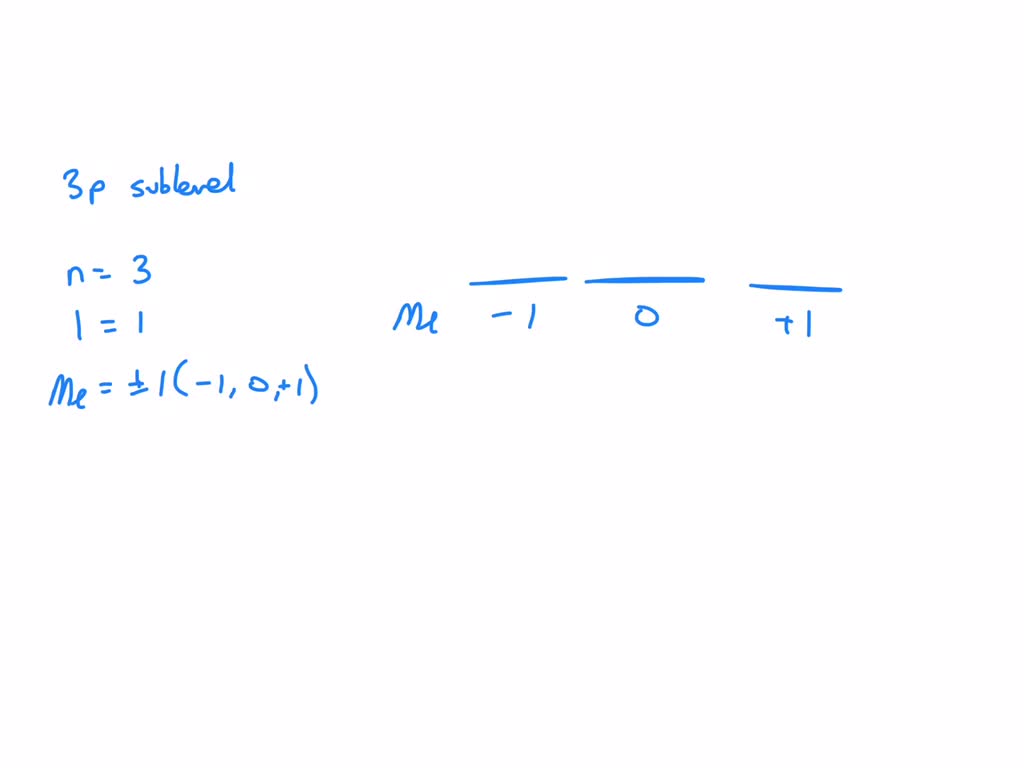
Hey there, coffee buddy! So, you've been dabbling in the wild and wonderful world of atoms, huh? Awesome! And now you've bumped into something called a "3p sublevel"? Don't sweat it, it's not as scary as it sounds. Think of it like… well, let's dive in, shall we?
You know how electrons are always buzzing around the nucleus, right? They don't just float around anywhere, though. Nope, they’re like picky little tenants, always looking for the best apartment. These apartments come in different sizes and shapes, and we call them "orbitals." Makes sense, right?
Now, these orbitals group together into "shells," and then, get this, they group again into what we call "sublevels." It’s like a Russian nesting doll, but way more… atomic. And the one we're chatting about today is the 3p sublevel. Fancy!
So, what's the deal with this "3p" thing?
Let's break it down. The "3" part? That's easy peasy. It tells us which energy shell we're talking about. Think of it as the floor in our apartment building. So, "3" means we're on the third floor. Higher floor, more energy, you get the picture. It's not the basement, that's for sure!
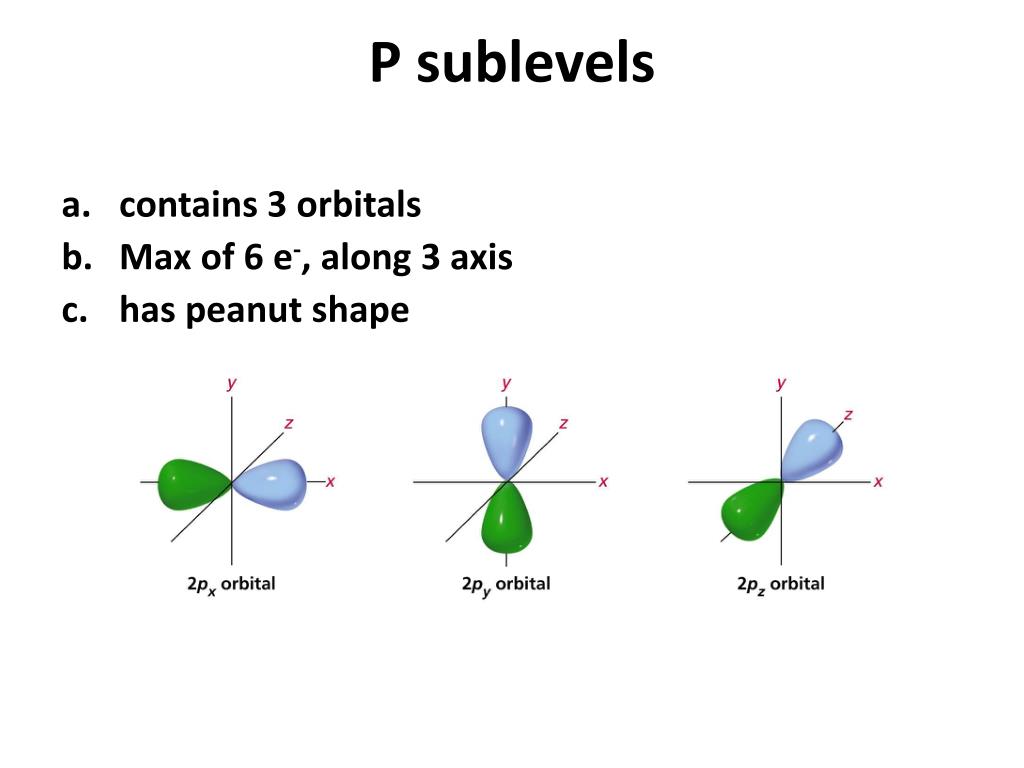
The "p" part? Ah, this is where it gets a little more interesting. The letter tells us the shape of the orbitals within that sublevel. And let me tell you, these shapes are not your average circles. Boring! The "p" orbitals? They're like little dumbbells. Or maybe tiny, elegant hourglasses. Seriously, the universe has a flair for the dramatic.
So, the 3p sublevel just means we're talking about the p-shaped orbitals that are on the third energy level. Boom! Not so mysterious anymore, right? It’s like saying "the comfy armchair on the third floor." See? Totally manageable.
What are the values we're looking for?
Okay, now to the nitty-gritty. When we talk about "values" in this atomic context, we're usually talking about quantum numbers. Don't let that big word intimidate you! It's just a way for scientists to precisely describe where an electron is and what it's doing. Like giving an electron its own personal address and its own little behavioral profile. Super detailed!

For the 3p sublevel, we’re specifically interested in two quantum numbers: the azimuthal quantum number (fancy name, I know, just call it 'l' for short, much friendlier!) and the magnetic quantum number (or 'm_l').
Let's tackle 'l' first. This guy tells us the shape of the orbital. Remember our dumbbell shapes for 'p'? Well, different shapes have different 'l' values. For an 's' orbital (which is just a plain old sphere, how quaint!), l = 0. For our beloved 'p' orbitals, l = 1. That's the key value for the shape! If you see a 'p' anything, you know l is 1. It's like a secret handshake.
Now, for 'm_l'. This is where things get a bit more spatially interesting. 'm_l' tells us about the orientation of that orbital in space. Think of our dumbbell shapes. Can they point along the x-axis? The y-axis? The z-axis? Yes, they can! And 'm_l' tells us which one.
For any given 'l' value, 'm_l' can take on a whole range of integer values. And the rule is, it goes from -l to +l, including zero. So, if l = 1 (which it is for our 3p sublevel!), then 'm_l' can be -1, 0, or +1. That's it! These are your three possible orientations for the 'p' orbitals.
So, for the 3p sublevel, we have:

- l = 1 (because it's a 'p' sublevel)
- m_l = -1, 0, +1 (the possible orientations for 'p' orbitals)
These are the core values that define the 3p sublevel. It’s like saying, "We're on the third floor, we've got dumbbell-shaped apartments, and they can point in three different directions." Simple, right? Well, as simple as quantum mechanics gets, anyway!
Why do we even care about these values?
You might be thinking, "Okay, I know these numbers. So what? Does it really matter?" Oh, my friend, it matters a ton! These little numbers are the building blocks for understanding how atoms behave. They dictate everything from why certain elements react the way they do to how light interacts with matter. It's like knowing the blueprint of a house – it tells you so much about how it's put together and how you can live in it.
For instance, the fact that there are three 'p' orbitals (because m_l goes from -1 to +1) is HUGE. It means that a 'p' sublevel can hold up to six electrons. Why six? Because each orbital can hold a maximum of two electrons, and we have three orbitals! Double that, and voilà! Six electrons can chill in the 3p sublevel. That's why elements like oxygen and fluorine, which have electrons filling up those 3p orbitals, have such interesting and active chemical personalities.
Think of it this way: if you only had one bedroom in your apartment (like an 's' sublevel), you could only fit two people comfortably. But with three "rooms" (our 'p' orbitals), you can invite more friends over! More electrons, more potential for chemical shenanigans!
The "n" number: Don't forget our main man!
Now, I know I focused on 'l' and 'm_l' for the sublevel itself, but we can't forget the first number, the principal quantum number, 'n'. We already said 'n=3' for our 3p sublevel. This 'n' number is super important because it also tells us the energy of those electrons. Higher 'n' means higher energy. So, electrons in the 3p sublevel have more energy than electrons in, say, the 2p sublevel, or even more than the 1s sublevel. It’s all about those energy levels!
The 'n' number is like the main address number. The 'l' number is like the street type (is it a boulevard? a lane?). And 'm_l' is like the house number on that street. And then, there's another quantum number, the spin quantum number ('m_s'), but that's a story for another coffee! It tells us if an electron is spinning "up" or "down," like a tiny little top. But for the 3p sublevel itself, we're mostly concerned with n=3, l=1, and m_l = -1, 0, +1.
Putting it all together: The magic formula
So, if someone asks you for the correct values for a 3p sublevel, you can confidently whip out this information:
- Principal Quantum Number (n): 3 (This is our "third floor"!)
- Azimuthal Quantum Number (l): 1 (This is our "p" shape, the dumbbell!)
- Magnetic Quantum Number (m_l): -1, 0, +1 (These are the three different ways our dumbbell can point in space!)
These are the fundamental properties that define what a 3p sublevel is. It’s not just a name; it's a set of characteristics that dictate its energy, its shape, and its spatial orientation. Pretty cool, right?
Think of it like this: if you're trying to find a specific type of book in a library, you need more than just the general section. You need the floor (the shell), the type of shelving (the sublevel shape), and maybe even where on that shelving it sits (the orientation). These quantum numbers are our library call numbers for electrons!
The visual aspect: Imagining those shapes
It’s helpful to actually visualize what these 'p' orbitals look like. They’re not just abstract numbers! You have three of them, and they are typically depicted as being aligned along the x, y, and z axes. So, you'd have a 'p_x' orbital, a 'p_y' orbital, and a 'p_z' orbital. Each one looks like two lobes, like a balloon that’s been pinched in the middle. And these are spread out in three dimensions!

The nucleus is right there in the middle, where the pinch is. And the electrons? They hang out in those lobes. It’s kind of mind-bending to think that this is where electrons actually are, or at least where they're most likely to be found. We can't pinpoint them exactly, but we can describe the space they occupy. It’s like a probability cloud, but with a very specific, dumbbell-like shape on the third floor.
And that's why the magnetic quantum number (m_l) is so important. It's what distinguishes these three different, mutually perpendicular orientations. Without m_l, we'd just have "a 'p' orbital on the third floor," but we wouldn't know which of the three orientations we were talking about. It’s the little details that make all the difference in the atomic world!
A quick recap to cement it in your brain
So, to recap our little atomic chat over coffee:
- The number in front (like the 3 in 3p) is the principal quantum number (n), telling us the energy level or shell.
- The letter (like the p in 3p) tells us the shape of the orbitals within that sublevel. 'p' means a dumbbell shape, and its azimuthal quantum number (l) is always 1.
- The possible values for the magnetic quantum number (m_l) depend on 'l'. For l=1, m_l can be -1, 0, or +1, indicating the different spatial orientations of these dumbbell orbitals.
And that's pretty much the whole story for the 3p sublevel's defining values! You've got this. Just remember the floor, the shape, and how it can point. It’s like giving directions to a very, very tiny, very energetic resident.
Next time you see "3p," you won't just see letters and numbers; you'll see a whole picture of electron apartments on the third floor, with their unique shapes and orientations. Go you! High five! Now, who wants more coffee?
.jpg)


+in+space..jpg)