Ever wondered how scientists keep track of all those wacky-sounding chemical names? It’s not as complicated as it might seem, especially when we’re talking about a special family of molecules called alkenes. Think of them as the slightly more exciting cousins of the more common alkanes (which you might know as the building blocks of things like natural gas and paraffin wax). What makes alkenes so fascinating? It’s a tiny, yet mighty, difference: they have a double bond between two carbon atoms!
This double bond isn't just a quirky feature; it's the key to alkenes' unique properties and their incredible versatility. It’s like giving a molecule a superpower, allowing it to react in ways that its simpler alkane relatives can't. This makes alkenes incredibly important in the world around us, even if you don't realize it. They are the unsung heroes behind so many of the materials we use every single day, from the plastics in our water bottles and toys to the synthetic rubber in our tires.
So, why is it fun to learn how to name them systematically? For starters, it’s like cracking a code! Once you understand the rules, you can take any alkene molecule and give it a precise, unambiguous name. No more confusion, no more guesswork. This systematic naming system, called IUPAC nomenclature (which stands for the International Union of Pure and Applied Chemistry – don't worry, you don't need to memorize that part!), is the universal language of chemistry. It ensures that a chemist in Japan understands the exact same molecule when a chemist in Brazil says its name. That’s pretty cool, right?
The benefits of mastering this system are huge. It's not just for aspiring chemists in labs. Understanding chemical names helps us appreciate the composition of everyday products, from food additives to pharmaceuticals. It allows us to delve deeper into how these substances are made and how they function. Plus, there’s a real sense of accomplishment that comes with learning a new skill, especially one that unlocks a whole new way of understanding the molecular world.
Today, we're going to focus on the simplest members of this exciting family: the first three alkenes. They’re the foundation upon which all other, more complex alkenes are built. Think of them as the “alpha, beta, gamma” of the alkene world. Getting comfortable with these basic names will make it infinitely easier to tackle more challenging structures down the line. So, let's dive in and discover the systematic names of these fundamental molecules!
The First Three Alkenes: Unlocking Their Systematic Names
The world of alkenes is governed by a set of rules that, once learned, make naming them a breeze. The key is to identify the longest carbon chain that contains the double bond and then adjust the suffix of the parent alkane name. For alkenes, the suffix changes from "-ane" to "-ene". Simple enough, right? Let's meet our first three alkene stars!
The Littlest Alkene: Ethene
Our first alkene is the absolute smallest one. It's made up of just two carbon atoms, and, as you’ve probably guessed by now, they’re linked by a double bond. Following the IUPAC rules, we start with the corresponding alkane, which has two carbons and is called ethane. Then, we swap the "-ane" for "-ene". And voilà! The systematic name for this simplest alkene is ethene.
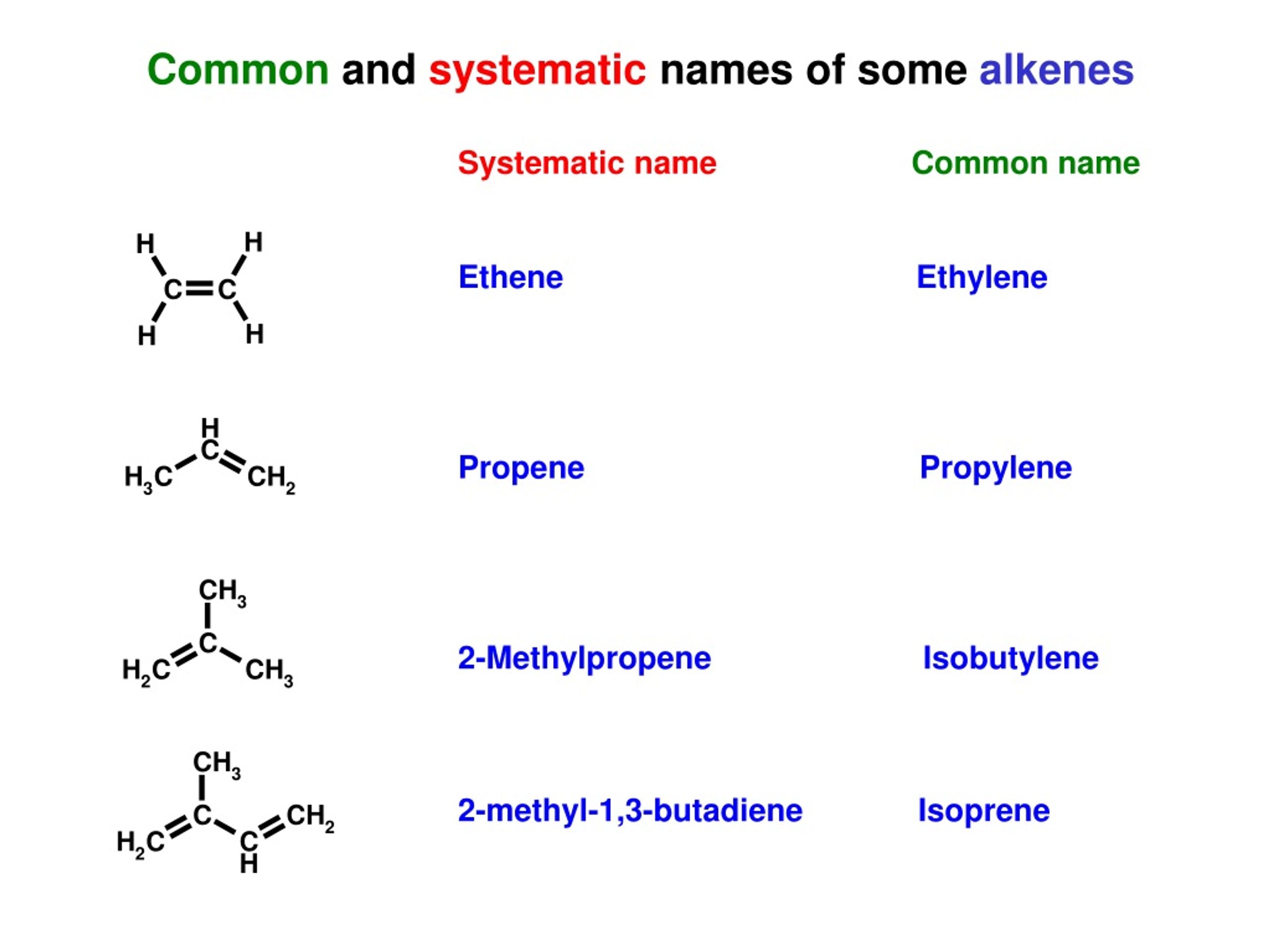
It’s also sometimes known by its common name, ethylene. While you might hear ethylene more frequently in certain industrial or biological contexts, ethene is the official IUPAC name. Ethene is a crucial molecule in industry; it’s a key starting material for making polyethylene, which is the most widely produced plastic in the world. Think of all the plastic bags, films, and containers – many of them start their life as ethene!
The structure of ethene is fascinating. It's a planar molecule, meaning all six atoms lie in the same plane. This specific geometry is a direct result of that double bond.
The Next Step Up: Propene
Moving on, we introduce a third carbon atom into our chain. This molecule now has a three-carbon backbone with a double bond somewhere within it. When there are only three carbons, the double bond can only be between the first and second carbon atom, or the second and third. However, in IUPAC nomenclature, we number the carbon chain so that the double bond gets the lowest possible number. With three carbons, there's really only one unique arrangement for the double bond that results in a distinct molecule. So, we start with the parent alkane name for three carbons, which is propane. Applying our "-ene" suffix, we get propene.
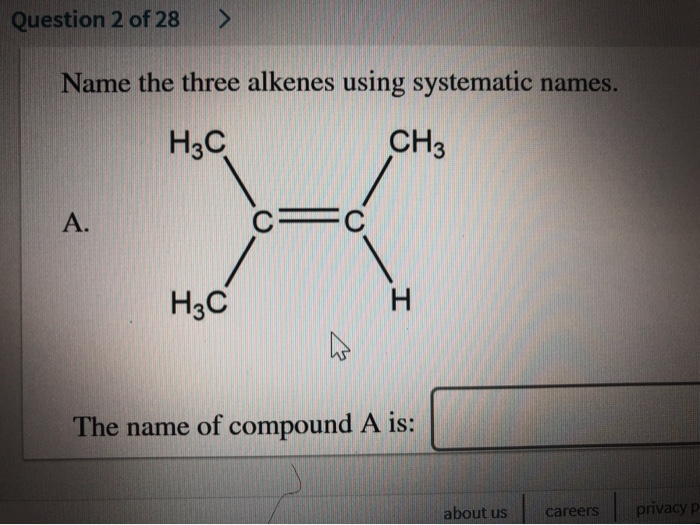
Just like ethene, propene also has a common name: propylene. Propene is another industrial powerhouse. It's used to create polypropylene, another extremely common plastic found in everything from carpets and clothing fibers to automotive parts and food packaging. It's also used in the production of other chemicals.
The presence of that double bond makes propene much more reactive than propane. This reactivity is what chemists exploit to build more complex molecules.
A New Possibility: Butene
Now, let’s add a fourth carbon atom. This is where things get a little more interesting and illustrate the importance of systematic naming even more clearly. With four carbons, we have the parent alkane name butane. Applying our "-ene" suffix, we might initially think of simply "butene." However, with four carbons, the double bond can actually be in two different positions within the main chain, leading to two different molecules that share the same molecular formula but have different structures and properties.
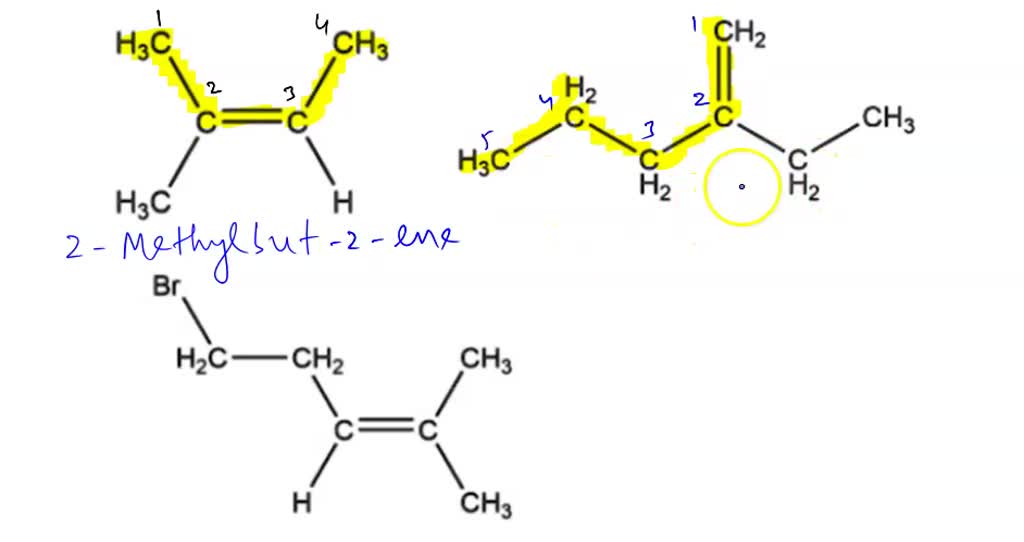
To distinguish between these, we need to use numbers. We number the carbon chain from the end that gives the double bond the lowest possible number. So, we can have the double bond between carbon 1 and carbon 2, or between carbon 2 and carbon 3. In the first case, the systematic name is but-1-ene. If the double bond is between carbon 2 and carbon 3, we would number it from the other end to get the lowest number, so it's still between carbon 1 and carbon 2 from that perspective. Therefore, the name is also but-1-ene. If the double bond is in the middle, between carbon 2 and carbon 3, numbering from either end will give the double bond a position of 2. Thus, the name becomes but-2-ene.
Both but-1-ene and but-2-ene are important industrial chemicals. But-1-ene, for instance, is often used as a comonomer in the production of polyethylene, helping to tailor its properties. But-2-ene is used in the synthesis of various other organic compounds.
The existence of but-1-ene and but-2-ene is a perfect example of isomers – molecules with the same chemical formula but different structural arrangements. This is why precise, systematic names are so vital!
Mastering these three – ethene, propene, and the various forms of butene – is your gateway to understanding the vast and dynamic world of alkenes. Each name tells a story about the molecule’s structure, hinting at its potential reactions and applications. So, next time you encounter a plastic product or a new material, you’ll have a better appreciation for the fundamental building blocks like these three alkenes!