Ever seen a chemistry set or a science documentary and noticed those little numbers with pluses or minuses next to chemical symbols? You know, like Na+ or Cl-? It can seem a bit intimidating at first, like a secret code only scientists understand. But guess what? That little number and sign are actually telling a really interesting story, and it's a story that impacts your everyday life in more ways than you might think. Think of it like a tiny tag that tells you what an atom is up to, and why it's behaving in a certain way. Let's dive in and decode this little mystery together!
So, what exactly is this number? It’s all about the tiny, buzzing particles that make up everything around us: atoms. Atoms are like the LEGO bricks of the universe. They're usually pretty balanced. Imagine a perfectly happy atom as a neutral person. They have an equal number of positively charged bits (called protons, found in the center) and negatively charged bits (called electrons, whizzing around the outside). For the most part, these positive and negative charges cancel each other out, and the atom is perfectly content, minding its own business.
But sometimes, atoms get a little… well, unbalanced. It's like when you're trying to share snacks with a friend. You have a certain number of cookies, and your friend has a certain number. If you have too many and your friend has too few, things can get a bit awkward, right? Atoms can experience a similar situation with their electrons. Electrons are the ones that tend to move around. They’re the most mobile part of the atom.
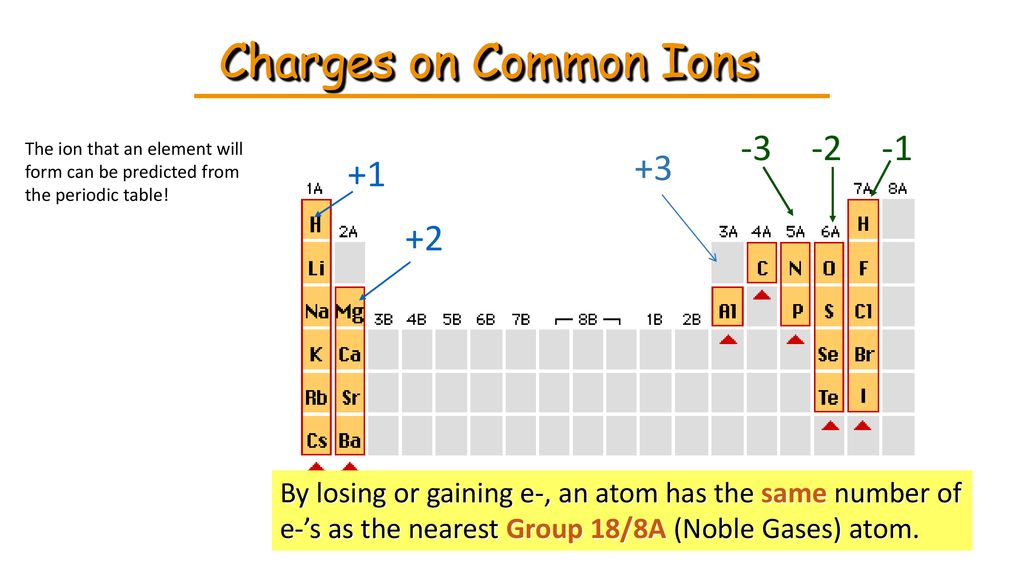
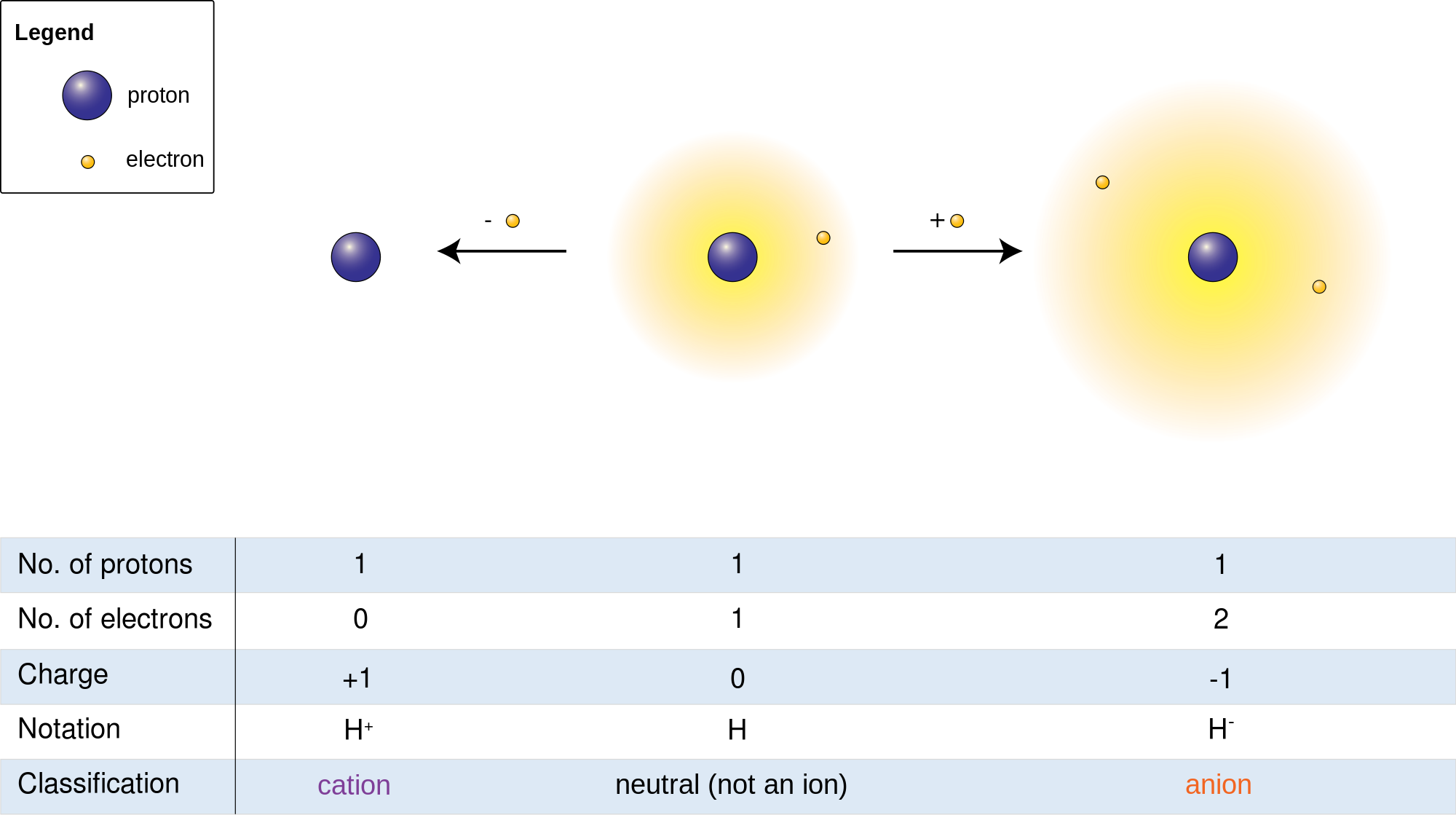
And that’s where those little numbers and signs come in! When an atom either gains or loses an electron, it’s no longer neutral. It’s become an ion. The sign tells us whether it’s lost or gained, and the number tells us how many electrons it’s played musical chairs with.
Let’s break down the signs. A plus sign (+) means the atom has lost electrons. Remember, electrons are negative. So, if you take away something negative, the atom becomes more positive overall. Think of it like this: you had five apples and two bananas. If you give away two bananas (which are like negative charges), you're left with a stronger sense of "appleness" (positivity). So, a Na+ ion, for example, is a sodium atom that has lost one electron. It’s now feeling a bit more positively charged.
On the other hand, a minus sign (-) means the atom has gained electrons. If you add more negative charges, the atom becomes more negative. Imagine you have three apples, and your friend gives you two more apples. Now you have a lot more "appleness" and might even feel a bit overwhelmed by all those apples! So, a Cl- ion, which is a chloride ion, is a chlorine atom that has gained one electron. It’s now carrying an extra negative charge.
So, Why Should We Care About These Wobbly Atoms?
This might sound like abstract science stuff, but it's actually super important for how the world works. These ions are the unsung heroes of so many everyday processes. They’re like the tiny workers that keep everything running smoothly.

Think about when you’re feeling a little dehydrated. You might reach for a sports drink, right? Those drinks often have electrolytes, which are basically ions! Sodium (Na+) and potassium (K+) ions are crucial for maintaining the right balance of fluids in your body. They help your nerves send signals and your muscles contract. Without them, you'd feel sluggish, and your body wouldn't function properly. It’s like your body’s electrical wiring needs these charged particles to send messages – no ions, no signal!
And what about something as simple as tasting your food? Those delicious flavors you experience? A lot of that comes down to ions interacting with your taste buds. The salty taste? That’s often due to sodium ions (Na+) and chloride ions (Cl-) working together. The sourness of a lemon? That’s often caused by hydrogen ions (H+). They’re literally tickling your tongue and telling your brain, "Hey, this is salty!" or "This is sour!"
Let's talk about something a bit more dramatic: electricity. How does that TV you’re watching get its power? In batteries, chemical reactions create ions that move around, generating an electrical current. It’s a whole dance of charged particles that eventually lights up your screen. So, those little numbers and signs are, in a way, the beginnings of electricity!

Even something as common as cleaning your house involves ions. Many soaps and detergents work by using ions to break down grease and dirt. They have molecules that can attract to both oily substances and water, and ions help facilitate this process, letting you wash away grime. It's like having tiny microscopic helpers that grab onto the dirt and escort it down the drain.
Consider the formation of rocks and minerals. Over millions of years, ions in water have dissolved and then recrystallized to form amazing structures like salt crystals or the shimmering veins in a gemstone. The beautiful patterns and colors are often a result of specific ions being incorporated into the crystal lattice. So, the next time you see a beautiful quartz crystal, you're looking at a history of ion interactions.

And what about the food you eat? The way your body processes nutrients, builds muscles, and even repairs itself involves a complex interplay of ions. Calcium ions (Ca2+) are essential for strong bones and teeth. Iron ions (Fe2+ or Fe3+) are vital for carrying oxygen in your blood. It’s a constant chemical ballet happening inside you, and ions are the lead dancers.
So, those little numbers and signs next to ion symbols aren't just random scientific jargon. They’re a snapshot of an atom’s charge, telling us if it’s feeling a bit positive after giving away some electrons or a bit negative after picking some up. This simple change in electrical charge is what allows atoms to interact with each other, form bonds, and create the incredible variety of substances and processes that make up our world.
Next time you see that Na+ or Cl-, don't just think of it as a complicated chemical formula. Think of it as a tiny, charged particle ready to do something important. It’s part of the reason you can taste your food, your muscles can move, and your lights can turn on. It’s a little bit of everyday magic, explained by the simple story of an atom losing or gaining an electron. Pretty cool, right?