Hey there, curious minds! Ever found yourself wondering about the tiny, invisible forces that keep our bodies humming along, or how plants manage to stand tall and proud? Well, today we're diving into something super cool, a concept that's surprisingly important and, dare I say, a little bit magical: osmolarity. Don't let the fancy word scare you; it's actually pretty straightforward once you break it down, and honestly, it’s a bit like a secret handshake for how liquids behave.
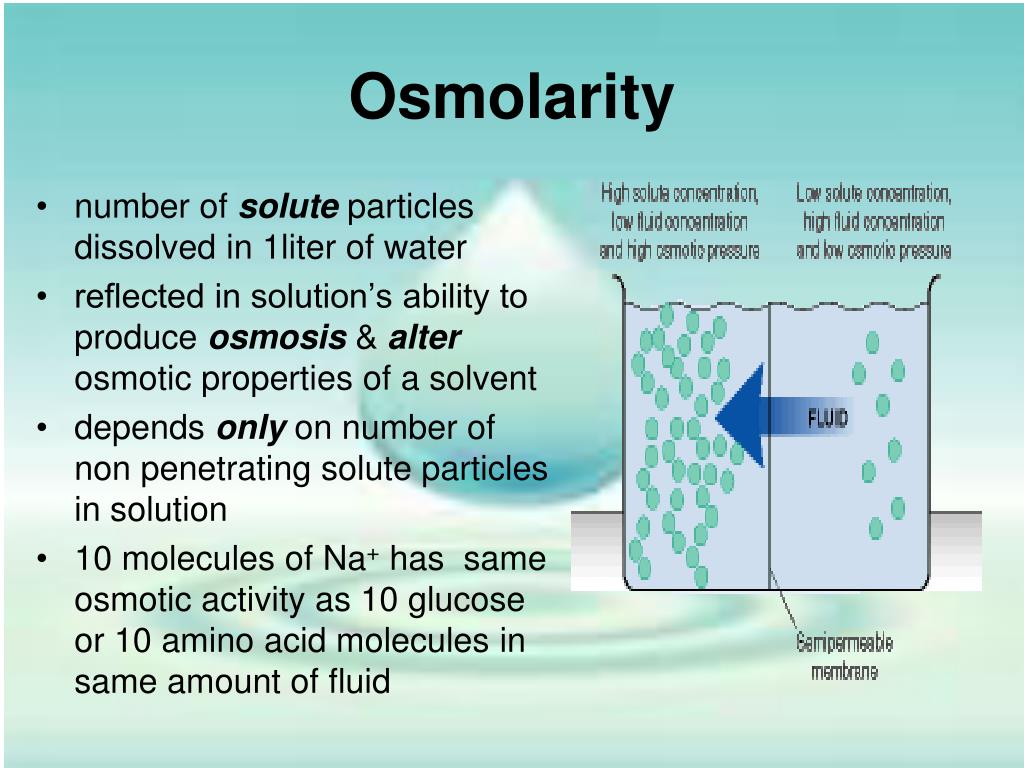
So, what exactly is this osmolarity thing? Think of it like this: imagine you have two different solutions separated by a special kind of barrier, like a super-fine sieve that only lets water molecules pass through, but not the stuff dissolved in it. This barrier is called a semipermeable membrane. Now, if these two solutions have different concentrations of dissolved stuff – let's call them "solutes" – something interesting happens. Osmolarity is basically a way to measure that concentration difference, and it tells us which way the water is going to move.
Think of it like a party. You’ve got a room with a handful of people (solutes) and a lot of empty space (water). Then, you have another room with way more people packed in, and less empty space. If you could magically open a door between these rooms that only let the "empty space" move, which way would it go? It would probably try to even things out, right? It would flow into the room where the "empty space" is more scarce, trying to dilute the crowd. Osmolarity is all about this tendency for water to move from an area of lower solute concentration (more water per solute) to an area of higher solute concentration (less water per solute).
Why is this water dance so important?
This might sound like something you'd only find in a chemistry lab, but it's happening inside you right now! Your cells are tiny little factories, and they're all surrounded by these semipermeable membranes. The fluid inside your cells, and the fluid outside your cells, have different concentrations of all sorts of dissolved things – salts, sugars, proteins, you name it.
If the osmolarity inside your cells is different from the osmolarity outside, water will start to move. If the outside solution has a higher osmolarity (more "stuff" dissolved in it), water will rush out of your cells to try and dilute that outside solution. This can cause your cells to shrink, which is definitely not ideal. On the flip side, if the outside solution has a lower osmolarity, water will rush into your cells, potentially making them swell and even burst!

Your body is a master at keeping these osmolarities balanced. It’s a constant, delicate juggling act. Your kidneys, for example, are like super-powered osmolarity regulators. They fine-tune the concentration of solutes in your urine, which in turn helps to maintain the right water balance throughout your entire body. Pretty neat, huh?
Osmolarity in Action: Beyond Your Body
But it's not just about our squishy insides. Osmolarity plays a crucial role in the plant world too! Ever wondered how a wilting plant perks up after you water it? That's osmolarity at work. Plant cells also have semipermeable membranes. When a plant is dry, the concentration of solutes inside its cells is higher than the surrounding soil. So, water moves from the soil into the plant cells, making them plump and firm – that’s called turgor pressure, and it’s what keeps plants from flopping over.
Think about a raisin. It’s all shriveled up because it's lost a lot of water. If you put that raisin in a bowl of pure water, the water has a much lower osmolarity than the inside of the raisin. So, water rushes into the raisin, and voilà! You get a plump grape (or at least, a much less wrinkly raisin!). It's like a mini-hydration spa for dried-up fruit.

Here's another fun comparison: imagine you’re trying to pack for a trip. If you have a huge suitcase (lots of "space" or water), and you're trying to pack a few small items (solutes), it’s pretty easy to fit them in. But if you have a tiny little bag (less "space" or water) and you're trying to cram in tons of stuff, it’s going to be a tight squeeze! Osmolarity is kind of like that, but with water trying to find its comfortable balance.
Let’s Get a Little More Specific (But Still Chill!)
Now, while we're talking about osmolarity, you might also hear terms like molarity or molality. These are related, but osmolarity specifically focuses on the number of osmoles. An osmole is a unit that measures the number of solute particles that contribute to osmotic pressure. Sometimes, when a solute dissolves, it breaks apart into multiple particles. For example, if you dissolve sodium chloride (table salt) in water, it breaks into sodium ions and chloride ions. So, one mole of NaCl actually creates two osmoles of dissolved particles. It's like one person walking through a door, but then they split into two distinct individuals on the other side – they both contribute to the "crowd" count!
Selecting The Correct Statement About Osmolarity
So, when you’re faced with statements about osmolarity, what should you be looking for? You want to find the one that accurately describes the movement of water based on concentration differences across a semipermeable membrane. Here are some things to keep in mind:
- Water moves from an area of low solute concentration to an area of high solute concentration. This is the golden rule. Think of it as water trying to dilute the more concentrated side to make things equal.
- Osmolarity is about the total number of solute particles. It doesn't matter what the solute is, as long as it contributes to the overall concentration.
- It involves a semipermeable membrane. Without this barrier, water would just mix freely, and you wouldn't have osmosis.
- It's about equilibrium. The ultimate goal is to reach a state where the concentration is the same on both sides, and water stops moving.
For instance, a correct statement might be: "When a cell is placed in a solution with a higher osmolarity than its cytoplasm, water will move out of the cell." Conversely, an incorrect statement might say: "Water moves from a high solute concentration to a low solute concentration." That's the opposite of what happens!
Understanding osmolarity helps us appreciate the incredible precision of biological systems and the fundamental physical principles that govern them. It's a reminder that even the smallest, most invisible processes are vital to life as we know it. So next time you sip a drink or see a plant reaching for the sun, give a little nod to osmolarity – the unsung hero of hydration and balance!