Ever felt like you were playing detective, but instead of a smoking gun, you were looking for... well, a cloudy mess? Welcome to the wild and wonderful world of precipitates and solubility rules! Think of it as a cosmic tea party where some guests just can't stand each other and decide to form a perfectly formed, often colorful, solid little clump at the bottom of the mug. And the best part? You get to be the super sleuth figuring out who's doing what!
So, what's the big fuss about these "precipitates"? Imagine you're making a super-duper fancy potion. You mix two clear liquids together, maybe something that looks like sparkling lemonade and something that's totally see-through, like the purest mountain spring water. You swirl it all up with great anticipation, expecting a beautiful, shimmering elixir. But instead, BAM! The whole thing turns foggy, like someone just blew a giant bubble of cotton candy into your beaker. That's your precipitate! It's basically the unexpected solid party guest that shows up when you mix certain liquids (we call them solutions) together. It's like when you try to mix oil and water – they just refuse to mingle and create a distinct layer. Except with precipitates, it's like they decide to get married and settle down as a solid.
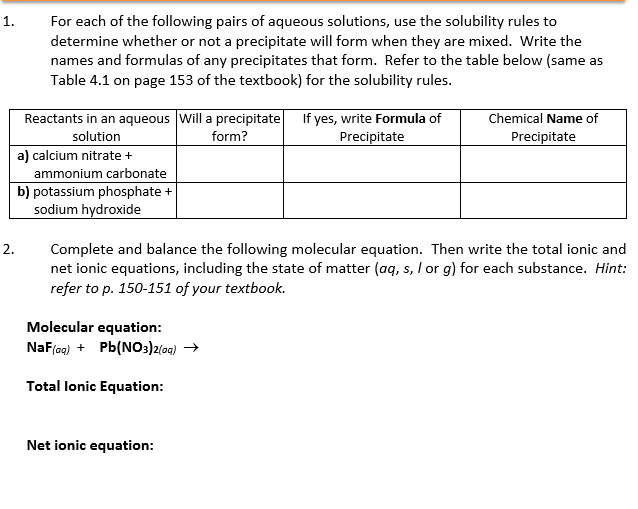
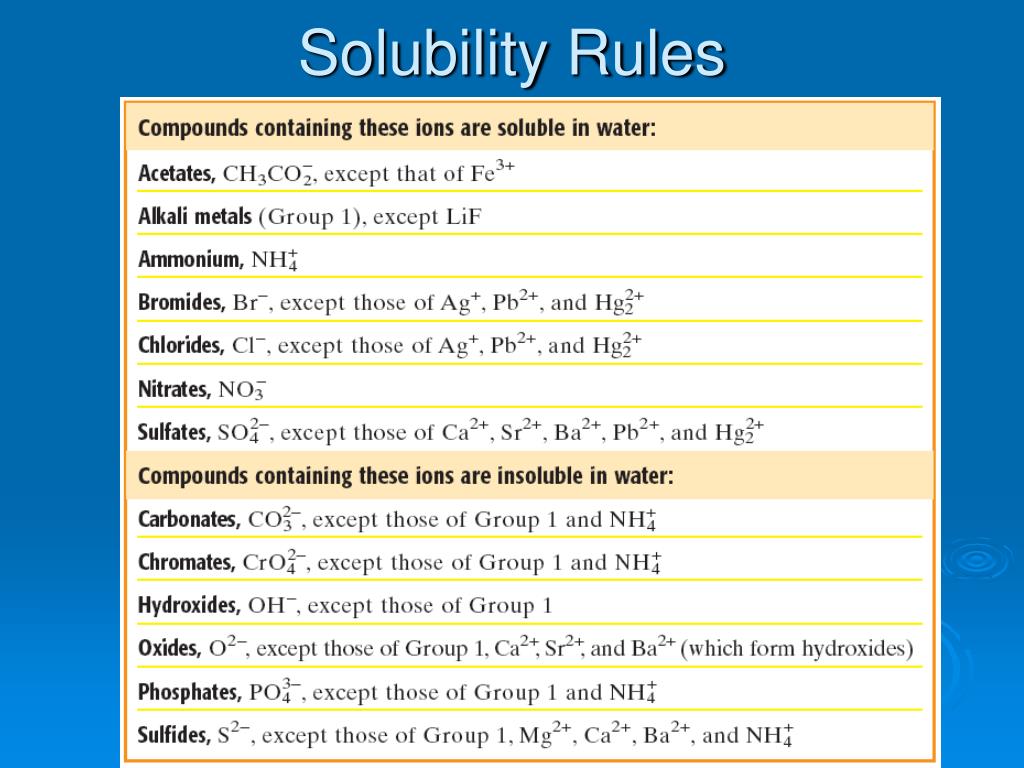
Now, how do we know if this solid party is going to happen? This is where our trusty solubility rules come in! These aren't your grandma's knitting patterns, oh no. These are the secret codes, the ancient scrolls that tell us which chemical compounds are the ultimate social butterflies and will happily dissolve in water, and which ones are the grumpy hermits, preferring to hang out as solids. Think of it like knowing which kids at school will always join the game of tag and which ones will hide behind the teacher. Solubility rules are the ultimate cheat sheet for predicting if a precipitate will make its grand entrance.
Our lab adventures were all about putting these rules to the test. We were like mad scientists in our own little laboratories, armed with beakers, test tubes, and a whole lot of exciting (and sometimes slightly alarming) chemicals. We'd take two perfectly clear solutions and, with a flourish and a dramatic whisper, mix them together. Then, the moment of truth! Would we see that glorious cloudiness, that magnificent precipitate? Or would our solutions just shrug and remain as clear as a freshly polished mirror? The suspense was palpable, folks! It was more thrilling than the season finale of your favorite reality show.
We were practically breathing the excitement, hoping for that satisfying 'plop' of a solid forming.
And when it happened, oh, when it happened! It was like watching a magic trick unfold right before your eyes. You'd mix two colorless liquids, and suddenly, a vibrant blue solid would appear, like a tiny sapphire jewel forming in the liquid. Or maybe a fluffy white cloud, as delicate as a snowflake, would drift down. It was pure, unadulterated chemical joy! And then, the even cooler part: using our solubility rules to figure out why that happened. It was like deciphering an ancient hieroglyph, but instead of pharaohs, we were talking about ions and charges.

For instance, let's say we mixed a solution of silver nitrate with a solution of sodium chloride (that's just fancy talk for table salt!). According to our magical solubility rules, most chlorides are super soluble, meaning they love to dissolve. But then there are a few grumpy exceptions, and silver chloride happens to be one of the grumpiest! So, when these two meet, they're like, "Nope, not playing nice!" and out pops a solid precipitate of silver chloride. We'd look at the white cloud and go, "Aha! You're the grumpy silver chloride, aren't you?!" It was incredibly satisfying.
Think about trying to dissolve sugar in water versus trying to dissolve sand. Sugar is a total social butterfly, it just melts into the water. Sand? Not so much. It just sits there, being sandy. Solubility rules are like the ultimate guide to which "sands" of the chemical world will dissolve and which will form their own little solid island. And in our lab, we were the captains of these chemical voyages, navigating the seas of solubility.

The "answers" we were looking for in our lab were basically confirmations. Did our observations match what the solubility rules predicted? If we mixed two things and expected a precipitate, did we see one? If we didn't expect one, did it stay crystal clear? It was all about seeing if our understanding of these chemical social dynamics was on point. And when it was, oh boy, did we feel like brilliant scientists! We were high-fiving, doing little victory dances (in our heads, of course, we're sophisticated scientists, after all).
Sometimes, things didn't go exactly as planned, and that's where the real fun began. Maybe a precipitate didn't form as strongly as expected, or perhaps a faint cloud appeared when we thought it should be crystal clear. These little anomalies were like intriguing puzzles. Were our solutions not quite pure enough? Did we accidentally grab the wrong bottle? It was a chance to become even better detectives, to really think about what was happening at the molecular level. It's like when your favorite recipe doesn't turn out perfectly, and you have to troubleshoot. You learn so much more from those little hiccups!
So, the next time you see something cloudy form in a liquid, don't just shrug. Think about the amazing world of precipitates and the clever solubility rules that help us understand this fascinating chemical dance. You're witnessing a tiny, solid party happening right before your eyes, and you, my friend, are the VIP guest who gets to figure out all the juicy details! It’s science, but it’s also like a really cool, visually interesting magic show. And who doesn't love a good show? We certainly did, and we’re pretty sure you would too! It’s a reminder that even in the seemingly invisible world of molecules, there’s plenty of action and plenty of fun to be had. Keep an eye out for those unexpected solids – they’re just waiting to tell you a story!