn’t use in the sp hybridization. These unused p orbitals are like secret weapons, ready to be deployed. In NO₂⁺, these two leftover p orbitals are perfectly positioned to form a double bond with each Oxygen atom. Think of a double bond as a super-strong handshake, twice as firm as a regular one!
So, what does all this clever orbital blending lead to? It leads to a molecule, or rather, an ion, with a fantastic shape! Because Nitrogen is using its sp hybridized orbitals, the bonds it forms are perfectly spread out. They’re not all squished together like a can of sardines. Instead, they’re arranged in a straight line, 180 degrees apart. Imagine holding two balloons and trying to tie them together with a string – they’ll naturally want to spread out as far as possible. That’s the vibe here!
This linear arrangement is super important for the NO₂⁺ ion. It makes the whole structure incredibly stable and efficient. It’s like the most aerodynamic design you can imagine, allowing the ion to zip through chemical reactions with grace and power. It’s the sports car of the molecular world!
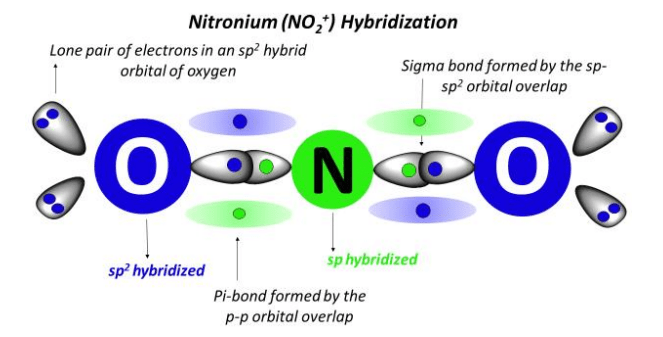
The hybridization of Nitrogen in NO₂⁺ is a testament to the incredible adaptability of atoms. They can literally reshuffle their internal structure to create new forms and new ways of interacting. It’s a beautiful dance of electrons, orchestrated by the fundamental laws of chemistry, all to achieve stability and create fascinating compounds like this positive, linear ion.
So, the next time you hear about sp hybridization, remember our Nitrogen chef, expertly blending its orbitals to create the perfectly shaped and stable NO₂⁺ ion. It's a little bit of molecular engineering, a dash of atomic creativity, and a whole lot of chemical coolness. Isn't science just the most entertaining thing ever?
And there you have it! The secret behind the neat and tidy structure of NO₂⁺ is its sp hybridized Nitrogen. It’s a fantastic example of how even the tiniest building blocks of the universe can perform amazing feats of coordination and bonding. So, let’s raise a metaphorical glass to Nitrogen and its brilliant sp hybridization! Cheers to chemistry!