Hey there, curious minds! Ever wondered what makes life, well, work? It's a pretty wild thought, right? We're talking about the tiny building blocks that create everything around us, from the fluffy clouds in the sky to the bouncy ball you might have played with as a kid. And guess what? One of these fundamental pieces, a little guy named methionine, has a super neat trick up its sleeve. Today, we’re going to dive into a really cool idea: modifying methionine to show its zwitterion form. Sounds fancy, I know, but stick with me, it's seriously interesting!
So, first things first, what even is methionine? Think of it as one of the 20 essential amino acids. Amino acids are like the LEGO bricks of proteins, and proteins are the workhorses of our bodies. They do everything: build muscles, carry oxygen, fight off germs – you name it. Methionine is special because it's one of the building blocks that starts the protein-making process. It’s like the “start” button on your favorite video game, but for making life itself!
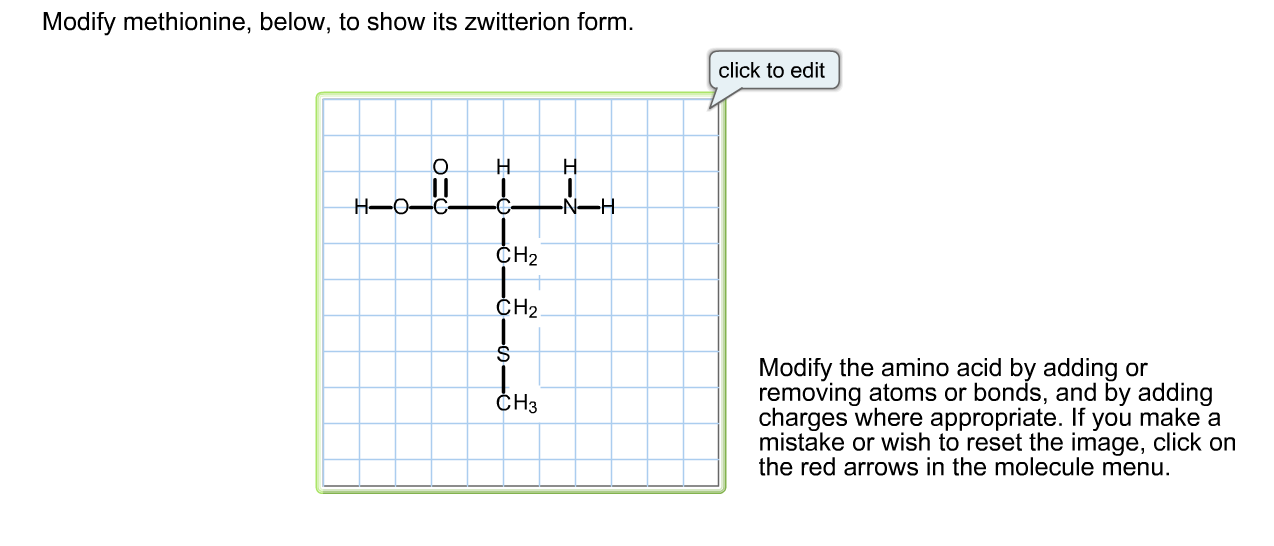
Now, what does it mean to "modify" something? In science, it usually means changing it a little bit, like adding a new feature or tweaking its structure. And what’s a "zwitterion"? This is where things get really fun. Imagine a tiny molecule that’s feeling a little conflicted. It’s got both a positive charge and a negative charge, all at the same time, but they're on different parts of the molecule. It’s like a tiny bipolar superstar!
Normally, when we think about molecules, they’re either positively charged, negatively charged, or neutral, right? Like a battery – it’s got a positive and a negative terminal. But a zwitterion is like having a tiny, perfectly balanced internal battery. It’s electrically neutral overall, but within itself, there's this fascinating push and pull of charges.
So, why would we want to make methionine, this crucial amino acid, into a zwitterion? Well, the natural form of methionine in our bodies and in many biological systems is actually a zwitterion! Isn't that cool? It's not like we're forcing it into something it never knew. We're just exploring and understanding a state it can exist in. Think of it like learning a new dance move – the basic steps are there, but you can refine and play with them to create something even more elegant.

But sometimes, to really understand how something works, scientists like to isolate and study different aspects of it. So, imagine we're trying to get a really good look at the zwitterionic personality of methionine. We might need to create specific conditions or make tiny adjustments to its environment to emphasize this zwitterionic form. It's a bit like asking a shy friend to stand in the spotlight so you can see all their amazing qualities more clearly.
How do we even do this? It often involves playing with the pH of the solution. pH is basically a measure of how acidic or basic something is. Think of it like a dial that can go from super sour (acidic) to super soapy (basic). Different molecules behave differently at different pH levels. By carefully adjusting the pH, we can create an environment where methionine’s positive and negative charges are perfectly balanced, making that zwitterion form shine!

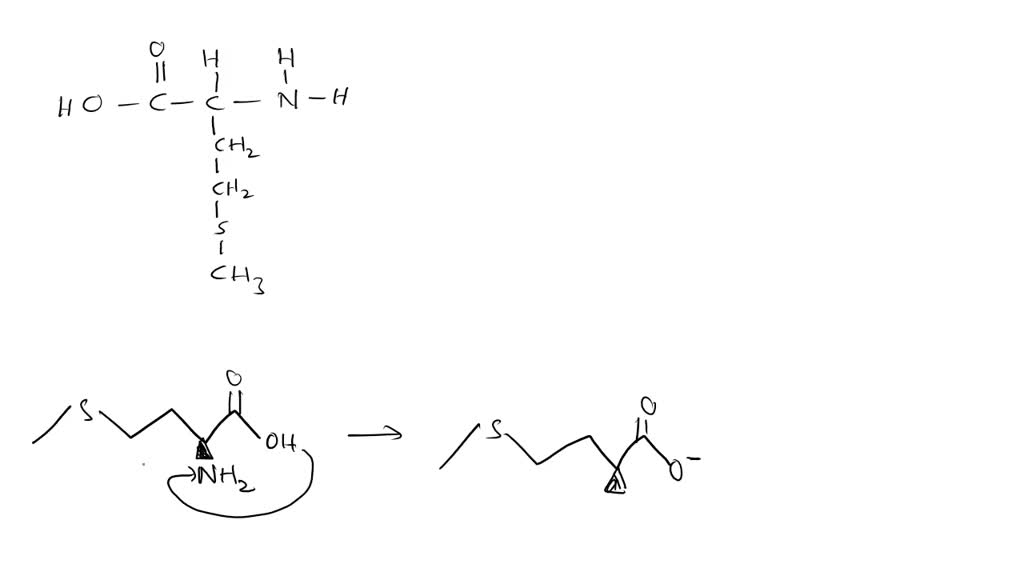
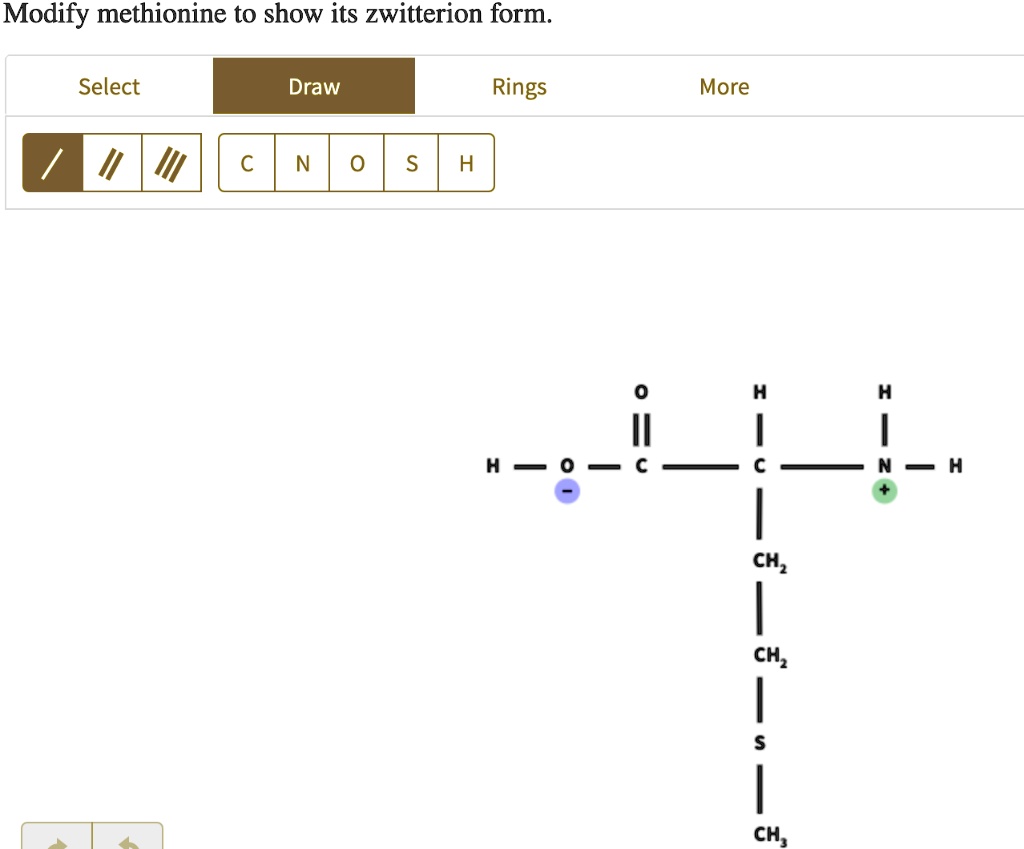
Let’s break down methionine's structure for a second. It has a central carbon atom, which is like the heart of the molecule. Attached to this carbon are a few key groups. There’s a carboxylic acid group (-COOH), which can lose a proton and become negatively charged (-COO⁻). And there's an amino group (-NH₂), which can gain a proton and become positively charged (-NH₃⁺). When these two groups are balanced, and the molecule is sitting at its "isoelectric point" (fancy term for the pH where it's most zwitterionic), voilà! Zwitterion time!
Why is this important, you ask? Well, understanding the zwitterionic form of methionine helps us understand how it interacts with other molecules. Remember those protein-making machines in our cells? The way methionine fits into the growing protein chain is heavily influenced by its charge. If it's in its zwitterionic form, it has a specific way of attracting or repelling other charged bits, guiding the protein to fold correctly.
Think about building with magnets. If you have a magnet that's neutral, it behaves one way. But if it has a positive and negative end, it'll interact with other magnets in a much more specific and dynamic way. Methionine's zwitterionic nature allows it to be a precise player in the complex dance of biochemistry.
Furthermore, by being able to create and study this zwitterionic form in a controlled way, scientists can design experiments to test specific hypotheses. They can look at how methionine behaves when it's definitely in this charged state, compared to when it might be in a more purely acidic or basic form. This helps them get to the heart of its functionality.
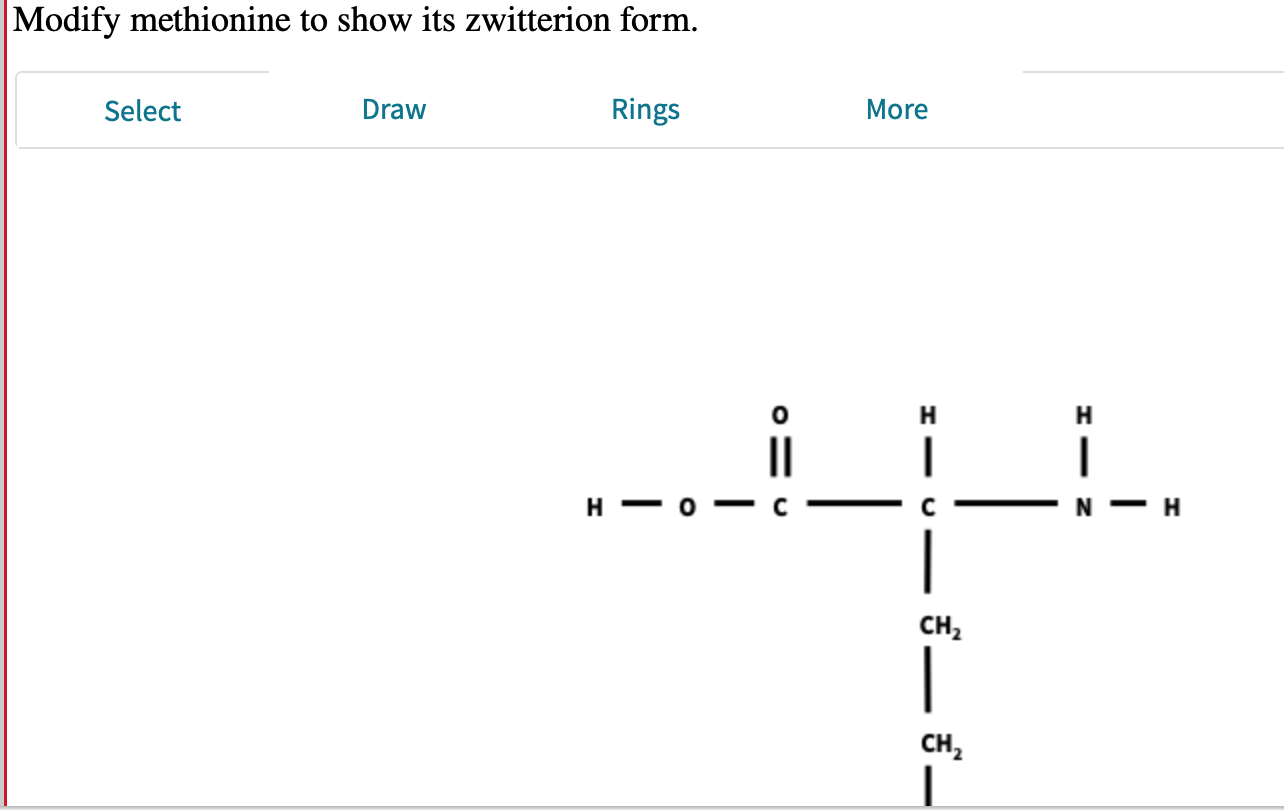
It’s like being a detective for molecules! You’re not just looking at the molecule itself, but you’re examining its different "attitudes" or "moods" depending on its surroundings. And the zwitterionic mood of methionine is a really crucial one for its job in life.
This kind of research also has practical applications. Understanding how amino acids behave, especially in their charged forms, can help in developing new drugs, designing better biomaterials, or even improving food science. For instance, if we want to create a protein-based material that sticks to a certain surface, knowing how the amino acids within that protein interact due to their charges is key.
So, the next time you think about the complex tapestry of life, remember methionine and its cool zwitterionic trick. It’s a testament to the elegant simplicity and incredible complexity of the molecular world. It reminds us that even the smallest building blocks have fascinating stories to tell, and by playing with their forms, we can unlock even more of nature's secrets. Pretty neat, huh?