Have you ever thought about the tiny building blocks of everything around us? We're talking about atoms, and they're pretty fascinating. Think of them like miniature solar systems, with a central core and little bits buzzing around. Now, within this atomic world, there are families of elements that are super interesting. Today, we're going to chat about one particular family called the Halogens. They're a bit like a quirky group of siblings, and we're going to uncover a secret about one of them.
The Halogens are elements 17 on the periodic table. You might have heard of some of them before, maybe in school or even in your own home! We've got Fluorine, Chlorine, Bromine, Iodine, and Astatine. They're known for being a bit reactive, which means they love to mix and mingle with other elements to form all sorts of cool compounds. Think about how salt is made – that's thanks to Chlorine and Sodium getting together! So, right off the bat, these guys are pretty important in our everyday lives.
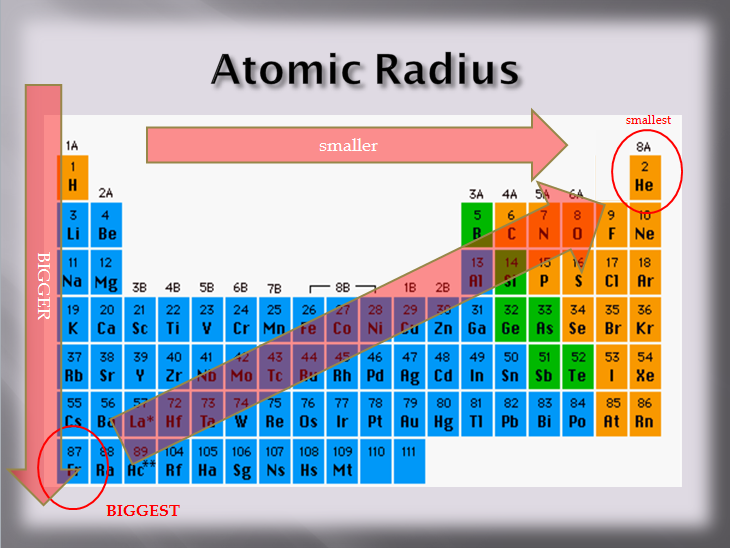
Now, let's zoom in on their size. Atoms, remember, are small. But even within this smallness, there are differences. Some atoms are a bit chunkier, and some are more delicate. The Halogens, despite being a family, also have variations in their size. It's like how in any family, some people are a little taller, and some are a little shorter. But with atoms, it's all about how many layers of electrons they have and how strongly the center pulls on them. It's a delicate balance!
So, the big question we're exploring today is: of all these Halogens, which one is the absolute teeniest, tiniest? Which one has the smallest radius? Imagine them all lined up, and you're trying to find the one that takes up the least space. It's a bit like a fun scavenger hunt in the microscopic world!
Let's meet our contenders. We have Fluorine, the very first member of the Halogen family. Then there's Chlorine, which we already mentioned with our salt example. Next up is Bromine, which is a liquid at room temperature and has a reddish-brown color. Then we have Iodine, a solid that gives us that nice purple vapor. And finally, Astatine, which is much rarer and more radioactive, making it a bit mysterious.

As you go down the Halogen family – from Fluorine to Chlorine, then to Bromine, and so on – the atoms tend to get a bit bigger. It’s like adding another floor to a building; it takes up more space. Each new element in the family has an extra layer of electrons, and that extra layer just adds to its overall size. So, if we're looking for the smallest, we should probably be looking at the top of the list, right?
And that brings us to our star of the show! Drumroll, please… the Halogen with the smallest radius is none other than Fluorine! Yes, that’s right. This feisty little element is the champion of tininess in the Halogen family. Isn't that neat? It’s the first one on the list, and it truly is the smallest.
Why is Fluorine so small, you ask? Well, it all comes down to its atomic structure. Fluorine has the fewest electron shells of all the Halogens. It’s like it only has one main "floor" for its electrons, while the others have more. Plus, the positive charge in its nucleus is strong enough to pull those electrons in nice and close. It’s a very efficient little atom!
Fluorine is the king (or queen!) of smallness when it comes to the Halogens.
Think about it – this tiny Fluorine atom plays a huge role in so many things. It's used in toothpaste to make our teeth strong and fight cavities. It’s also used in making Teflon, that super slippery stuff that coats our non-stick pans. So, even though it's the smallest, it's incredibly mighty and important!

It’s kind of inspiring, don’t you think? This little element, the smallest of its family, has such a big impact. It’s a testament to how even the tiniest things can be incredibly powerful and essential. It makes you wonder what other amazing secrets are hidden in the world of atoms, just waiting to be discovered.
So next time you brush your teeth or cook with a non-stick pan, you can give a little nod to Fluorine, the smallest Halogen, and appreciate its incredible contribution. It’s a fun little fact to know, and it reminds us that science is all around us, even in the most unexpected places. It’s definitely worth exploring more about these amazing building blocks of our universe!





