Ever wonder what happens when you mix a few common ingredients and get a little chemistry show? It’s like a mini-science magic trick happening right before your eyes! Today, we're going to peek behind the curtain at one of these exciting reactions. It’s a real crowd-pleaser, and you don't need a lab coat to appreciate the spectacle. Think of it as a little performance, starring some familiar faces from the periodic table.
Let's talk about our main stars. We’ve got Zinc, which you might know as the shiny metal that’s sometimes used to coat other metals. It’s a bit of a solid performer, always ready to jump into action. Then, we have Hydrochloric Acid, or HCl. This one’s a bit feistier, a liquid that’s eager to get things moving. You might have heard of acids before; they can be quite reactive!
Now, imagine our buddy Zinc (that's Zn for its shorthand name) taking a dive into the bubbly world of Hydrochloric Acid (HCl). What happens next is where the real fun begins. It’s not just a quiet little fizz; it's a lively exchange, a chemical dance that produces something pretty neat. It’s like these two are meeting for the first time, and they’re really hitting it off, but in a very scientific way.
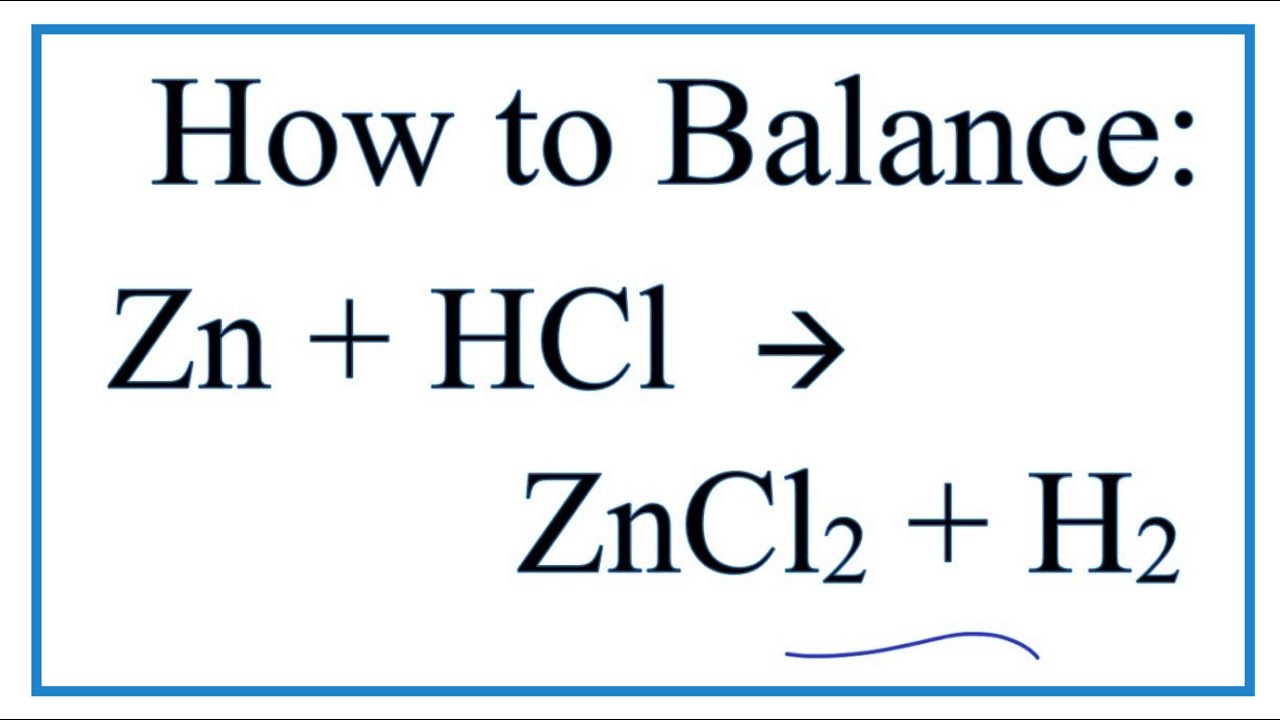
As Zinc and HCl get together, they start rearranging themselves. It’s not a complicated process, but the results are fascinating. They're not just hanging out; they’re actively participating in a swap. The Zinc atom decides to team up with the Chlorine part of the Hydrochloric Acid. So, goodbye, Hydrogen, it was nice knowing you! This new partnership forms something called Zinc Chloride (ZnCl2). You can think of Zinc Chloride as the new kid on the block, a compound that’s a result of this energetic meeting. It's stable and formed from the two main players.
But wait, there's more! Remember that Hydrogen that got swapped out? Well, it doesn't just disappear. It’s too energetic to sit on the sidelines. The Hydrogen atoms, which were once attached to the Chlorine in the HCl, now find themselves free. They’re like little unbound dancers, looking for a partner. And they find one in each other! Two Hydrogen atoms easily team up to form Hydrogen gas (H2). This Hydrogen gas is a real showstopper.
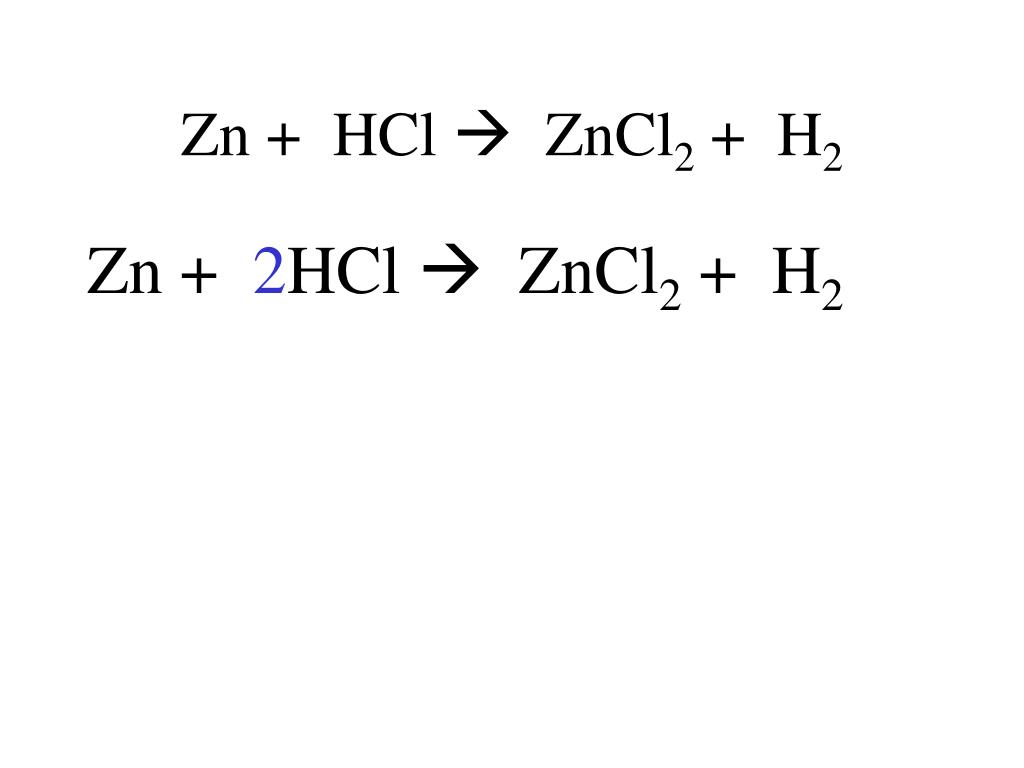
This is where the entertainment really kicks in. When Hydrogen gas is produced, it doesn't just sit there. It wants to escape! You'll see bubbles, lots and lots of bubbles, rising up. It’s like the reaction is breathing out, letting off some steam. This bubbling is a visual cue, a sign that something exciting is happening. It’s the reaction’s way of saying, “Look at me! I’m creating something new!”
And the gas itself? Hydrogen gas is actually very light. It's lighter than air. So, these bubbles are eager to float up and away. If you were to get really close (but safely, of course!), you might even hear a tiny little popping sound if you were to introduce a flame. This little ‘pop’ is the signature of Hydrogen gas, and it’s a classic chemical demonstration that gets people excited. It’s like a tiny celebration for the newly formed gas!

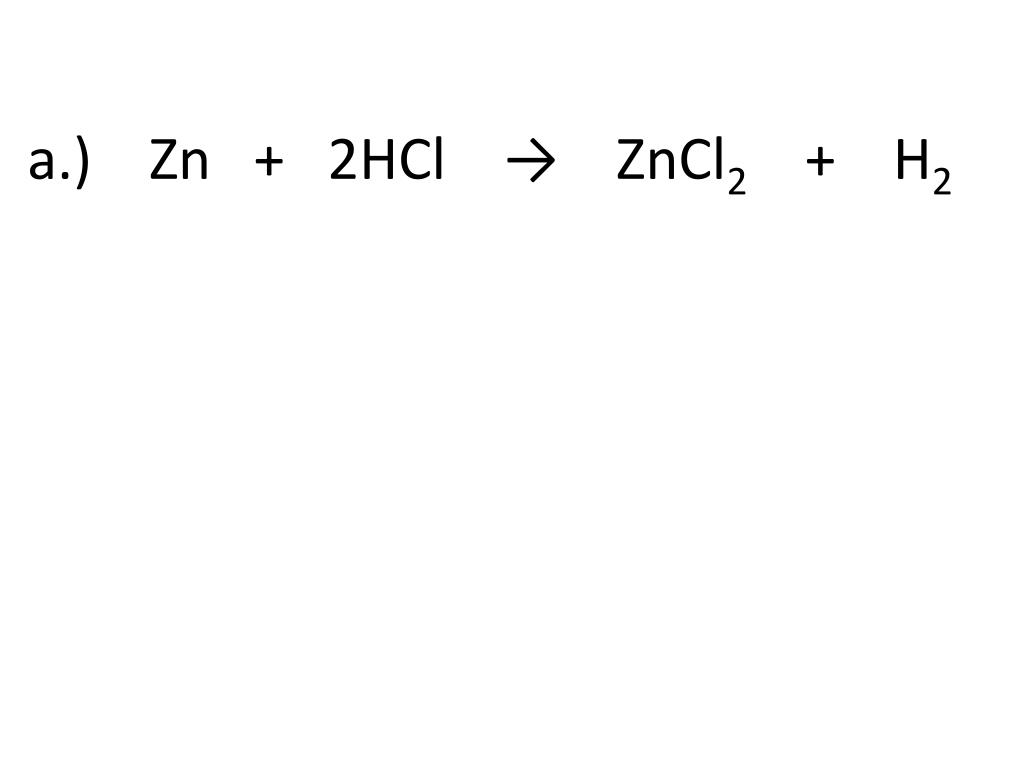
So, to recap our little drama: we started with Zinc (Zn) and Hydrochloric Acid (HCl). They got together, and in a whirlwind of chemical activity, they produced Zinc Chloride (ZnCl2) and Hydrogen gas (H2). It’s a neat transformation, a perfect example of a specific type of chemical reaction that’s not only important in science but also visually quite engaging.
What makes this so special? It’s the simplicity and the clear outcome. You can see the bubbles, you can imagine the escape of the gas. It’s a very tangible way to witness chemistry in action. It’s not some abstract concept; it’s a physical event that you can observe. This is why it’s often shown in introductory chemistry classes – it’s a fantastic illustration of how atoms and molecules interact and rearrange themselves.

It's like a tiny chemical party where new friends are made and some energetic guests are released!
This type of reaction is called a single displacement reaction. Think of it like this: the Zinc comes in and ‘displaces’ or kicks out the Hydrogen from the Hydrochloric Acid. The Zinc then takes the Hydrogen's place, forming Zinc Chloride. The kicked-out Hydrogen, being a bit of a free spirit, then pairs up with another Hydrogen to form Hydrogen gas. It’s a really neat way to visualize how elements can swap partners in a chemical compound.
The beauty of this specific reaction, Zn + HCl → ZnCl2 + H2, is its clarity. You start with two distinct substances, and you end up with two new substances, one of which is a gas that actively shows itself through bubbling. It’s a satisfying demonstration of chemical change. It’s a little bit of a spectacle, a quick burst of activity that showcases the fundamental principles of chemistry. So, the next time you hear about Zinc reacting with Hydrochloric Acid, remember the energetic dance, the bubbly escape, and the formation of new chemical friends. It’s a small reaction, but it packs a punch in terms of what it teaches us and how engaging it can be to watch!