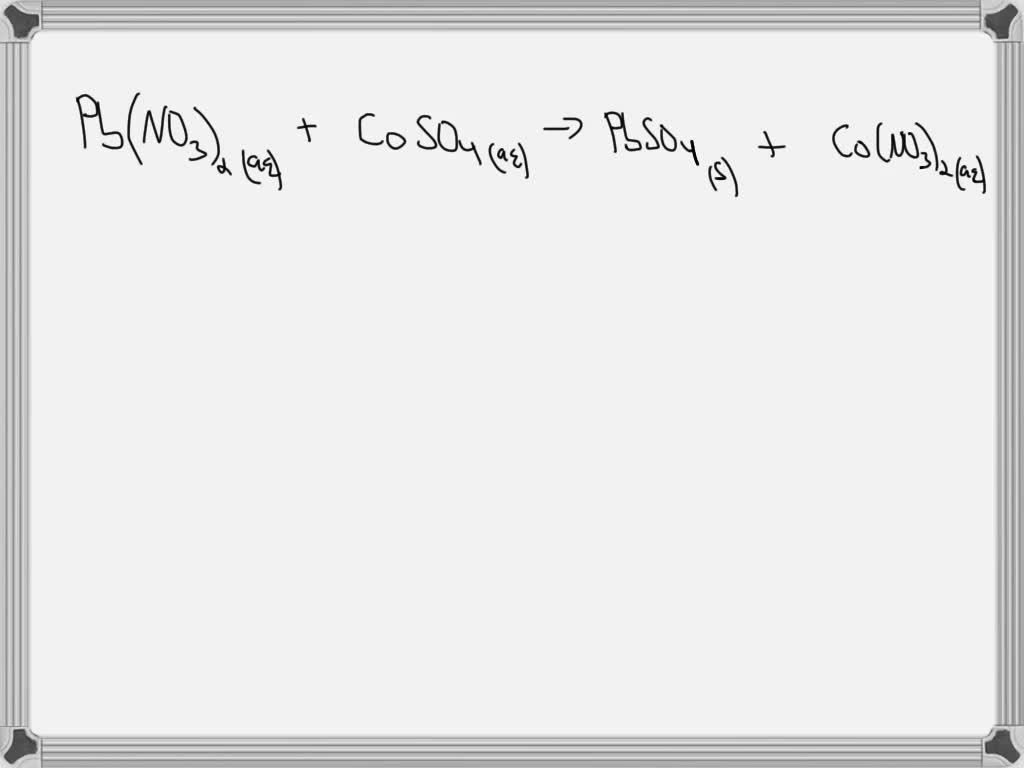Hey there, lovely people! Ever feel like science is this big, scary monster lurking in a lab coat, muttering about things like "molecular equations" and "reactants"? Yeah, me too sometimes. But what if I told you that even those fancy-sounding terms can have a sprinkle of everyday magic in them? Today, we're going to chat about something called the lead nitrate sulfuric acid molecular equation. Now, before you picture bubbling beakers and explosions (though sometimes that is chemistry!), let's break it down into something we can all get our heads around.
Think of chemistry like cooking. You've got your ingredients, right? And when you mix them in a certain way, you get a delicious meal. Chemistry is kind of the same, but instead of flour and eggs, we have tiny little building blocks called molecules. And just like you can't just throw random stuff in a pot and expect a gourmet dish, you can't just mix any molecules together and expect something cool to happen. You need a recipe – and that’s where the molecular equation comes in.
Our star players today are lead nitrate and sulfuric acid. Now, lead nitrate sounds a bit dramatic, doesn't it? Like something out of a gothic novel. But in the real world, it's a white crystalline solid. Think of it like a super-concentrated sugar that you definitely wouldn't want to put in your coffee! Sulfuric acid, on the other hand, is a strong acid. We use it in things like car batteries, but it's also a powerful cleaning agent. So, these are not your average kitchen ingredients, for sure!
When we talk about the lead nitrate sulfuric acid molecular equation, we're essentially writing down the recipe for what happens when these two ingredients meet. It's like telling a story about how they interact. In this story, lead nitrate is one of our main characters, and sulfuric acid is the other. They're going to have a little chemical dance, and something new is going to be made.
Imagine you have two groups of friends, Group A and Group B. Group A has two people, let's call them Alex and Brenda. Group B also has two people, Charlie and Daisy. Now, what if Alex and Charlie really hit it off? And Brenda and Daisy decide they'd rather hang out with each other? The molecular equation is like saying, "Okay, Alex and Brenda were together, and Charlie and Daisy were together. But when they got mixed, Alex paired up with Daisy, and Brenda paired up with Charlie!" See? A little bit of swapping and rearranging.
In our lead nitrate and sulfuric acid situation, the molecules are made up of even smaller bits called ions. Lead nitrate is made of lead ions and nitrate ions. Sulfuric acid is made of hydrogen ions and sulfate ions. When they get together, it's like those groups of friends. The lead ions and sulfate ions decide they make a great pair, and the hydrogen ions and nitrate ions also form a new, happy couple.
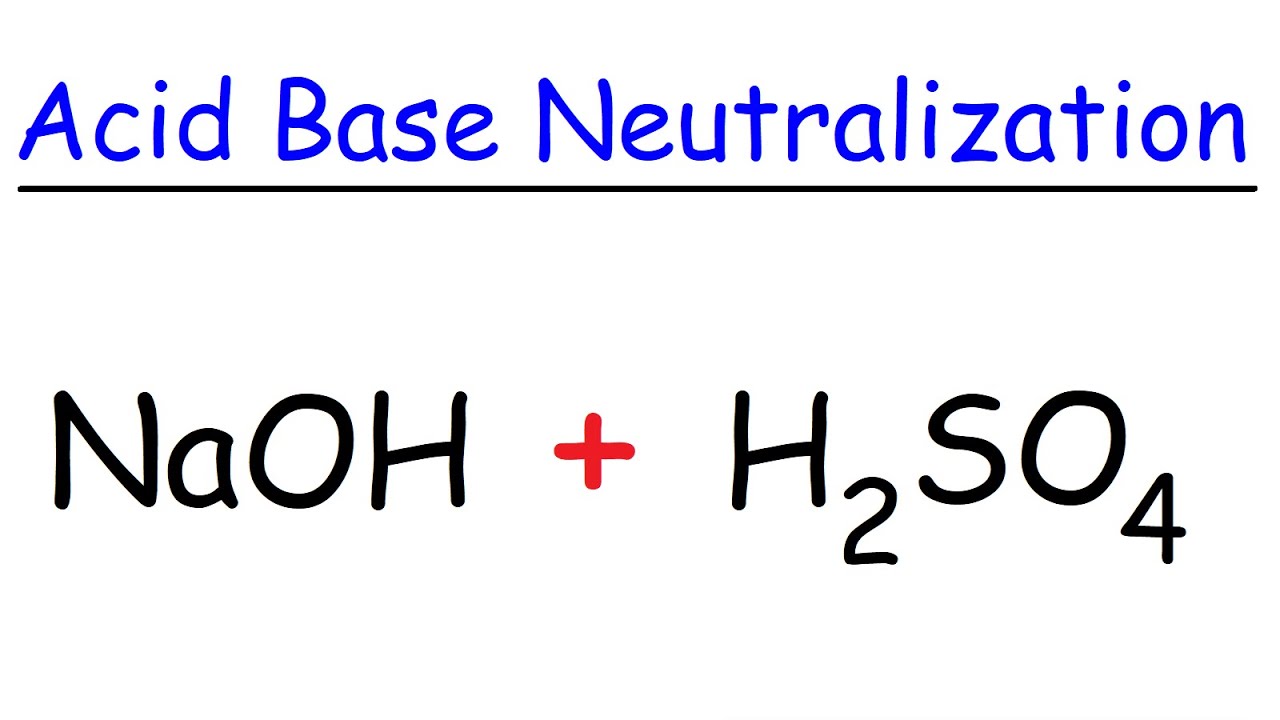
So, the lead nitrate sulfuric acid molecular equation shows us that lead nitrate (Pb(NO₃)₂) and sulfuric acid (H₂SO₄) react to form lead sulfate (PbSO₄) and nitric acid (HNO₃). It’s like the old partners splitting up and finding new ones. Pretty neat, right?
Now, you might be thinking, "Okay, that's cool, but why should I, a person who probably hasn't touched a chemistry textbook since high school, care about lead sulfate and nitric acid?" Well, that's a fantastic question! It's all about understanding how the world around us works, even the bits we don't see.
For example, lead sulfate is a pretty important compound. It's a white solid and, importantly, it's insoluble in water. This means it doesn't dissolve easily, like sand in water. This insolubility is actually a big deal. Think about it: if you're trying to get rid of something harmful, like lead, making it insoluble is a way to help keep it from spreading around. In environmental cleanup, understanding reactions that produce insoluble compounds is super important for safely dealing with pollution.

And then there's nitric acid. While it's not something you'd whip up in your kitchen, nitric acid has a whole bunch of uses. It's used in making fertilizers, which help our food grow. It's also used in making explosives (don't worry, that’s a whole other story!), and in some metal cleaning processes. So, even though we're talking about a specific chemical reaction, the results of that reaction have real-world impacts on things like agriculture and industry.
Think of it this way: you know how sometimes a small change in a recipe can completely alter the taste of your favorite dish? Maybe you accidentally added too much salt, or maybe you forgot the vanilla extract. Chemistry is like that, but on a much, much smaller scale. The way molecules interact and rearrange can have huge consequences for the materials and processes that shape our lives.

The lead nitrate sulfuric acid molecular equation is just one tiny example of this grand chemical dance. It’s a way for scientists to precisely describe what's happening at the molecular level. It’s their shorthand for saying, "These things went in, and these things came out." And understanding this shorthand helps them predict what will happen when other chemicals are mixed, or how to create new materials, or how to clean up the environment.
It's like learning a new language. Once you understand a few basic phrases, you can start to read books, watch movies, and understand conversations. With chemistry, once you understand these basic molecular equations, you start to see the intricate workings of the world in a whole new light. You begin to appreciate how everything is connected, from the tiniest atom to the vastness of our planet.
So, the next time you hear about a "molecular equation," don't let the big words intimidate you. Remember the friends swapping partners, or the ingredients in a recipe. The lead nitrate sulfuric acid molecular equation is just a story about molecules doing their thing, a story that helps us understand how things are made, how things change, and ultimately, how our world functions. And that, my friends, is pretty darn cool, wouldn't you agree?