Hey there! So, you know how sometimes you’re just chilling, maybe contemplating the universe, and then BAM! A random fact pops into your head? Yeah, me too. And today’s random fact is about… wait for it… rubidium. I know, exciting stuff, right? But stick with me, because this little element is actually pretty cool. And guess what? It’s got a secret. Or, well, not really a secret, more like a… two-for-one deal.
See, most things in life aren't just one thing, are they? Think about it. You've got your chocolate chip cookies, but some have nuts, some don't. Or your favorite band, they have their classic hits, but then they also have those… let's call them 'experimental phases.' Rubidium is kinda like that. It's not just plain old rubidium. Nope. It comes in a couple of different flavors, or as the science folks like to say, isotopes.
Now, before you start picturing tiny little rubidium atoms wearing different hats, let’s break it down. What even IS an isotope? It’s like having two brothers who look almost exactly the same, right? Same basic structure, but maybe one’s a little taller, or has a slightly different hair color. Atoms are like that too. They have a core, called the nucleus, with protons and neutrons. The number of protons is what makes an atom that element, you know, like the DNA of its identity. But the number of neutrons? That can change. And when it changes, you get an isotope.
So, for rubidium, the number of protons is always the same. That’s non-negotiable. It’s what makes it rubidium. But the neutrons? They can play a little game of musical chairs. And in rubidium’s case, there are two main variations that nature just decided to hand out. Pretty neat, huh?
The Usual Suspects
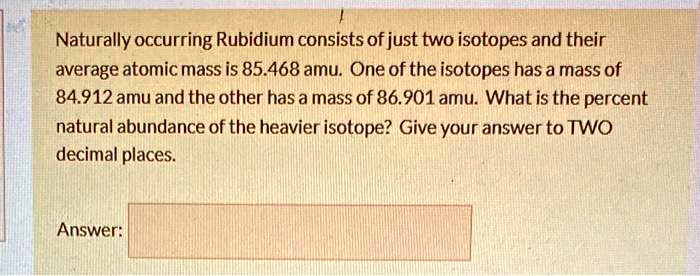
The two naturally occurring isotopes of rubidium are named after their total number of protons and neutrons. We've got rubidium-85 and rubidium-87. Think of them as Rubidium Lite and Rubidium Extra. You know, if atoms had marketing departments.

Rubidium-85 is the more common one. Like, way more common. If you were to grab a random handful of rubidium atoms (don't do that, by the way, it's not recommended), about 72% of them would be the -85 variety. It’s the laid-back, easygoing sibling. It just hangs out, does its thing, and doesn't cause too much fuss. It's stable, meaning it’s not going to spontaneously decide to fall apart. And that’s a good thing, stability. We all like stability, don’t we? Especially in our coffee orders.
Then there’s rubidium-87. This one's a bit more… interesting. It makes up the remaining chunk, about 28%. Now, 28% might sound small, but in the grand scheme of things, that’s a significant amount of ‘extra neutron’ action. And here’s where it gets really fun. Rubidium-87 isn't entirely stable. Gasp! I know, right? The very idea!
The Radioactive Rock Star (Kind Of)
Okay, when I say "not entirely stable," I don't mean it's going to explode like a rogue firework. Let's not get too dramatic. What it means is that rubidium-87 is radioactive. It undergoes a process called beta decay. Basically, it's a bit like a slow-motion transformation. Over a very, very long time, a neutron in its nucleus decides it's had enough and turns into a proton. This changes the rubidium-87 atom into a completely different element: strontium. Yep, it morphs!
And the timeframe for this transformation? Get this: the half-life of rubidium-87 is an absolutely mind-boggling 48.8 billion years. Billion! With a 'B'! That’s longer than the universe has even existed. So, while it's technically radioactive, you're not going to see your rubidium-87 sample suddenly glowing in the dark or anything. It’s more like a really, really, really patient form of decay.
Think of it like this: imagine you have a super, super, super old piece of chewing gum. And one day, after eons of waiting, it slowly turns into a tiny piece of rock. That’s kind of what rubidium-87 is doing, but on a cosmic scale. And that’s actually super useful, as it turns out. Who knew a slightly unstable atom could be so helpful?
Why Should We Care About Rubidium’s Dual Nature?
You might be thinking, "Okay, so it’s got two kinds, one’s a bit bouncy. So what?" Well, my friend, this little quirk has some seriously cool applications. It's not just some obscure fact for your next trivia night (though it would totally win you that round). It’s actually fundamental to how we understand the age of things.

Remember that rubidium-87 decays into strontium? This is the key! Scientists can use this. Imagine you’ve found a really old rock. How old is it? Like, really old? You can’t just ask it, can you? (Though that would be a fun conversation.) But what you can do is look at the ratio of rubidium-87 to strontium in that rock. Because when the rock formed, it had a certain amount of rubidium-87. As time went on, that rubidium-87 slowly turned into strontium. So, the more strontium you find, and the less rubidium-87 there is (relative to how much you’d expect), the older the rock is!
It's like finding a time capsule, but instead of letters, you're reading radioactive decay. This method is called rubidium-strontium dating, and it's a big deal for figuring out the ages of rocks, meteorites, and even the Earth itself. It’s like a geological clock. How awesome is that?
So, the next time you hear about rubidium, don’t just think of some random metal. Think of its two identities. Think of the stable, common rubidium-85, just chilling. And then think of the patient, slowly transforming rubidium-87, acting as a cosmic timer. It’s a perfect example of how even the smallest differences in atoms can lead to some pretty huge discoveries. It’s like finding out your quiet neighbor is secretly a master cryptographer. You just never know!

And it's not just about dating rocks. Rubidium itself, in various forms, has other uses. It’s used in atomic clocks, which are the most accurate timekeeping devices we have. Imagine something that’s so precise, it only gains or loses a second over millions of years. That’s some serious timekeeping power, and rubidium plays a part. It's like the superhero of punctuality.
It's also used in some niche scientific applications, like in vacuum pumps because it can absorb gases very effectively. And historically, it was even considered for use in fireworks due to its vibrant crimson flame, though that's not a common use today. Still, a metal that burns red? Pretty cool visually, wouldn't you say? Almost like a little bit of that rock-star radioactive energy showing itself in a less… geological way.
So there you have it. Rubidium. Not just a single entity, but a duo. A partnership of isotopes, each with its own role to play. One is the steadfast foundation, the other the subtle storyteller. They remind us that even within something as fundamental as an element, there’s complexity, variation, and ultimately, immense usefulness. It’s a little reminder that the universe, at its most basic building blocks, is anything but simple. And that, my friends, is why we keep looking, keep asking questions, and keep enjoying these little scientific wonders. Now, about that coffee… is it time for a refill?