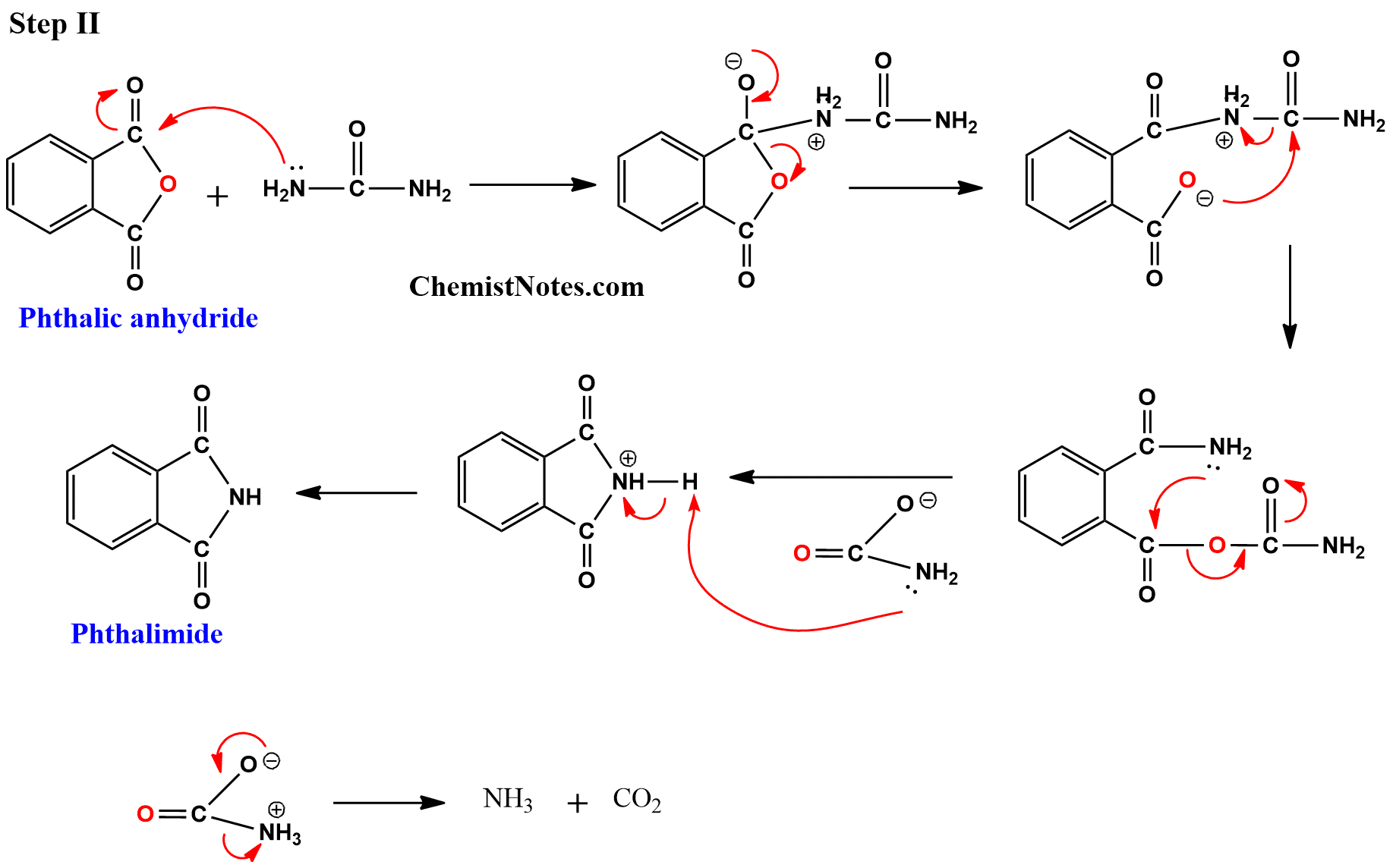
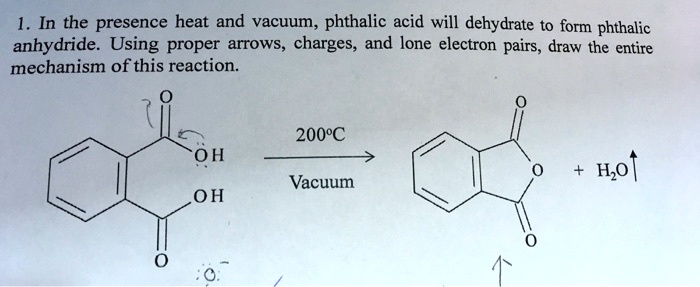
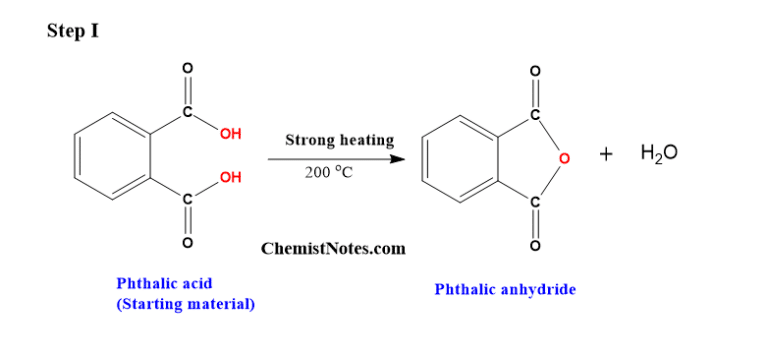
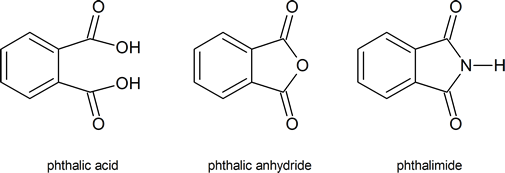Ever wonder what gives your favorite nail polish its glossy finish, or how those durable plastic pipes are made? Well, the answer might lie in a fascinating chemical transformation involving something called phthalic acid and its close cousin, phthalic anhydride. It might sound a bit technical, but understanding how these two are related is actually quite cool and has a surprisingly big impact on our everyday lives. Think of it as a little chemical secret that helps make a lot of the stuff we use possible!
So, what's the big deal with this conversion? Essentially, phthalic acid is a molecule that can be persuaded to lose a little bit of water, transforming into phthalic anhydride. This might seem like a small change, but it’s a really useful one. Phthalic anhydride is more reactive and stable in many industrial processes, making it a much more sought-after building block. The primary purpose of this transformation is to create a versatile intermediate for a wide range of materials. It’s a bit like taking a raw ingredient and refining it into something that’s easier and more efficient to work with, leading to a whole lot of benefits!
The benefits are pretty significant. For starters, phthalic anhydride is a key ingredient in the production of plasticizers. These are the clever compounds that make plastics, especially PVC, more flexible and less brittle. Without them, those garden hoses, electrical cable coatings, and even some types of flooring would be stiff and prone to cracking. Beyond plastics, phthalic anhydride is also crucial for making polyester resins, which are used in fiberglass for boats and car parts, as well as in coatings and paints for durability and shine. It's also a precursor to certain dyes, contributing to the vibrant colors we see in textiles and inks.
Where can you spot its influence? In education, this reaction is a classic example used to teach students about dehydration reactions and the interconversion of organic acids and anhydrides. It’s a tangible way to illustrate fundamental chemical principles. In daily life, you’re interacting with its products constantly. Think about the resilience of your shower curtain, the flexibility of some children's toys, or the protective coating on your car. These are all areas where phthalic anhydride plays a supporting role, ensuring performance and longevity.
Curious to explore this a little further? While you won't be performing chemical reactions in your kitchen, you can start by simply observing the world around you. Look for products that highlight terms like "PVC," "plasticized," or "polyester resin." You can also do a quick online search for "phthalate plasticizers" to see the vast array of everyday items that benefit from their inclusion. Sometimes, just understanding the 'why' behind the materials we use can be an incredibly rewarding journey, opening your eyes to the fascinating chemistry that shapes our modern world. It’s a reminder that even seemingly complex chemical processes have a story to tell and a real-world application.