Hey there! So, you've probably stumbled across the magical world of mass spectrometry, right? It's like this super cool science detective tool. And if you're knee-deep in organic chemistry, chances are you've heard whispers (or maybe even loud shouts) about the Mclafferty rearrangement. Yeah, that one. It's a bit of a tongue-twister, but oh-so-important for figuring out what's really going on inside those molecules. And, of course, when you're trying to understand it, you're probably looking for resources. Enter the humble PDF. Specifically, the Mclafferty PDF.
Now, I know what you're thinking. "Another PDF? Really?" But trust me, this isn't just any PDF. This is the key, the Rosetta Stone, the secret handshake to decoding those crazy zig-zag lines on your mass spec output. Think of it as your cheat sheet for winning at chemistry. And who doesn't want that, right? Especially when exams are looming, and your brain feels like it's been run through a centrifuge. Been there, done that, got the slightly-less-than-perfect grade.
So, What's the Big Deal About Mclafferty Anyway?
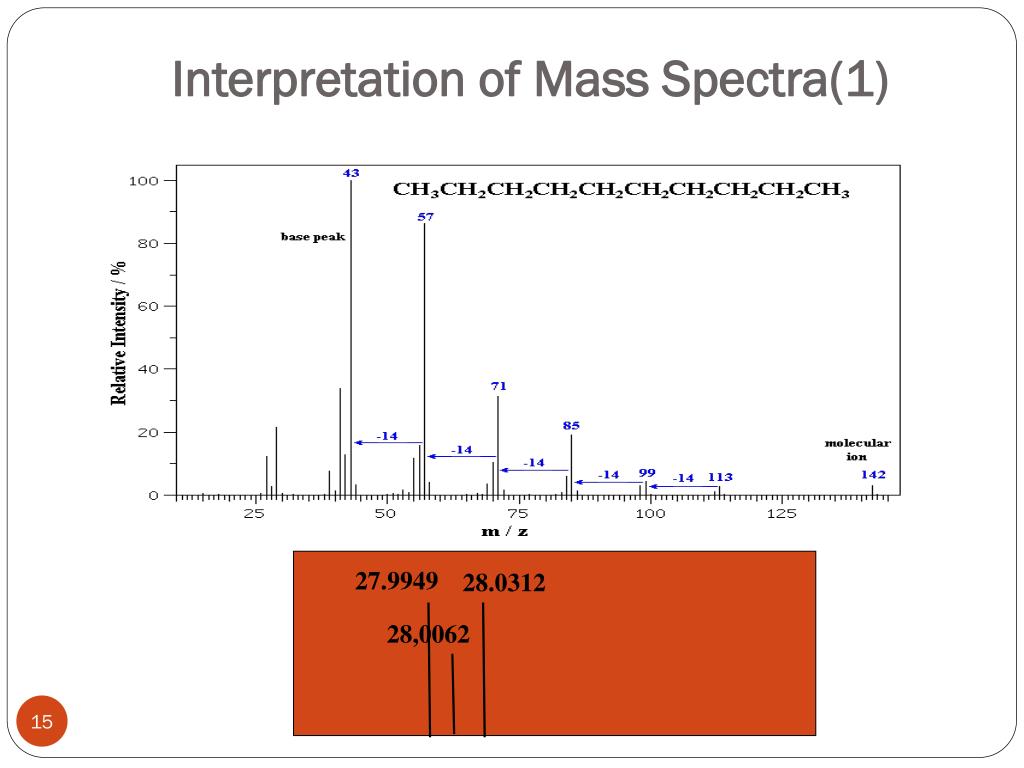
Okay, let's get down to brass tacks. Mass spectrometry, at its core, is all about zapping molecules and seeing what little bits fly off. When you zap 'em, they fragment. It's like when you drop a cookie – it doesn't just break into two perfect halves, does it? You get crumbs, big chunks, little bits. Mass spec is kind of the same, but with way less mess and a lot more data. And the Mclafferty rearrangement is this specific way certain molecules like to break apart. It's a predictable dance of electrons and atoms.
Imagine you have a molecule with a carbonyl group – you know, the C=O thing. And then, somewhere nearby, it's got a hydrogen atom hanging out on a carbon that's at least three atoms away. This little setup is like a perfectly poised domino. The energy from the mass spec blast is the push. What happens? The hydrogen hops over to the oxygen, and simultaneously, the molecule does this neat little six-membered ring thing and breaks apart. Poof! You get two new fragments. Revolutionary, right?
And the best part? This rearrangement produces a very specific type of fragment. It's called an enol fragment. They have this characteristic peak in the mass spectrum. So, if you see that peak, and you know your molecule has the right structural features, you can confidently say, "Aha! Mclafferty happened!" It’s like finding a fingerprint at a crime scene. Except, you know, less… forensic. More academic.
Why Is This Little Hop So Important?
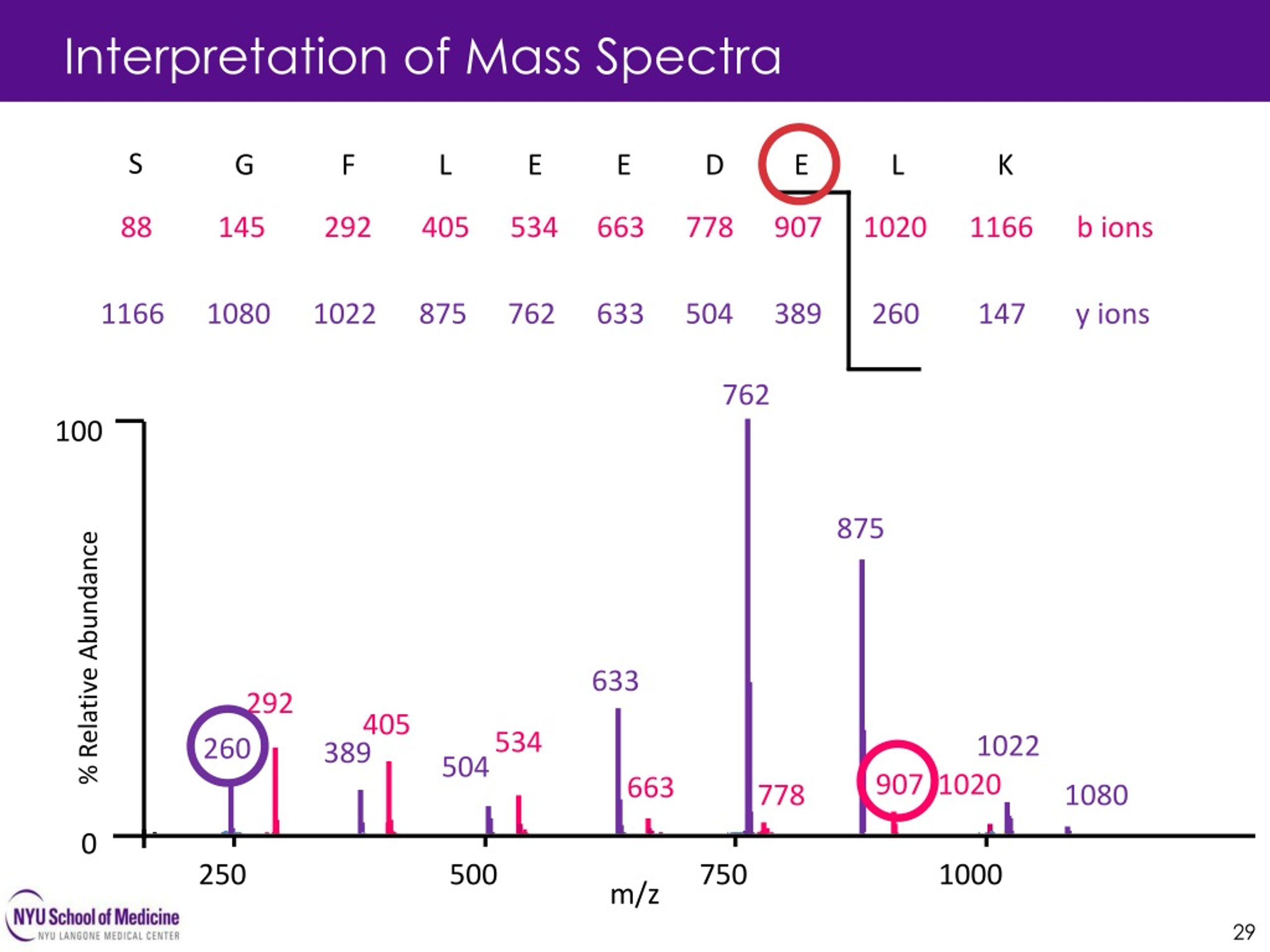
Well, for starters, it helps you identify unknowns. If you're given a mystery compound and you get a mass spectrum, looking for the Mclafferty peak can give you a huge clue about its structure. It's a structural fingerprint, remember? It tells you there's a carbonyl and a specific type of alkyl chain attached. Without understanding Mclafferty, you're basically staring at a bunch of squiggles and hoping for the best. And let's be honest, hoping isn't the best study strategy, is it?

It also helps you confirm structures. So, you've synthesized a new compound. You've done all the reactions, crossed your fingers, and now you need proof it's what you think it is. Mass spec is your go-to. And if your compound is capable of a Mclafferty rearrangement, seeing that characteristic peak is a sweet confirmation. It’s like the universe giving you a little nod of approval. “Yep, you did good, kid.”
And let’s not forget about elucidating reaction mechanisms. Sometimes, chemists are trying to figure out how a reaction happens. The fragments you see in a mass spec can tell you a lot about the intermediate stages and how the molecule broke down. Mclafferty’s contribution is a classic example of a well-understood fragmentation pathway. It's a building block for understanding more complex fragmentation patterns.
So, Where Do I Find This Magical Mclafferty PDF?
Ah, the million-dollar question! Or, you know, the slightly-less-than-a-million-dollar question, because usually, these things are free. You'll find them in a few key places. Most likely, your university library website is your best bet. They often have access to a gazillion journals and databases. Just do a quick search for "Mclafferty rearrangement mass spectrometry" and see what pops up. You might get lucky with a review article or even a dedicated chapter in an online textbook.

Then there's the ever-reliable Google Scholar. Seriously, this thing is a lifesaver. Type in "Mclafferty rearrangement mechanism PDF" or "interpretation of mass spectra Mclafferty" and prepare to be overwhelmed. You'll find papers, lecture notes, even old thesis chapters. Just be a little discerning. Some older PDFs might be a bit… quaint. But the fundamental principles are usually still solid. Think of it as a vintage wine; might have a bit of sediment, but the flavor is still there.
And of course, there are the dedicated chemistry education websites. Some professors are super generous and put their lecture notes online. These are often goldmines! They're usually simplified, focused on the key takeaways, and often come with diagrams. Diagrams! The unsung heroes of understanding chemistry. Seriously, a good diagram can save you hours of mental anguish.
You might even find PDFs specifically focused on interpreting mass spectra in general. These will usually have a section dedicated to common fragmentation patterns, and Mclafferty will be front and center. It's like a "greatest hits" album for molecule breaking. You'll learn about other cool fragments too, like alpha-cleavage or retro-Diels-Alder reactions. It’s a whole symphony of molecular destruction!
Tips for Navigating Your Mclafferty PDF
Okay, so you've found a PDF. Hooray! Now, what do you do with it? Don't just skim it like you're speed-reading the cereal box. Dive in! Here are a few pointers:

- Focus on the Diagrams: Seriously, the diagrams are your best friend. Trace the electron movements. See how the hydrogen hops. Watch the bonds break. It's a visual story.
- Identify the Key Players: Look for the conditions that allow for Mclafferty. Remember that gamma-hydrogen? It’s crucial! And that carbonyl group? Non-negotiable.
- Look for the Product Peaks: What m/z values (mass-to-charge ratios) are characteristic of the Mclafferty fragments? Memorize those. Or at least know where to find them in your PDF!
- Practice, Practice, Practice: The PDF is a guide, but the real learning happens when you apply it. Look at example spectra. Can you spot the Mclafferty peak? Can you predict it based on the structure?
- Don't Be Afraid of the "Why": Why does this rearrangement happen? It’s driven by the formation of a stable six-membered cyclic transition state. That’s a fancy way of saying it’s a really stable way for the molecule to break. Stability is king in chemistry, people!
And if your PDF is a bit dense, don't despair. Sometimes, you need to read a concept from a few different sources to really get it. One explanation might click where another didn't. It's like trying on different hats until you find the one that fits just right. Or in this case, the explanation that makes your brain go "Aha!" instead of "Huh?"
Beyond the PDF: Other Ways to Master Mclafferty
While a good PDF is a fantastic starting point, don't stop there! Think of it as your initial training montage. You need the actual workout to get buff.
Online Videos: YouTube is a treasure trove. Search for "Mclafferty rearrangement explained" and you'll find animated videos showing the process. It’s like watching a molecular ballet. Some of these are incredibly clear and can really solidify your understanding. I've spent hours just watching molecules do their thing in little animated clips. It's my guilty pleasure.

Practice Problems: Your textbook probably has a bunch of practice problems. Do them! Don't just look at the answers. Try to work them out yourself. If you get stuck, go back to your PDF, your notes, your videos. The struggle is part of the learning process, even if it feels like wrestling a slippery eel sometimes.
Study Groups: Talking through concepts with friends can be super helpful. You might explain it to them, and in the process, you'll realize what you truly understand and what you're still fuzzy on. Plus, you can commiserate about the late-night study sessions. Misery loves company, as they say. Or maybe it's just shared caffeine addiction.
Professor's Office Hours: Don't be shy! Your professor is there to help. If you're genuinely stuck on Mclafferty, go ask them. They've explained it a hundred times, so they can probably explain it to you in a way that makes sense. It’s like getting insider tips from the head chef.
Ultimately, the Mclafferty rearrangement is a fundamental concept in mass spectrometry. It's a reliable pattern that helps us unlock the secrets of molecular structure. And that PDF? It’s your starting point, your roadmap, your trusty companion on this journey of chemical discovery. So, grab your virtual coffee, find that PDF, and get ready to become a mass spec master. You got this!