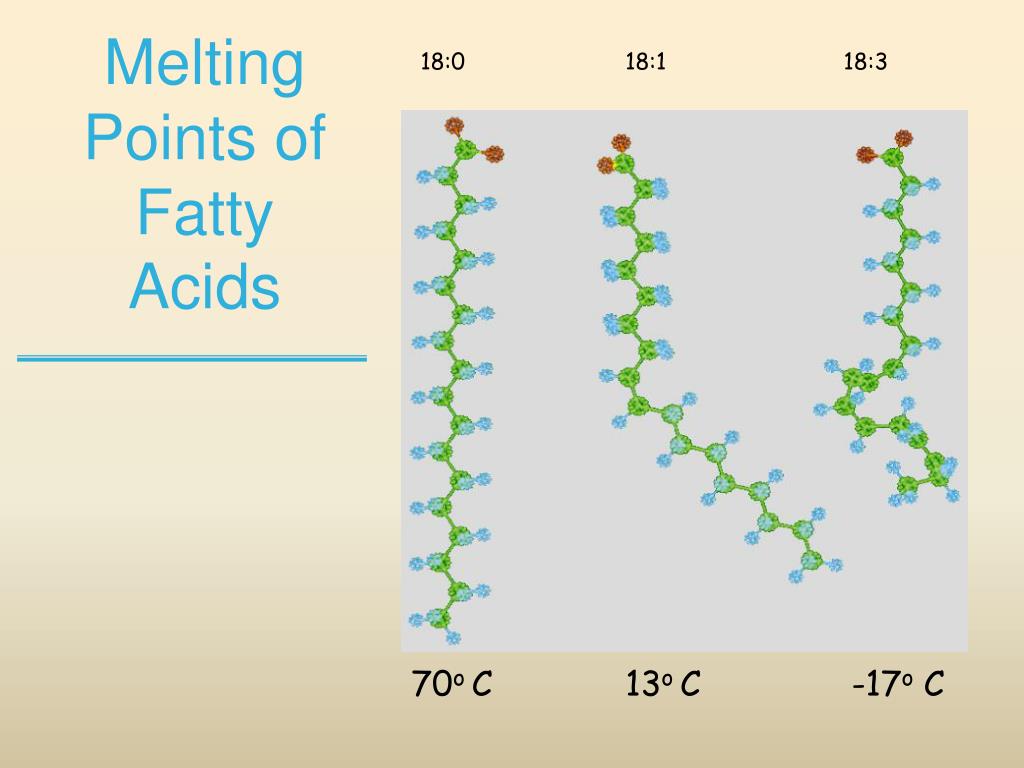
Okay, confession time. I have a secret, slightly nerdy obsession. It involves... fats. Yes, those slippery, sometimes-vilified molecules that make our food taste amazing. Now, before you click away thinking this is about calorie counting, hold up! My fascination is a little more… chilling. Literally. We're talking about their melting points.
Think of it like this: you know how ice melts when it gets warm? Fats do that too! But instead of a nice, predictable 0 degrees Celsius (that's 32 Fahrenheit for my Fahrenheit-inclined friends), different fats have their own little melt-times. It's like a slow-motion party for molecules, and I find it oddly fascinating. Don't judge me.
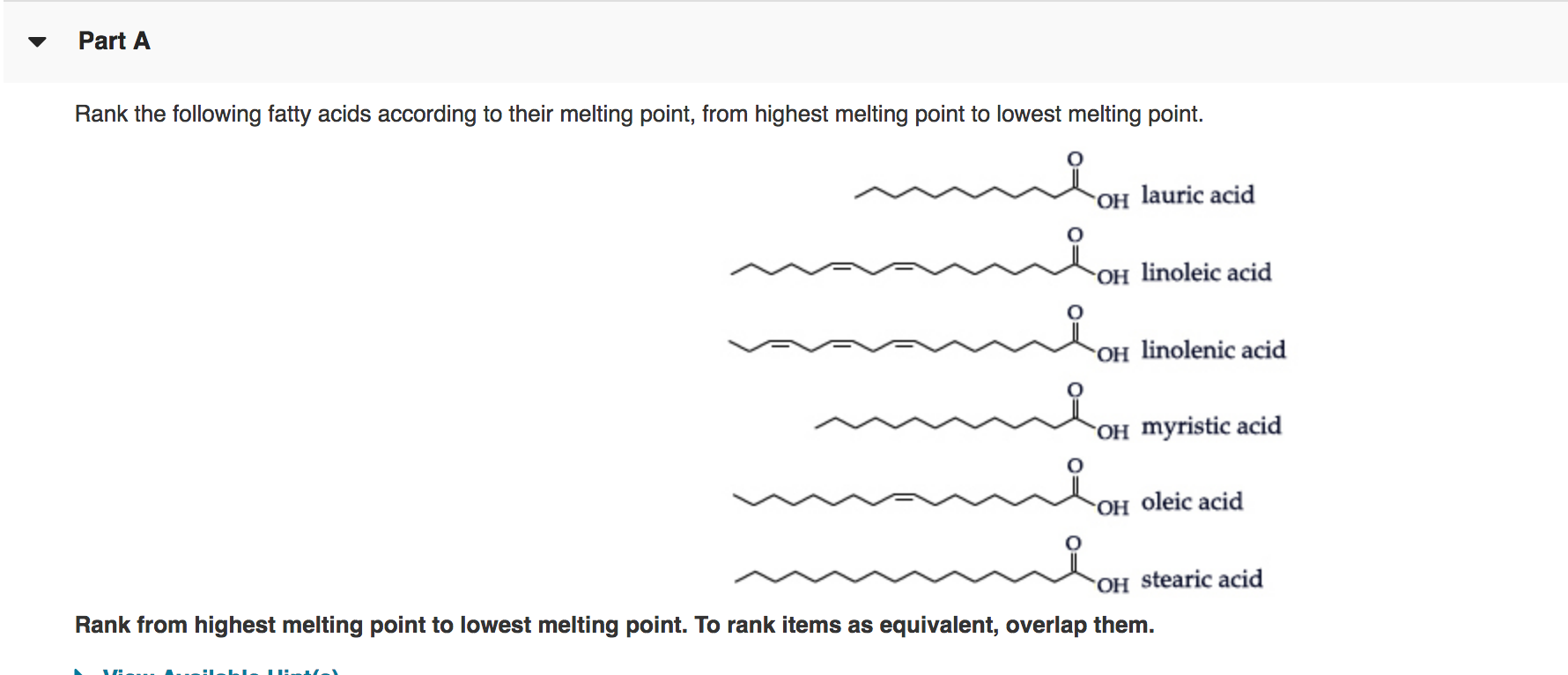
So, let's play a little game. I've got some common fatty acids here, the building blocks of all those delicious fats. And I've got a list of melting points. Your mission, should you choose to accept it (and why wouldn't you, it’s all about food!), is to play matchmaker. It's like a culinary dating app, but with chemistry.
The Contenders: Fatty Acids!
First up, we have Stearic Acid. This guy is a bit of a heavyweight. You find him chilling in lots of animal fats, like butter and lard. He’s pretty solid, both in personality and in temperature. He likes to hold his ground. Think of him as the dependable, slightly stoic friend. He doesn't get flustered easily.
Then there's Palmitic Acid. Another saturated fatty acid, and a very common one indeed. Palmitic acid is like the workhorse of the fat world. It’s found in palm oil, as you might guess, but also in a gazillion other things. It's got a good presence. It’s not as unyielding as Stearic, but it's still not exactly a wilting flower.
Now, let's dip into the world of unsaturated fatty acids. First, we meet Oleic Acid. Ah, Oleic! This is the star of olive oil, that liquid gold. It's monounsaturated, meaning it has one little double bond where it could have had more hydrogens. This little kink makes it more flexible, more… fluid. It's the charmer, the smooth operator. It's not afraid to bend a little.
Next, we have the double-bonder extraordinaire, Linoleic Acid. This one's an omega-6 fatty acid, often found in vegetable oils like sunflower and soybean oil. It's got two double bonds, making it even more bendy than Oleic. Linoleic is the adventurous one, the free spirit. It’s always looking for new experiences, which translates to a lower melting point.
And finally, let’s welcome Alpha-Linolenic Acid. This is the omega-3 cousin, found in things like flaxseed and canola oil. It's got three double bonds! Three! That's a whole lot of kinks. Alpha-Linolenic is the ultimate free spirit, the bohemian of the fatty acid family. It’s practically a liquid at room temperature.

The Melting Pot: Temperatures!
Now for the melting points. We've got:
- 70°C (158°F)
- 63°C (145°F)
- 13.4°C (56.1°F)
- 5.3°C (41.5°F)
- -5°C (23°F)
So, who’s going to melt at what temperature? It’s a bit like a personality test for fats, really. The more kinks (double bonds) a fatty acid has, the less neatly its molecules can stack together. And when they can’t stack neatly, they don’t need as much heat to start wiggling around and becoming liquid. It's like trying to stack a pile of perfectly straight logs versus a pile of twisty branches – the branches are much harder to get organized!
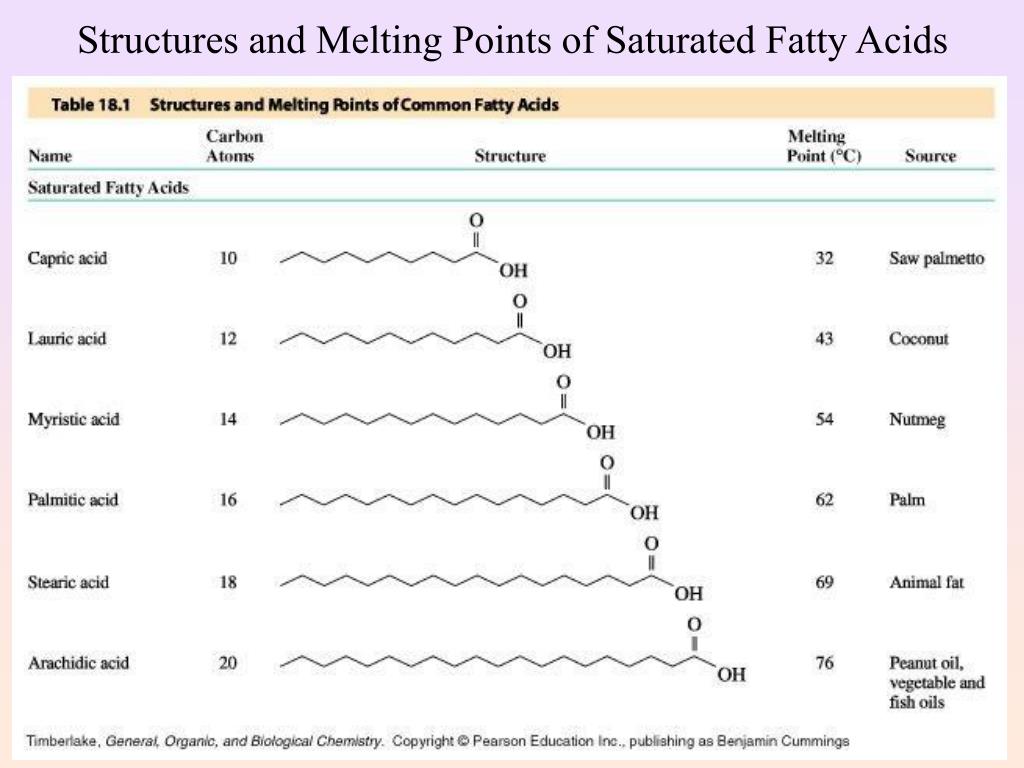
Let's start with the most rigid, the one that takes the most convincing to melt. That would be our friend Stearic Acid. He's a saturated fat, no kinks at all. He's packed tightly, like sardines in a can. He needs a serious oven to get going. So, Stearic Acid, you're matching with 70°C (158°F). A well-deserved retirement temperature, I say!

Next up is Palmitic Acid. Also saturated, but a tiny bit less tightly packed than Stearic. He's still pretty solid, but he'll give in a little sooner. Palmitic, you're joining the club at 63°C (145°F). A respectable showing!
Now, the unsaturated ones start to loosen up. Let's look at Oleic Acid. This is our single-kink wonder. It’s that lovely olive oil component. It’s liquid at room temperature, but it does have a point where it’ll start to solidify if things get really cold. Oleic Acid, you're the smooth operator, melting at 13.4°C (56.1°F). Just above freezing, like a cool breeze.
Then we have Linoleic Acid. This one has two double bonds. More kinks, more flexibility. It's found in many common cooking oils. Linoleic Acid, you're practically a liquid in most kitchens, and your melting point reflects that. You're matching with 5.3°C (41.5°F). Getting chilly!
And finally, the reigning champion of fluidity, Alpha-Linolenic Acid. With three double bonds, this guy is all about the wiggles. It’s the most sensitive to temperature changes. Alpha-Linolenic Acid, you're practically a liquid in your sleep. Your melting point is a frosty -5°C (23°F). Brrr! You're the one who makes your fridge look like a freezer!
There you have it! A little peek into the surprisingly exciting world of fatty acid melting points. It's not just about what they do in your body, or how they make your cookies crumbly. It's also about their unique personalities, their willingness (or unwillingness!) to loosen up when things get warm. And honestly, isn't that a little more fun than just seeing them as numbers on a nutrition label? I think so.