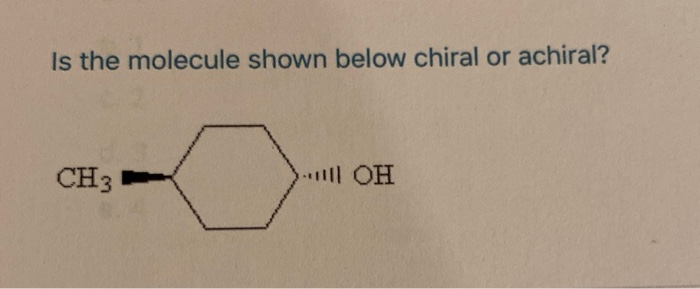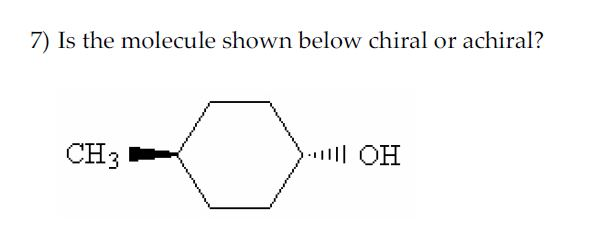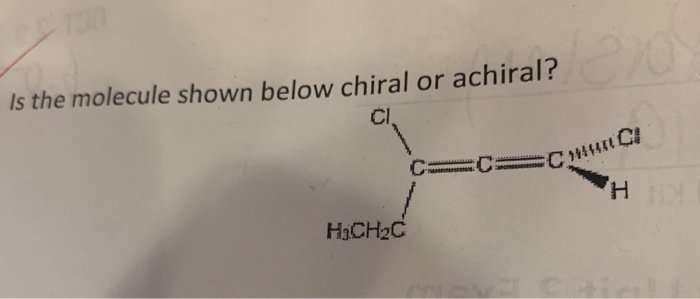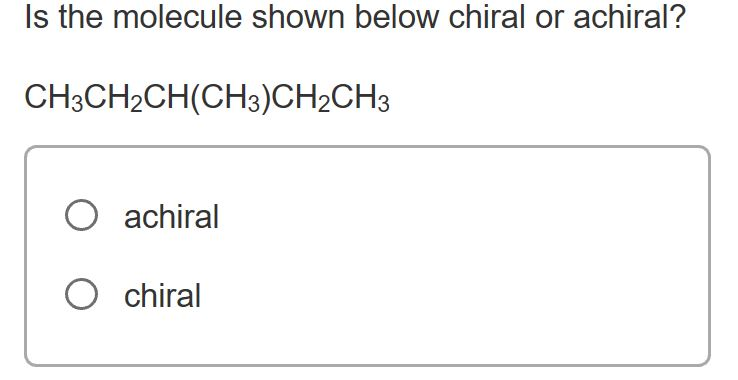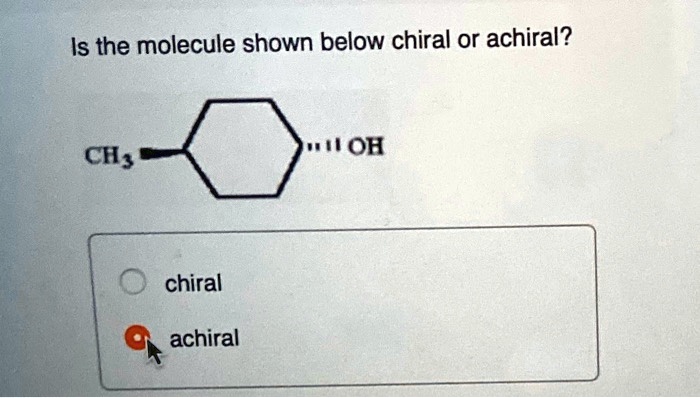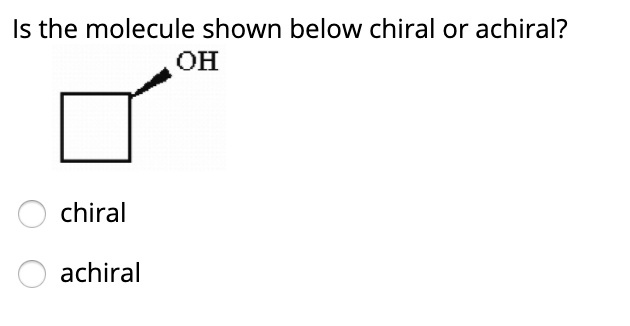
Imagine you're holding a pair of your favorite sneakers. You've got the left one, and you've got the right one. They look almost identical, right? But try to put the left sneaker on your right foot – it just doesn't feel right, does it? They're like twins, but one is definitely the mirror image of the other. This is a little bit like what we're talking about when we discuss whether a tiny, invisible thing called a molecule is chiral or achiral.
Now, I know "chiral" sounds like something you'd find in a dusty old chemistry textbook, and maybe it is. But stick with me, because it's actually a concept that pops up in some surprisingly delightful places. Think of it like this: a chiral molecule is like our sneaker – it has a distinct left-handed and right-handed version. They are non-superimposable mirror images of each other. An achiral molecule, on the other hand, is like a perfectly symmetrical coffee mug. If you hold it up to a mirror, its reflection is exactly the same, and you can lay the reflection right on top of the original. No matter how you twist it, it always matches itself. It's… well, it's just plain.
So, how do we figure out if a molecule is a fancy sneaker or a plain mug? We look at its structure. Specifically, we look for a special kind of carbon atom. This isn't just any carbon atom; it's a carbon atom that’s playing host to four different friends – four different groups or atoms attached to it. If a molecule has a carbon atom like this, and it can't be flipped or rotated to perfectly match its mirror image, then BAM! We've got ourselves a chiral molecule. It’s a molecule with a twist, a personality, a definite handedness.
Why should we care about this molecular handedness? Well, it turns out that nature is a HUGE fan of chirality. Many of the building blocks of life – like our DNA and the proteins that do all the work in our bodies – are chiral. They exist in only one specific handedness. Think about it: your body is built from these chiral molecules. They fit together in very specific ways, like a lock and key. If you try to use the wrong key (the wrong handedness of a molecule), it just won't work.
This is why, for example, some medicines are chiral. A drug that works wonders in its "right-handed" form might do absolutely nothing, or worse, cause harmful side effects in its "left-handed" form. It's like trying to put on a glove designed for your right hand onto your left. It's just not going to fit properly, and things can get a bit… awkward. Chirality in medicine is a big deal, and scientists spend a lot of time making sure they're using the correct "handedness" of a drug to make it as safe and effective as possible. It’s a tiny detail with enormous consequences!
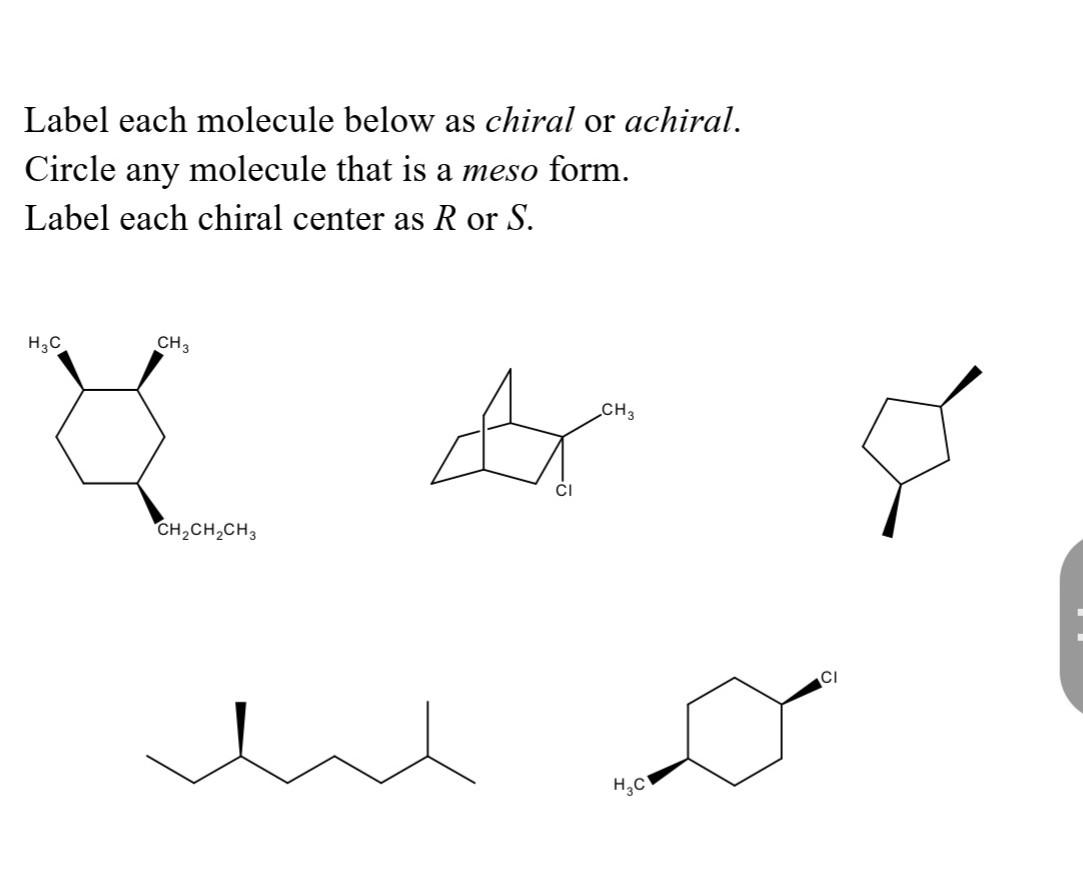
But it’s not all serious science and medicine. Chirality also plays a role in the things we enjoy every day. Did you know that the difference between a lemon's zest and a lime's zest, or even the way we perceive smells, can be due to chirality? Some molecules that smell like lemons are actually different "handed" versions of the same basic molecule that smells like limes! Isn't that wild? A tiny change in how atoms are arranged in space can completely alter how we experience the world through our senses. It’s like having a secret code that dictates whether something tickles your nose with citrusy delight or a more pungent aroma. The sense of smell, that marvelous, often overlooked sense, is deeply intertwined with molecular handedness.
Consider the humble amino acids, the building blocks of proteins. Almost all the amino acids used in life are “left-handed.” This is a fundamental rule of biology. Imagine a world where all our proteins were built with the "wrong" amino acids. It would be a bizarre, non-functional mess. Our bodies wouldn't be able to build muscles, digest food, or even think! It’s a testament to how precisely nature orchestrates these molecular interactions. It’s like a cosmic dance where every molecule has its designated step and partner. When the steps are right, life flourishes. When they're wrong, well, things fall apart.
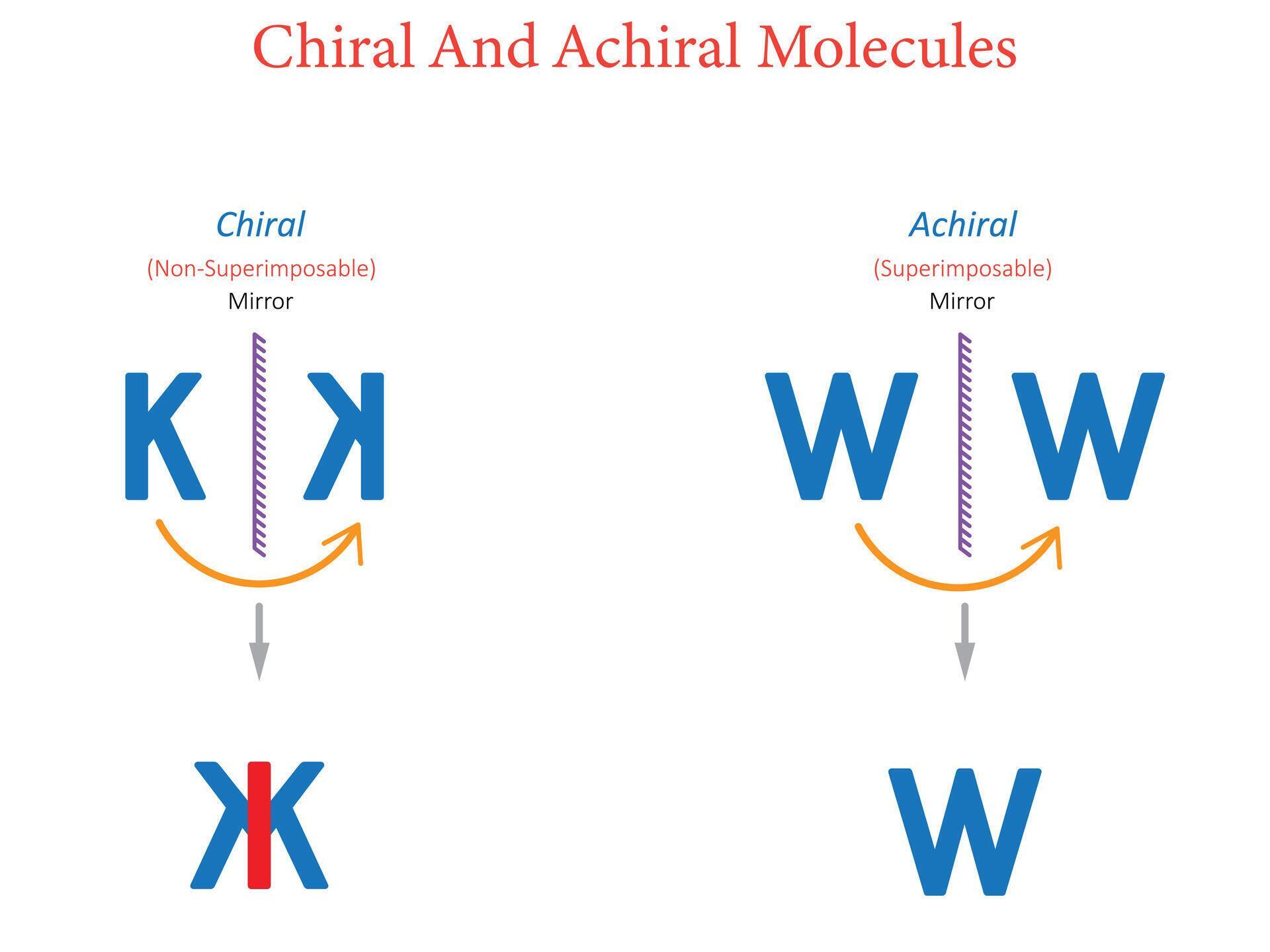
So, the next time you look at a molecule, don't just see a jumble of atoms. Think about its potential for "handedness." Is it a sturdy, symmetrical coffee mug, perfectly happy being itself no matter how you look at it? Or is it a sleek, sophisticated sneaker, with a distinct left and right, ready to play its part in the grand symphony of life? It’s a hidden world of molecular personality, a subtle but significant difference that shapes everything from the medicines we take to the scents that drift on the breeze. It's a reminder that even in the smallest, most invisible corners of existence, there's a fascinating story to be told, a story of right and left, of mirror images, and of the elegant complexity of the world around us. Chirality is a fundamental principle that underlines so much of what we understand about chemistry and biology, proving that even the most abstract scientific concepts can have a surprisingly tangible and delightful impact on our lives.
And if you were to show me a specific molecule right now, I’d be able to tell you if it was a chiral sneaker or an achiral mug!