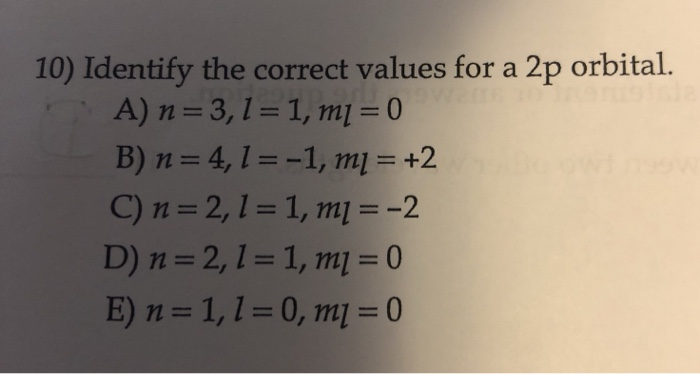Ever wondered what makes atoms tick? It's like a tiny, incredibly organized universe, and understanding it can be surprisingly fun! Today, we're going to peek into the world of electron shells and specifically, what makes a 2p sublevel special. Think of it as discovering the specific flavor of a very important ingredient in the recipe of all matter. It’s not just for super-smart scientists; it’s a little secret that unlocks a deeper appreciation for the world around us.
So, why should you care about a 2p sublevel? For the beginner learner, it’s a fantastic stepping stone into the basics of chemistry and physics. It introduces the idea that electrons aren't just floating around randomly, but have specific addresses within an atom. For families looking for a way to make science engaging, discussing how atoms are built can be a fun bedtime story or a kitchen table experiment. Imagine explaining that the carbon in your pencil lead and the oxygen you breathe are both made from these fundamental building blocks. For hobbyists, whether you're into collecting minerals, understanding electronics, or even appreciating the science behind cooking, knowing about electron configurations can add a whole new layer of understanding to your passions. It’s like learning the secret handshake of the universe!
What exactly are the correct values for a 2p sublevel? When we talk about electron sublevels, we're referring to how electrons are organized within an energy shell. The '2' in '2p' tells us it's in the second energy shell from the nucleus. The 'p' tells us about the shape of the region where these electrons are most likely to be found – think of it as a dumbbell shape, rather than a simple sphere. For a 'p' sublevel, there are always three orbitals. Each orbital can hold a maximum of two electrons. So, a 2p sublevel can hold a total of six electrons (3 orbitals x 2 electrons/orbital).
Let's break this down with some simple examples. In the second energy shell, you'll find both the 2s sublevel (which is spherical and holds up to 2 electrons) and the 2p sublevel. Elements like Nitrogen (N), with 7 electrons, have the electron configuration 1s² 2s² 2p³. See that '2p³'? It means three electrons are occupying the 2p sublevel. Oxygen (O), with 8 electrons, goes a step further: 1s² 2s² 2p⁴. That fourth electron in the 2p sublevel is important for how oxygen forms bonds!

Getting started with understanding sublevels is easier than you think. You don't need a fancy lab! You can start by looking at an electron configuration chart online. Many websites offer interactive versions. Try looking up the electron configurations for the first 20 elements of the periodic table. See if you can identify where the 2p sublevel is filled. You can even get creative: draw simple diagrams of 's' (circle) and 'p' (dumbbell) orbitals and show how electrons fill them. It’s a visual way to grasp the concept.
Exploring the world of electron sublevels like the 2p might seem like diving into complex science, but it’s really about understanding the fundamental organization of everything. It’s a fascinating puzzle that helps us appreciate the intricate beauty of atoms. So, next time you look at anything, remember the tiny, organized dance of electrons within its atoms – it’s a truly rewarding insight!