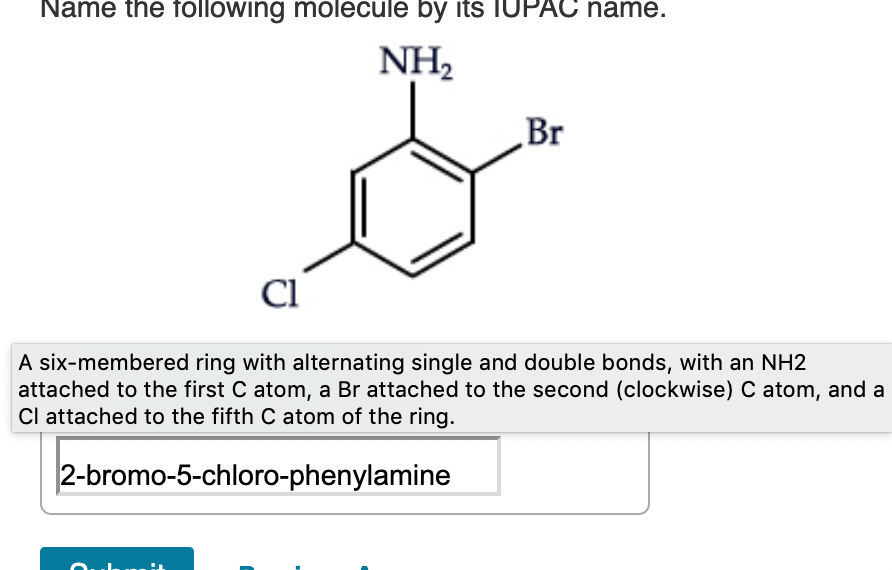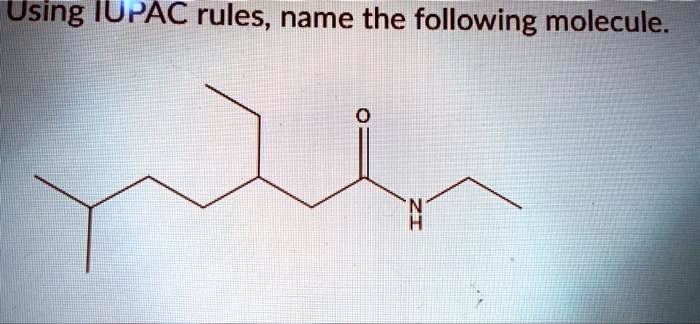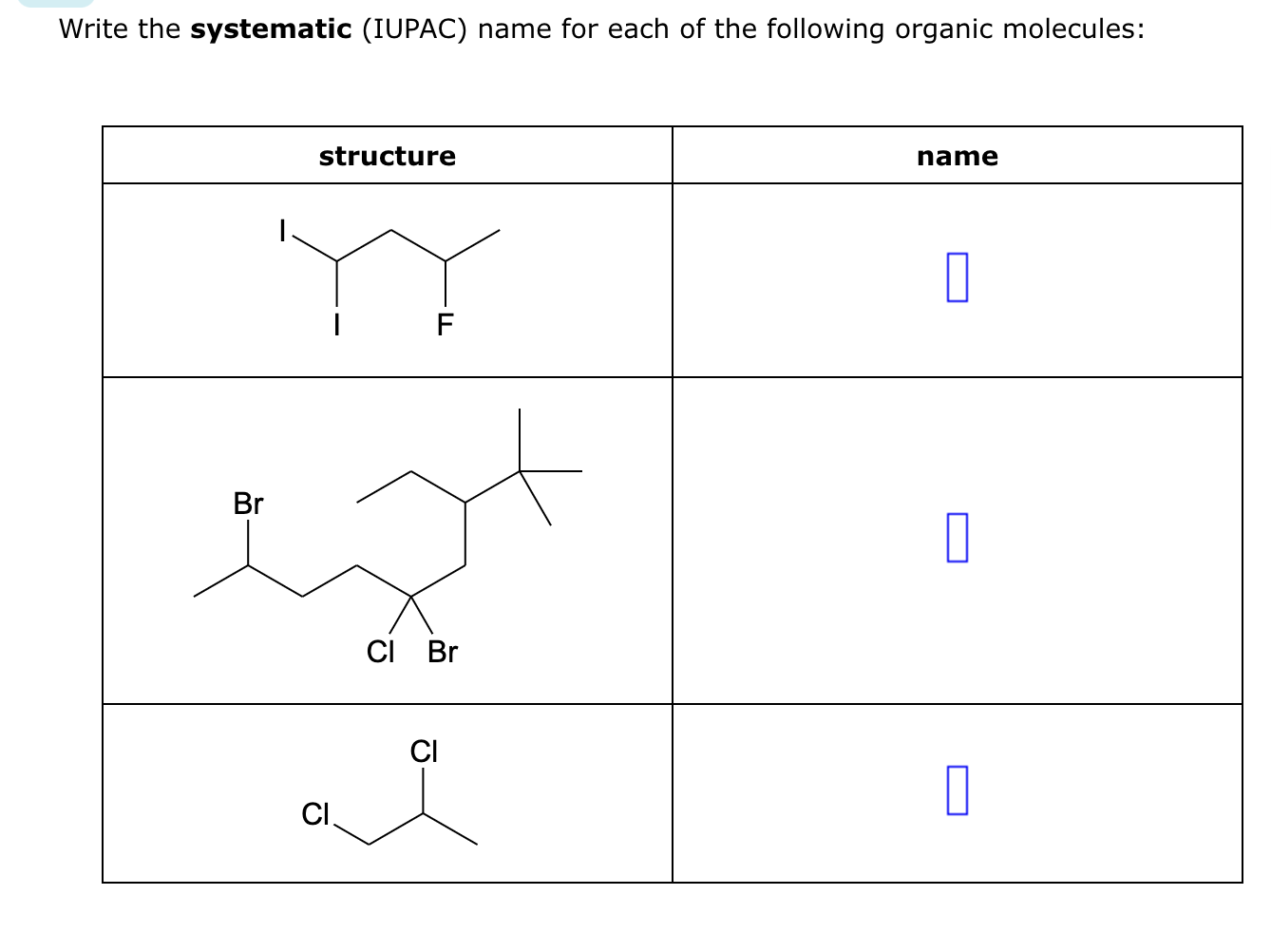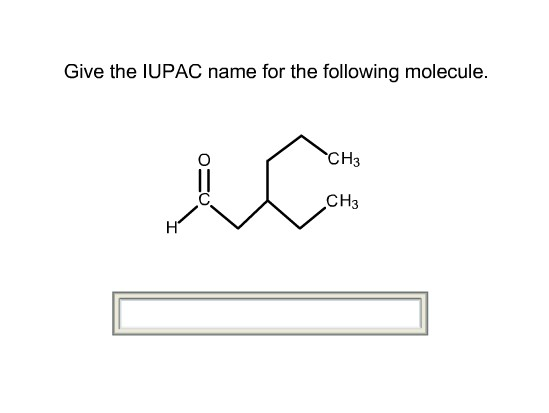
So, picture this. My friend, bless her chemically-challenged heart, was trying to bake a cake the other day. She’s usually a whiz in the kitchen, but this time, something went spectacularly wrong. She’d accidentally grabbed a container of baking soda and, instead of the sugar, decided to use a generous scoop of it. The result? A cake that looked more like a deflated, vaguely foamy science experiment than a dessert. She held it up, looking utterly bewildered, and asked me, "What is this thing?"
And that, my friends, got me thinking. We all encounter things – objects, foods, even abstract concepts – that we can’t quite identify. In the kitchen, it might be a weird ingredient or a failed culinary creation. In everyday life, it's that vaguely familiar tune you can't place, or that person you swear you've met before but can't put a name to. It’s this fundamental human need to label, to categorize, to give things names. And in the fascinating, sometimes baffling world of chemistry, this need is absolutely crucial. Today, we’re diving headfirst into the wonderfully organised chaos of naming molecules, specifically, one rather special little guy. Let’s call him… well, we’ll get to that. But first, we need to understand the system.
The Grand Unification of Molecule Names
Imagine a world where every single chemical compound had its own unique, quirky, made-up name. It would be utter pandemonium! You’d have “Fluffy-Cloud-Maker” for water, and “Sparkly-Boom-Powder” for something else entirely. Thankfully, for the sake of sanity and scientific progress, brilliant minds came up with a system. It’s called the International Union of Pure and Applied Chemistry (or IUPAC, if you want to sound like you know what you’re talking about) naming system. It’s basically a set of rules, a chemical Rosetta Stone, that allows chemists all over the globe to identify a molecule just by its name, and vice versa.
It’s like a secret handshake for scientists. You see a name, you know the structure. You see a structure, you can (with a bit of practice!) derive the name. It’s elegant, it’s systematic, and it’s incredibly powerful. Think of it as a highly detailed address for each molecule. It tells you not only the main “street” it lives on but also the “house number,” the “floor,” and even the “apartment number” of every atom within it.
So, Who's Our Mystery Guest?
Alright, enough preamble. Let’s meet the molecule we’re going to name today. Drumroll, please. It's this little fella:
``` O // H3C-C-CH3 ```
Looks simple enough, right? Just a few atoms hanging out together. But even these simple structures have a proper, official name. And before we get to the IUPAC name, let’s talk about what we see here. We’ve got a central carbon atom bonded to an oxygen atom with a double bond (that’s the `//` bit), and then two other carbon atoms, each bonded to three hydrogen atoms (that’s the `H3C-` and `-CH3` bits). So, in essence, we have a carbon atom double-bonded to an oxygen, and then two methyl groups attached to that same carbon.
Now, if you’ve done any chemistry before, you might already have a hunch about what this is. It’s a pretty common functional group, a familiar sight in organic chemistry labs and, indeed, in the kitchen. But we’re not going for the common name here; we’re going for the full, unabridged, IUPAC experience.
Deconstructing the IUPAC Beast
The IUPAC system has a hierarchical structure. Think of it like building a house. You need a foundation, then walls, a roof, and then all the decorative bits. Similarly, IUPAC names have prefixes, a root word, and a suffix. The root word tells you the length of the longest carbon chain, the suffix tells you the type of functional group, and the prefixes describe any substituents (things hanging off the main chain).
Let’s break down our molecule piece by piece, following the IUPAC logic.
Step 1: Find the Longest Carbon Chain
This is our foundation. We need to identify the longest continuous chain of carbon atoms. In our little molecule:
``` O // H3C-C-CH3 ```
We have three carbon atoms arranged in a line, with the central carbon also attached to the oxygen. So, the longest continuous carbon chain is three carbons long. This is our starting point for determining the root name.
Step 2: Identify the Functional Group
This is like choosing the type of house you’re building – is it a bungalow, a townhouse, a mansion? The functional group is the most important reactive part of the molecule. In our case, we have a carbon atom double-bonded to an oxygen atom. This is characteristic of a very important class of compounds called ketones.
Ketones have the general formula R-CO-R', where R and R' are alkyl groups (like our methyl groups) and the 'CO' part is the carbonyl group. The IUPAC suffix for ketones is -one.
Step 3: Determine the Root Name Based on the Carbon Chain Length
Now we combine the chain length with the functional group type. A three-carbon chain has the root name prop- (derived from propane, a three-carbon alkane). Since it’s a ketone, we add the suffix -one.
So far, we have propanone. It's a three-carbon chain with a ketone functional group. Pretty neat, huh?
Step 4: Numbering and Locating Substituents (If Any)
This is where we get into the nitty-gritty, the house numbers and apartment details. If there were other groups attached to our main carbon chain (like halogens, other carbon chains, etc.), we’d need to number the carbon chain to give these substituents the lowest possible numbers. However, in our specific molecule, we have a very symmetrical arrangement. The carbonyl group (the C=O) is right in the middle of our three-carbon chain.
Let’s visualize our three-carbon chain: C1-C2-C3. If we number from left to right, the carbonyl group is on carbon 2. If we number from right to left, it’s still on carbon 2. Since there’s only one possible position for the carbonyl group in a three-carbon ketone without it being at the end (which would make it an aldehyde), we don’t actually need to specify its position with a number. The name propanone inherently implies the carbonyl is on the second carbon. It’s like saying “two-bedroom apartment” when there’s only one way to arrange two bedrooms in that specific building layout.
So, to recap, we have a three-carbon chain (prop-) and it’s a ketone (-one). The carbonyl group is in the central position, which is the only possible location for a ketone in a three-carbon molecule. Therefore, no numerical locator is needed.
The Glorious, Official IUPAC Name Revealed!
Putting it all together, our molecule, with its central carbonyl group and two methyl groups attached, is officially named:
Propanone
There you have it! A perfectly functional, scientifically sound name for our little chemical friend. It’s not as flashy as “Sparkly-Boom-Powder,” but it’s infinitely more useful. It tells anyone who sees it exactly what the molecule looks like, its structure, and its chemical properties.
Now, you might be thinking, "Okay, that's all well and good, but does anyone actually call it propanone?" And this is where the irony of chemistry naming conventions often creeps in. While propanone is the correct IUPAC name, this particular molecule is so common, so ubiquitous, that it has a very widely accepted and frequently used common name: acetone. Yes, the stuff you use to remove nail polish! Chemistry can be a bit of a diva sometimes, insisting on its fancy IUPAC titles while the common names get all the airtime.
Why Does This Matter, Anyway?
You might be asking, "Why bother with these complicated names if there are simpler ones?" Great question! While acetone is familiar, imagine a molecule with ten carbons and several different functional groups. Trying to remember a common name for every single one would be an impossible task. IUPAC provides a universal language. It ensures that no matter where you are in the world, or what your native tongue is, you can communicate precisely about a chemical structure.
It’s also about clarity and avoiding ambiguity. Sometimes common names can be misleading or applied to different substances. IUPAC cuts through that confusion. It’s the bedrock of communication in fields like medicine, pharmaceuticals, materials science, and environmental chemistry. Without it, drug discovery would be a nightmare, new materials would be difficult to synthesize and describe, and understanding environmental pollutants would be a much harder task.
So, the next time you see a chemical formula, whether it’s for a life-saving drug, a delicious (or, in my friend’s case, not-so-delicious) food ingredient, or a material that makes up your phone, remember the IUPAC system. It’s the silent architect of chemical understanding, ensuring that we can all speak the same language, even when we’re talking about something as seemingly simple, yet as profoundly important, as a molecule.
And as for my friend's cake? Well, let's just say we ended up ordering pizza. Sometimes, even with the best naming systems, a culinary disaster is just a culinary disaster. But hey, at least now she knows the difference between sugar and baking soda! And if she ever accidentally creates a ketone with a more complex structure, she’ll have the tools to name it. You, my curious reader, now have those tools too. Go forth and name those molecules!