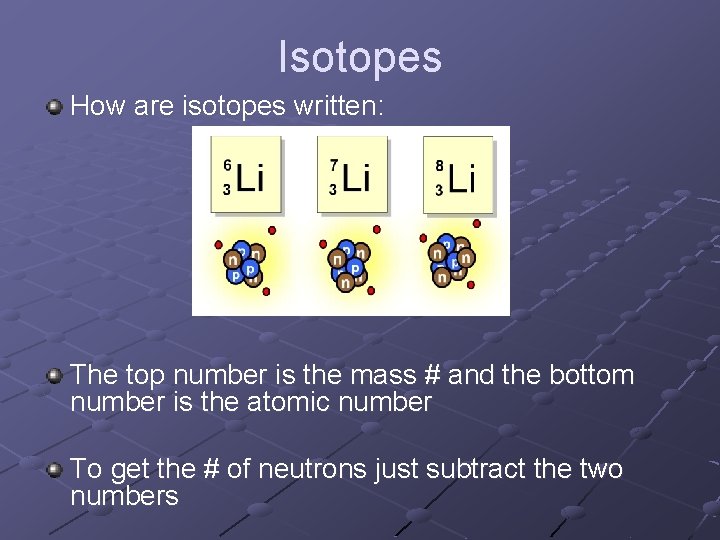
Okay, so you've probably seen these weird numbers next to element names, right? Like, Carbon-12 or Uranium-238. What's the deal with that number? It’s not just some random digit, I promise! It's actually a pretty big deal, and it tells us something super important about those atoms. Think of it like their own personal ID number, but way cooler.
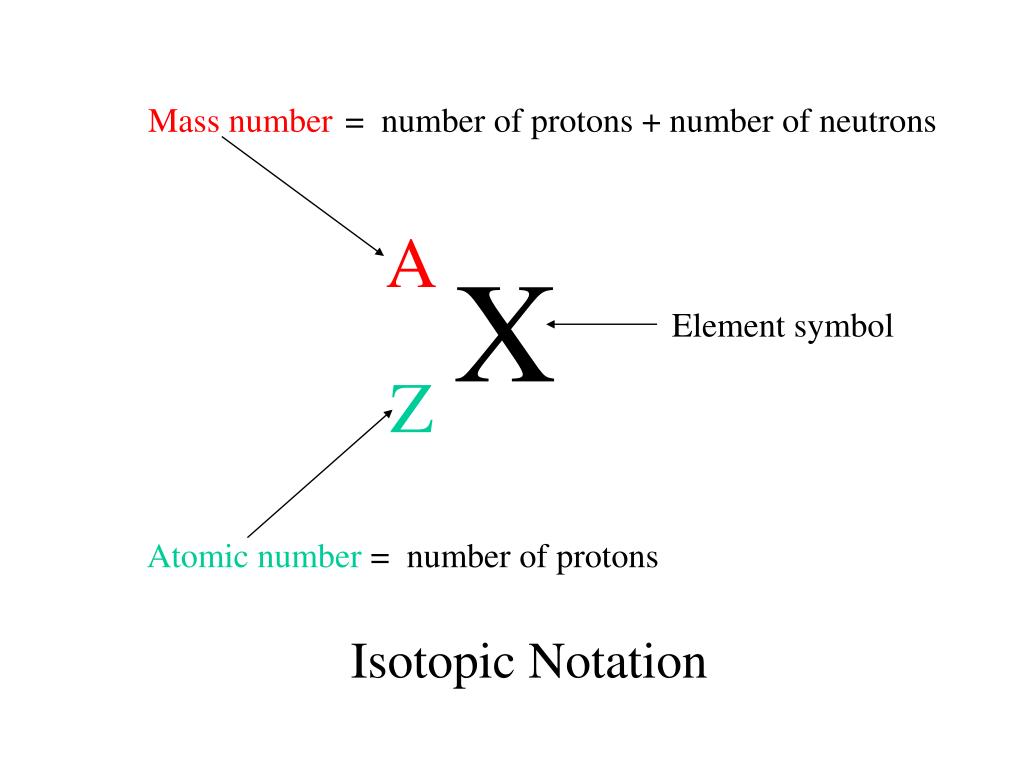
So, what's the scoop? That number, my friends, is the mass number. Pretty straightforward, I know. But what is the mass number, exactly? It’s not quite as simple as just counting all the bits inside an atom. Nope, it’s more specific than that.
Imagine an atom is like a tiny little LEGO set. It’s got protons, neutrons, and electrons. Now, the electrons? They’re super light, practically weightless in the grand scheme of things. So, when we’re talking about an atom’s mass, we’re mostly focusing on the other two guys: the protons and the neutrons. These guys hang out in the center of the atom, in the nucleus. They're the heavy hitters, literally!
So, the mass number is simply the total count of protons and neutrons in an atom's nucleus. That's it! Just add 'em up. Easy peasy, lemon squeezy. But here's where it gets really interesting.
Why do we even care about this mass number? Well, it’s what defines something called an isotope. Ever heard that word? Isotopes are like siblings from the same atom family, but with a slight difference. They're the same element, meaning they have the same number of protons (that’s what makes them that element, like Carbon always has 6 protons), but they can have a different number of neutrons.
And guess what? That difference in neutrons directly affects their mass number! So, Carbon-12 has 6 protons and 6 neutrons. Add 'em up, and voilà! 12. Makes sense, right? But then you might have Carbon-13. Still 6 protons (because it's Carbon, duh!), but now it’s got 7 neutrons. So, 6 + 7 = 13. See? The number changes!

And then there’s Carbon-14. Yep, you guessed it, 6 protons and 8 neutrons. 6 + 8 = 14. This little guy is actually pretty famous for its role in carbon dating, which is pretty wild when you think about it. We're using the "extra" neutrons to figure out how old ancient stuff is! Science is so cool, right?
So, when you see Carbon-12, Carbon-13, and Carbon-14, they're all versions of carbon. They all behave pretty much the same chemically because, again, same number of protons, same number of electrons (which are involved in chemical reactions). But their masses are different. That's the key takeaway!
Think about it like this: Imagine you have a bunch of identical twins. They look the same, they have the same parents, but one twin is, say, a little bit stronger because they went to the gym more often. They’re still the same person, but they have a different physical characteristic. Isotopes are kind of like that, but with neutrons.
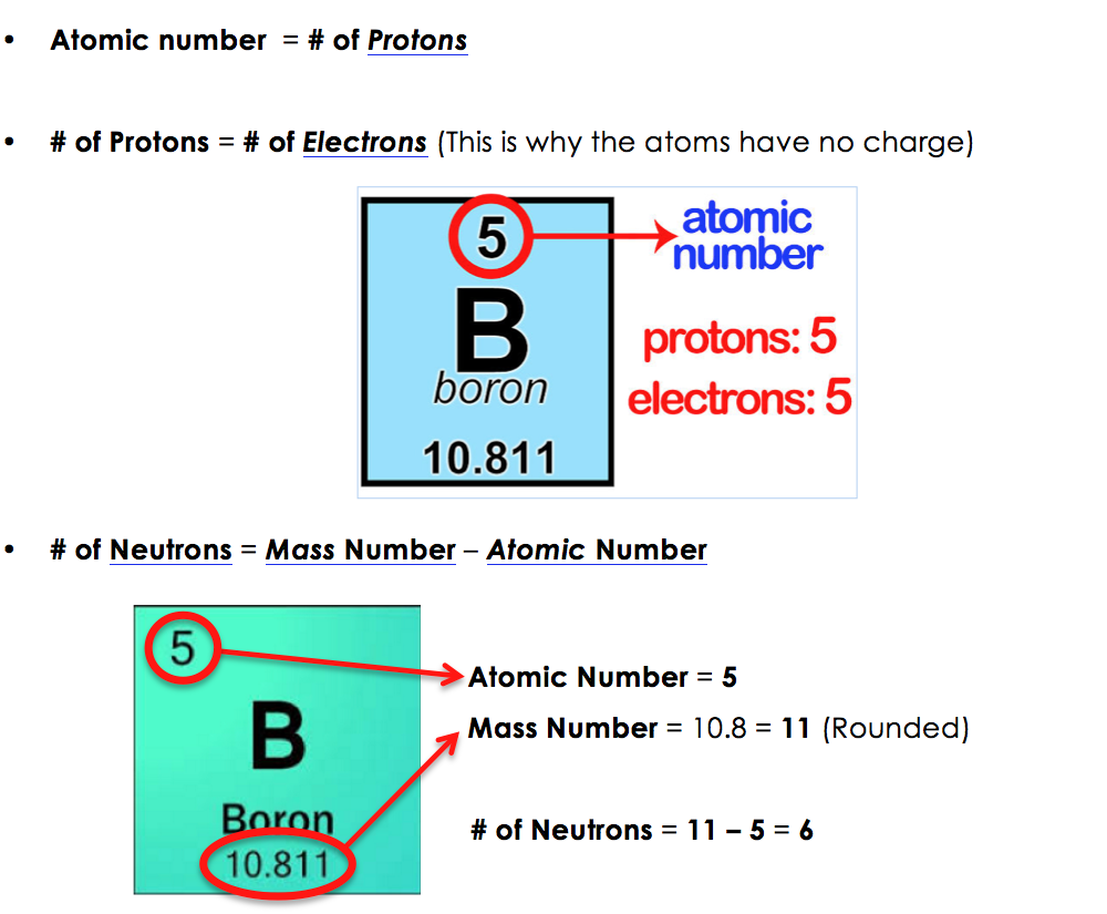
The number next to the element name, the mass number, is basically a way to tell you which specific version of that element you're dealing with. It’s like saying, "Hey, this isn't just any old oxygen, this is oxygen that weighs this much because it has this many neutrons."
Why is this important? Oh, you have no idea! Different isotopes of the same element can have vastly different properties, especially when it comes to their stability. Some isotopes are super stable, like a rock. They’ll just hang out forever. Others? Not so much. They're a bit… well, radioactive.
Radioactivity is a whole other can of worms, but in simple terms, it means an unstable nucleus is breaking down, releasing energy and particles. And guess which isotopes are usually the unstable ones? The ones with a different number of neutrons!
For instance, most Uranium we find is Uranium-238. That 238 is its mass number. It's got 92 protons (that's what makes it Uranium) and a whopping 146 neutrons. 92 + 146 = 238. Now, there’s also Uranium-235. Still 92 protons, but only 143 neutrons. 92 + 143 = 235. This Uranium-235 is the one that’s fissile, meaning it can be used in nuclear reactors and, well, other things you see in movies. The extra neutrons make all the difference!

So, that little number next to the element name? It’s not just decoration. It’s the key to understanding if you’re looking at a common, stable version of an element or a rarer, sometimes more exciting (and dangerous!), radioactive version. It’s the difference between a peaceful atom and one that’s a bit more… energetic.
When you’re looking at the periodic table, you’ll often see the average atomic mass listed. This is like an average of all the isotopes of an element, weighted by how common they are. It’s useful for calculations, but it doesn't tell you about the individual isotopes. That's where the mass number comes in. It's the specific identity of that particular atom or sample.
Think of it like this: If you’re talking about dogs, you might say the average dog weighs 50 pounds. But if you have a specific dog, you’d say, "Oh, that's a Golden Retriever, and he weighs 75 pounds." The 75 pounds is like the mass number for that specific dog (or isotope). It gives you more precise information.

So, the next time you’re staring at a chemistry textbook or a science documentary and you see that number, you’ll know what it means. It's not just a random digit. It's the total count of protons and neutrons, the very thing that defines an isotope and can tell us so much about an atom's properties, from its stability to its potential uses. Pretty neat, huh? It’s like having a secret code that unlocks a deeper understanding of the tiny building blocks of everything around us. Who knew something so small could hold so much significance?
It's funny how a simple addition problem can lead to such complex and fascinating behaviors. The number of neutrons is like the secret ingredient, the wildcard in the atomic recipe. And that mass number? It’s the direct consequence of that wildcard choice. So, next time you're at a science museum or just doodling in your notebook, remember that little number. It’s a whole story in itself!
And it’s not just about the big, dramatic stuff like nuclear reactions. Even in everyday chemistry, knowing about different isotopes can be important. For example, in medical imaging, doctors sometimes use radioactive isotopes of elements to track how things are working inside your body. Those specific isotopes have a mass number that, along with their other properties, makes them detectable and useful for diagnosis. So, that number is pretty vital for saving lives, too!
It really highlights how much variation can exist even within what we consider to be the same "thing." Like, all carbons are carbons, but they're not all exactly the same. They have these subtle, yet significant, differences that have massive implications. It’s a testament to the complexity and wonder of the universe at its most fundamental level. Just imagine all the possibilities that are packed into these tiny atomic nuclei, all represented by that single, important number!