Hey there, science curious! Ever heard of covalent bonds? Nope? Well, buckle up, buttercup, 'cause we're about to dive into the wild and wonderful world of atoms getting all buddy-buddy. Think of it like a dating app for the microscopic world. Super intense, right?
So, what's the big deal? Covalent bonds are basically how a ton of stuff around you sticks together. Your water bottle? Covalent bonds. The air you breathe? Covalent bonds. Even that slice of pizza you're craving? Yep, you guessed it. Pretty much the glue of the universe, but way cooler.
Sharing is Caring, Even for Atoms!
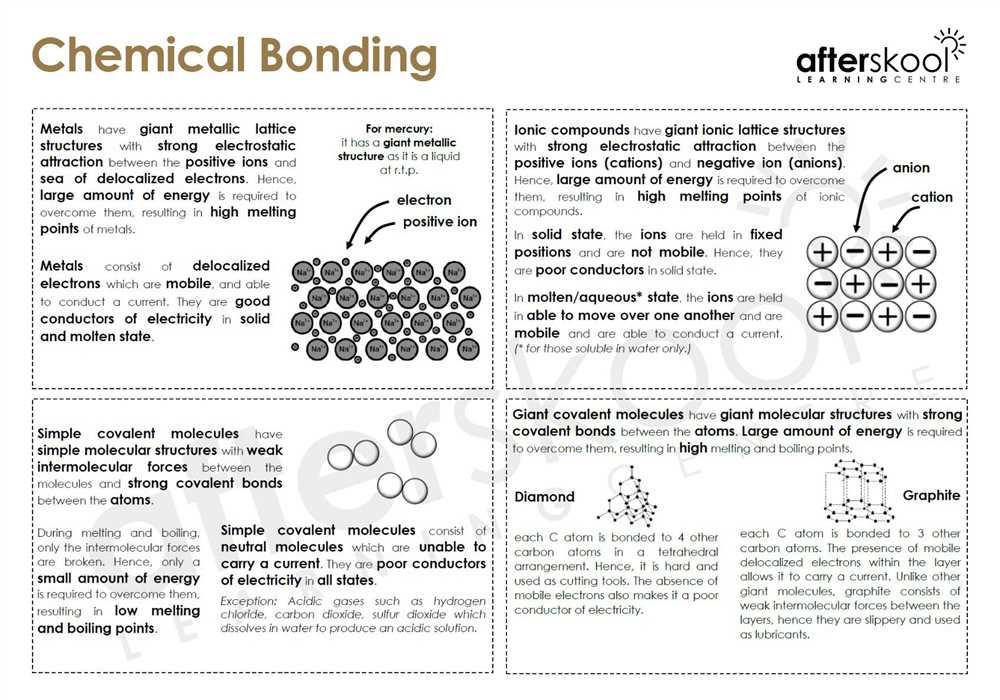
The real magic of covalent bonds is sharing. Unlike some other atomic relationships (we're looking at you, ionic bonds, you dramatic things!), these guys share their electrons. Imagine two friends at a playground, and one has a really cool toy. Instead of one friend snatching it, they decide to play with it together. That's a covalent bond in a nutshell.
These electrons are like the ultimate shared resource. Atoms, you see, are a little bit obsessive about having a full outer shell of electrons. It's their version of being "complete." So, when they can't quite get there on their own, they team up. They look at each other and go, "Hey, wanna share? We can totally achieve electron nirvana together!"
The Nitty-Gritty (But Still Fun!)
Let's get a tiny bit technical, but promise, it's not painful. We're talking about shared pairs of electrons. These shared electrons orbit both atoms. It's like a co-parenting situation for electrons, and it makes both atoms super happy and stable.

There are different flavors of covalent bonds too! You've got your single bonds, where they share one pair of electrons. Think of it as a casual handshake. Then there are double bonds, where they share two pairs. That's like a high-five, a lot more enthusiastic! And for the truly committed couples? You've got triple bonds, sharing three pairs. That's basically a full-on group hug, super tight and strong.
Why Is This So Rad?
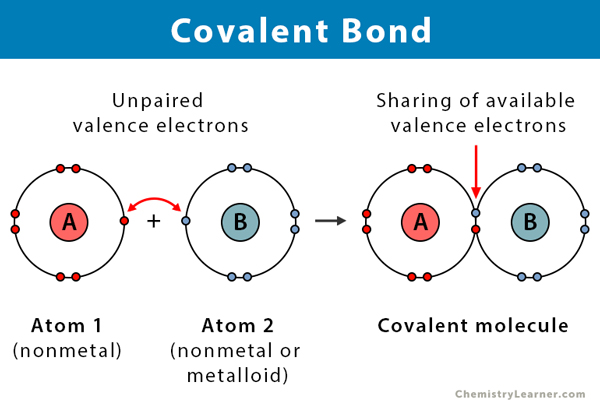
Okay, so why should you care about atoms holding hands? Because it explains everything! It explains why water is so chill and doesn't just fly apart. It explains why carbon can form these crazy, intricate structures that make up living things. It’s the foundation of life, people!
Think about it. Without covalent bonds, there'd be no molecules as we know them. No complex compounds. No us! It’s like the ultimate "how-to" guide for building reality. Kinda makes you feel like a cosmic architect, doesn't it?

Quirky Covalent Facts You Can Drop at Parties
Here's where things get really fun. Did you know that the strongest known chemical bond is a covalent bond? We're talking about the carbon-carbon triple bond in certain molecules. It’s like the Hulk of atomic bonds. Super strong, super tough.
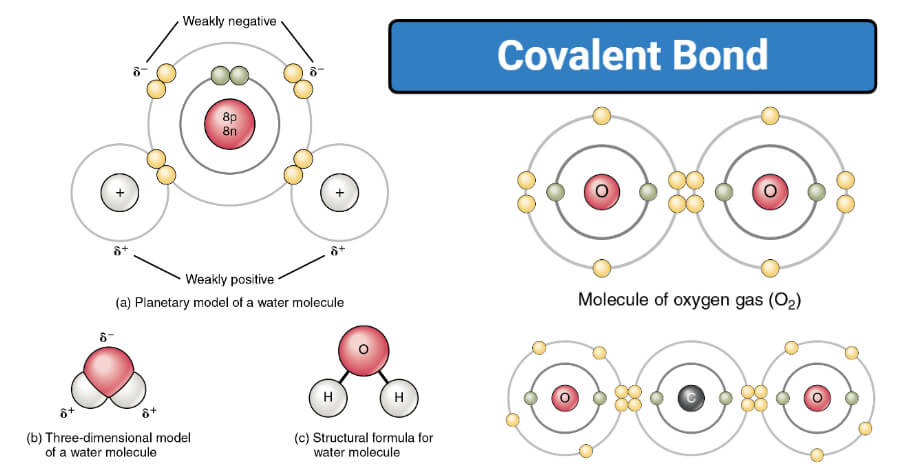
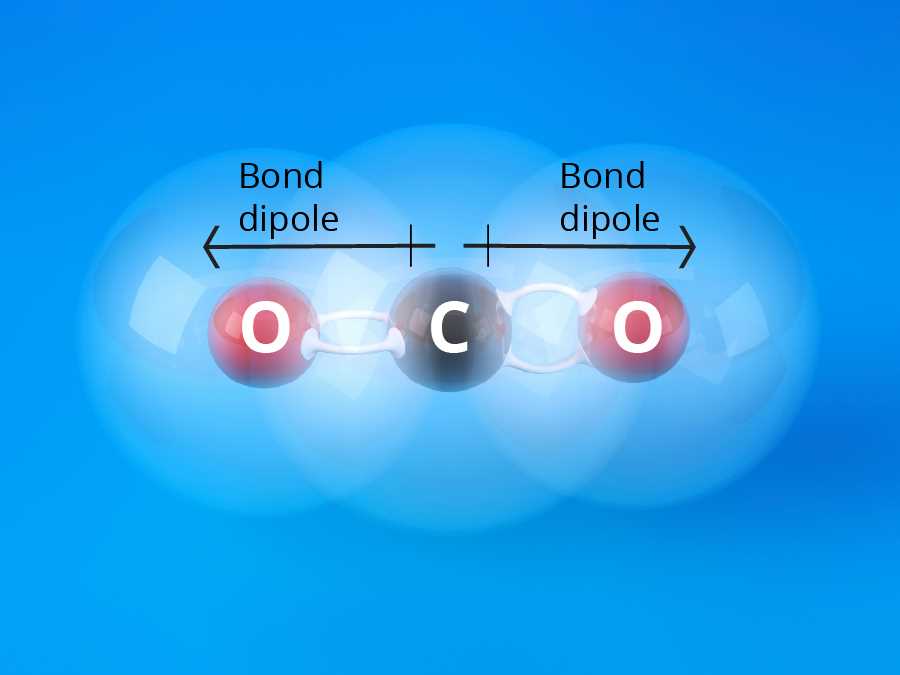
And get this: some molecules have what we call polar covalent bonds. This is like one atom being a little bit more "popular" with the electrons than the other. It's not a full transfer like in ionic bonds, but it's like one friend hogging the remote a tiny bit more often. This slight imbalance is super important for how water interacts with other things. It’s what makes it the universal solvent!

Ever heard of diamonds? Those sparkly beauties are held together by an epic network of covalent bonds. That’s why they're so hard! They're basically a giant, interconnected covalent hug. Talk about commitment!
The "Answer Key" Vibe
Now, you might be wondering about this "answer key" stuff. Think of it as the secret decoder ring for understanding how the world works at a fundamental level. When you’re looking at a molecule and trying to figure out how it’s put together, the principles of covalent bonding are your cheat sheet.
It's about recognizing those patterns. Seeing which atoms are likely to share, how they'll share, and what kind of structure that will create. It's not about memorizing a bunch of facts; it's about understanding the underlying logic. Once you get it, you start seeing these connections everywhere. It’s like unlocking a hidden level in a video game.
It's Not Rocket Science... Well, It Kinda Is!
Okay, okay, it is science. But it doesn't have to be scary! When you see a problem about covalent bonds, it's like a puzzle. You’ve got your pieces (atoms) and you know the rules of how they like to connect (sharing electrons). Your job is to figure out the best way they can fit together to become stable and happy.
And that's where the "answer key" comes in. It’s not about giving you the answers, but about giving you the tools to find the answers. It's about building your scientific intuition. So next time you're looking at a chemical formula, don't just see a jumble of letters. See the story of atoms sharing their lives, forming bonds, and creating the amazing world around us. Pretty cool, right?
So go forth and explore the covalent universe! You might be surprised at how much fun you have. And who knows, maybe you'll even start seeing your own relationships in terms of electron sharing. Just kidding... mostly!