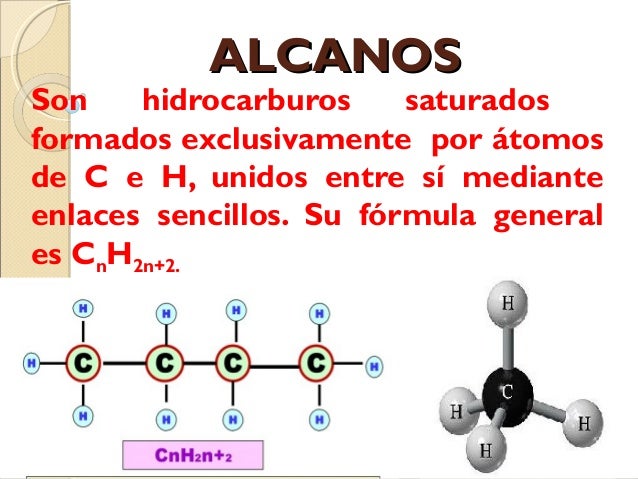
Hey there! So, you wanna chat about alkanes? Awesome! They’re, like, the simplest hydrocarbons out there, you know? Think of them as the vanilla ice cream of the organic chemistry world. No frills, just pure, unadulterated hydrocarbon goodness. We’re talking about molecules made only of carbon and hydrogen, and in alkanes, they’re connected in the most straightforward way possible. Like, super chill. No double bonds, no triple bonds, nothing fancy. Just single bonds holding hands, all day, every day. So, grab your mug, let’s dive in!
First things first, what makes them tick? We’re talking about their physical properties, right? These are the things you can observe or measure without actually changing what the alkane is. Like, is it a gas? Is it a liquid? Does it smell weird? All that jazz. And the coolest part? It all comes down to how these little molecules interact with each other. It’s like a big ol’ party, and the size and shape of your guests totally dictate how the party plays out. Makes sense, right?
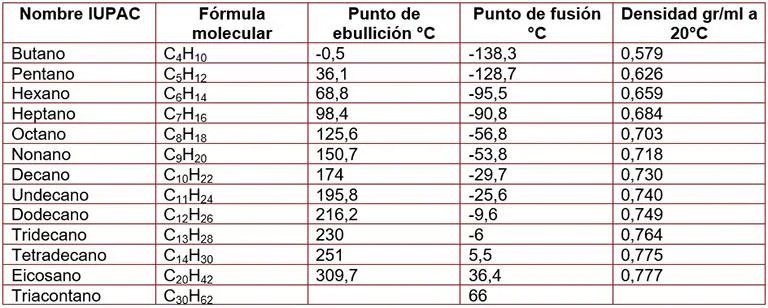
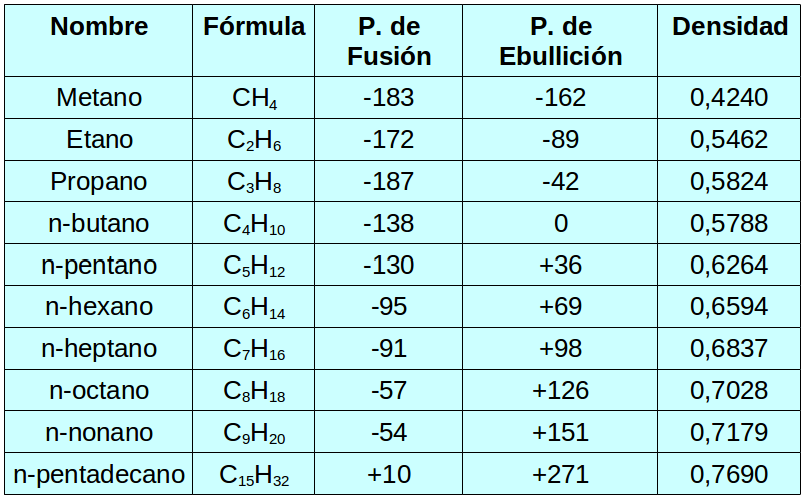
So, let’s talk about their boiling points. This is a biggie! Imagine trying to get these molecules to break free from each other and, you know, boil. The weaker their grip on each other, the less energy you need, and the lower the boiling point. Simple as that. Now, with alkanes, the main thing that messes with this grip is their size. Smaller alkanes, like methane (that’s just one carbon, super tiny!), they’re practically BFFs with each other and don't cling on too tight. So, boom, they’re gases at room temperature. Think about natural gas – that’s mostly methane! See? It all connects.
As you start adding more and more carbon atoms, these molecules get bigger and, dare I say, heavier. And what happens when things get bigger? They tend to stick together a little more. It’s like inviting a few more friends to your party – suddenly, there’s more chatter, more mingling, and it takes a bit more effort to get everyone to spread out. So, those bigger alkanes, like octane (think gasoline, eight carbons!), they’re liquids. You gotta heat ‘em up a bit more to get them to chill out and become a gas. It’s all about those intermolecular forces, my friends. These are like the invisible handshakes between molecules. For alkanes, they’re mostly van der Waals forces, and they get stronger as the molecules get bigger. Who knew a little bit of size could make such a difference, right?
Now, what about melting points? It’s kinda the same story. Getting those molecules to stop being solids and start wiggling around as liquids. Again, bigger molecules mean stronger attractions, so you’ll need more energy to melt them. Makes perfect sense, doesn’t it? But here’s where it gets a tiny bit sneaky. Branching! Oh, branching. Sometimes, instead of a long, straight chain, an alkane might have a little side branch. Think of it like a tangled spaghetti instead of a nice, neat pile. These branched molecules can’t pack together quite as snugly as their straight-chain cousins. So, even if they have the same number of carbons, a branched alkane might actually have a lower melting point because they’re not as good at that tight, cozy packing. Isn’t chemistry full of delightful little surprises?

Let’s talk about something you probably don’t want to do with your alkanes: dissolve them in water. Seriously, don’t. Alkanes are famously insoluble in water. Like, legendarily insoluble. You ever seen oil and water mix? Yeah, neither have I. And that’s because alkanes are nonpolar molecules. Water, on the other hand, is super polar. They’re like two people who just don’t click. One’s all structured and has these positive and negative ends (that’s water), and the other is just, like, evenly distributed charge, all chill and neutral (that’s our alkane friend). They just don’t have anything to grab onto each other with. So, they’ll just hang out separately, like at a party where no one knows anyone. It’s a classic case of “like dissolves like,” and alkanes and water are definitely not like.
But, guess what? If you try to dissolve them in other nonpolar solvents, like, say, gasoline or mineral oil, they’re super happy to mix! It’s like finding your tribe. All these nonpolar molecules can interact with each other just fine, and the alkane feels right at home. So, if you ever have a greasy stain (which is probably made of some sort of oily, nonpolar stuff), you’re gonna want to use a nonpolar cleaner to get it off. See? Practical applications everywhere!
Now, what about their density? Generally, alkanes are less dense than water. That’s why oil floats on water! They’re lighter, less packed together. As the alkane chain gets longer, the density increases a little bit, but they pretty much stay less dense than water. So, if you were ever in a situation where you needed to know if something was going to float or sink in water, and it was an alkane, you’d be pretty safe betting on it floating. Unless it’s, like, a really, really, really long alkane, but we’re talking about the common ones here.
Okay, so we’ve covered the pretty stuff, the physical stuff. Now let’s get into the nitty-gritty: their chemical properties. This is about how they react, what they can become. And here’s the kicker: alkanes are, for the most part, super unreactive. Like, incredibly boringly stable. They’re the introverts of the chemical world. Why? Remember those single bonds we talked about? They’re really strong and stable. It takes a lot of effort to break them. So, unless you’re really trying to push it, they’re just gonna sit there, being alkanes.
The main way alkanes do react is through combustion. And this is a HUGE deal, my friends. This is how we get energy from fuels like natural gas, propane, and gasoline. Basically, you take an alkane, you throw in some oxygen (O₂), and with a little spark of energy (like a match or a spark plug), WHOOSH! You get carbon dioxide (CO₂) and water (H₂O), and a whole bunch of energy released as heat and light. It’s the ultimate chemical party trick, and it powers our world!

Let’s take methane, CH₄, as an example. The balanced equation for its complete combustion is: CH₄ + 2O₂ → CO₂ + 2H₂O + Energy. See? Simple, clean (well, mostly clean, depending on how complete the combustion is), and generates tons of useful energy. If the combustion isn't complete, though, you can get other stuff, like carbon monoxide (CO), which is, you know, really not good for you. So, complete combustion is the goal!
Another way alkanes can react is through halogenation, specifically free radical halogenation. This is where a halogen atom, like chlorine (Cl) or bromine (Br), gets shoved into the alkane molecule. It’s a bit of a violent process, usually requiring UV light or heat to get started. Think of it like a karate chop to the alkane. The UV light or heat helps create these super reactive things called free radicals, which are atoms or molecules with an unpaired electron. They’re like little troublemakers, going around stealing electrons from other molecules, and in the process, they can break apart a C-H bond in the alkane, allowing the halogen to jump in. It’s not the most controlled reaction, and you can end up with a mix of products, but it’s a way to start modifying alkanes.
For example, if you react methane with chlorine gas (Cl₂) under UV light, you can get chloromethane (CH₃Cl) and hydrochloric acid (HCl). But it doesn’t necessarily stop there! You can get dichlorinated, trichlorinated, and even tetrachlorinated methane if you keep going. It’s like a game of chemical whack-a-mole. You’re trying to replace one hydrogen, but oops, another one gets replaced too! So, while alkanes are generally unreactive, they can be coaxed into doing things, especially under vigorous conditions.

What about oxidation? Well, as we saw with combustion, oxygen is their nemesis (or their best friend, depending on your perspective). Complete oxidation yields CO₂ and H₂O. Incomplete oxidation can lead to carbon monoxide or even just carbon (soot). So, their reaction with oxygen is pretty much their main claim to chemical fame, besides just existing.
One of the cool things about alkanes is their isomerism. Remember that branching we talked about? Well, different arrangements of the same atoms can lead to different molecules with different properties. Take butane. You can have n-butane, which is a straight chain (CH₃CH₂CH₂CH₃). Or you can have isobutane, which is branched (CH(CH₃)₃). They have the same chemical formula (C₄H₁₀), but they behave differently. Isobutane has a lower boiling point than n-butane because, as we discussed, those branches make it harder for them to pack together. It’s like having the same ingredients but making two different dishes. Mind-bending, right?
So, to sum it all up, alkanes are like the reliable, no-nonsense backbone of organic chemistry. They’re mostly found as gases, liquids, or low-melting solids depending on their size. They’re insoluble in water but love hanging out with other nonpolar substances. And chemically? They’re pretty chill, mostly reacting through combustion (which is super important for energy!) and a more complex process called halogenation. They might seem a bit boring at first glance, but honestly, without these simple molecules, a lot of what we rely on wouldn’t exist. Pretty neat, huh? So next time you see a flame or smell gasoline, give a little nod to the humble alkane!