Imagine you've baked a batch of your grandma's famous chocolate chip cookies. They're legendary, right? Everyone loves them. Now, what if I told you that sometimes, in the world of tiny, invisible molecules, things can be a bit like having two versions of those cookies? Not identical twins, but more like mirror images. They look the same in the mirror, but if you try to put your left glove on your right hand, it just doesn't fit. These molecular twins are called enantiomers, and understanding how many of each you have is surprisingly important, especially in medicine!
So, how do we figure out if we have a perfect batch of one "cookie" or a mix of both "mirror image" cookies? This is where our trusty tool, NMR spectroscopy, comes to the rescue. Now, NMR sounds fancy, like something you'd need a lab coat and a secret handshake to understand. But at its heart, it's like giving your molecules a very gentle, very polite poke and listening to how they respond. Think of it like this: you have two identical balls, but one is a left-handed screw and the other is a right-handed screw. If you spin them, they'll behave slightly differently. NMR is our way of "spinning" molecules and hearing their unique "sounds."
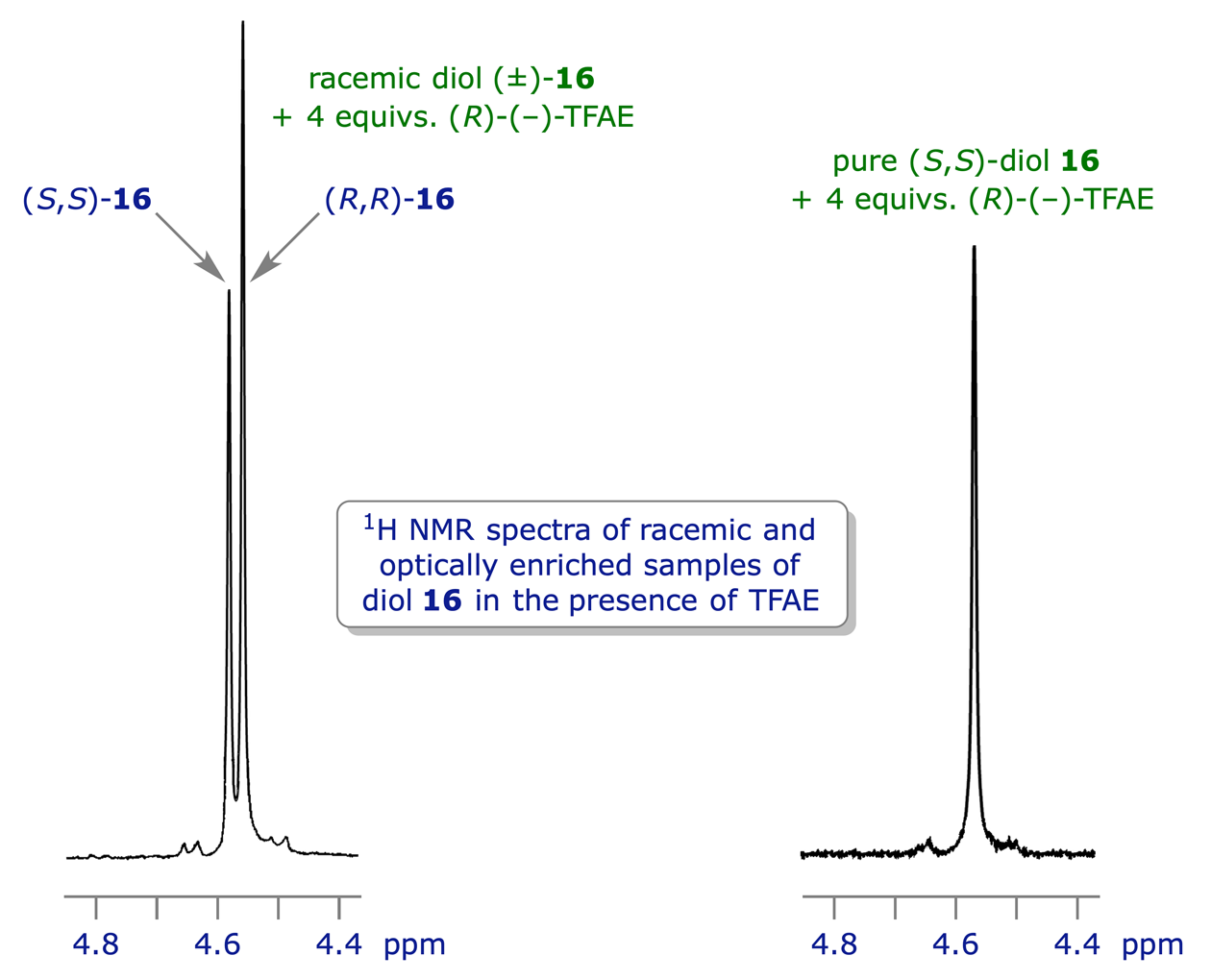
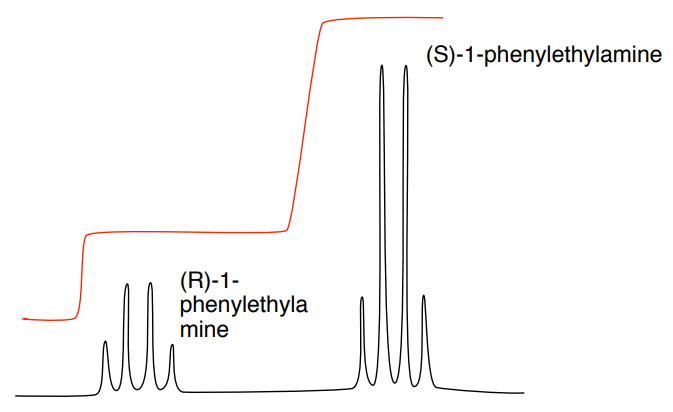
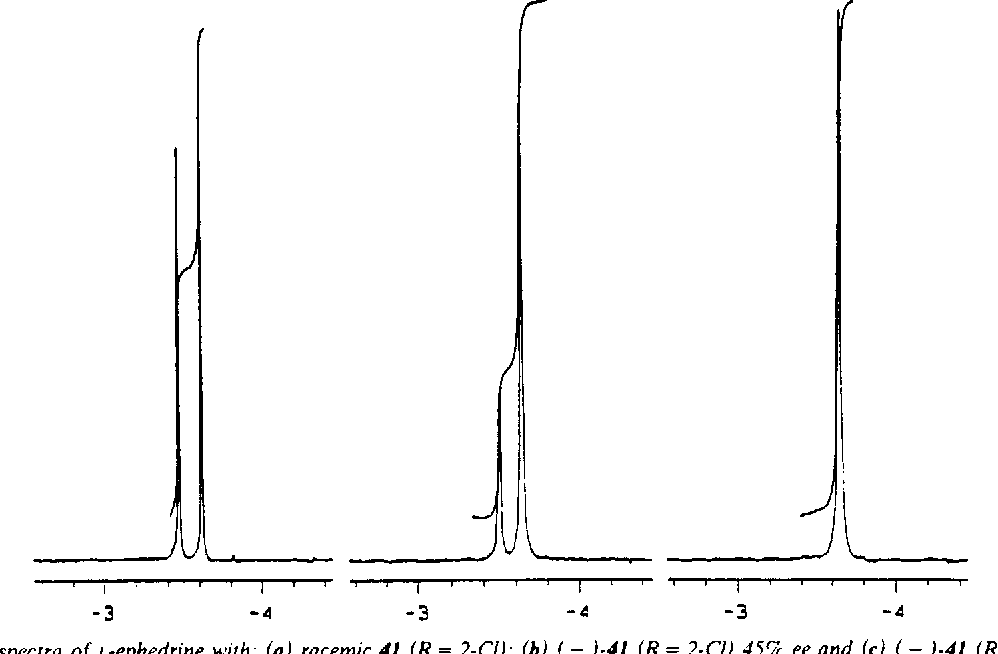
Here's where the fun really begins. Most of the time, NMR will show us a bunch of signals, like different instruments in an orchestra playing their notes. When we have a perfect batch of just one enantiomer, it's like having a symphony with only violins. Beautiful, clean, and all playing the same tune. But if we have a mix, it's like adding in some cellos – they're still musical, but their notes are just a little bit different, enough for a keen ear to notice. These subtle differences in their "notes" are what tell us we have a mixture!
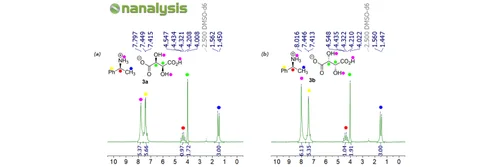
Now, calculating the enantiomeric excess, or ee, is basically figuring out how much of your "preferred cookie" you have compared to its mirror image. It's like asking, "Out of all the cookies I have, what percentage are the real grandma's recipe, and what percentage are the slightly-off mirror images?" We do this by looking at the heights of those different "notes" we hear from the NMR machine. The louder the note (the taller the peak on the NMR chart), the more of that particular enantiomer is present.
Let's say you have two distinct peaks in your NMR spectrum, and one peak is a little bit taller than the other. We'll call the height of the taller peak "Big Peak" and the height of the smaller peak "Small Peak." The calculation is surprisingly straightforward, almost like dividing up a pizza! You take the height of the "Big Peak," subtract the height of the "Small Peak," and then divide that difference by the total height of both peaks (which is just "Big Peak" + "Small Peak"). Finally, you multiply by 100 to get a percentage. Voila! That percentage is your enantiomeric excess.
It might sound complex, but think about it in everyday terms. If you have a bag with 90 red marbles and 10 blue marbles, and you want to know the "excess" of red marbles, you'd say you have 80 more red than blue (90-10). Then you'd figure out what percentage of the whole bag that 80 represents. It's the same idea, just with molecules and their special NMR "songs."
The heartwarming part? In drug development, for example, one enantiomer of a molecule might be a life-saving medicine, while its mirror image could be completely inactive, or even worse, cause harmful side effects. Think of the thalidomide tragedy – a devastating example where one enantiomer helped with morning sickness, but its mirror image caused severe birth defects. So, being able to accurately measure this enantiomeric excess is like a quality control check, ensuring that patients get the safe and effective version of a drug. It's a silent guardian, making sure the "cookies" we're giving out are exactly what they're supposed to be.
And the surprising twist? This little calculation, this peek into the molecular mirror, relies on a fundamental property of these molecules – their interaction with a magnetic field. It’s like they have a tiny, internal compass that spins in a slightly different direction depending on whether they’re the "left hand" or the "right hand" version. NMR is essentially eavesdropping on these compasses, and by comparing how loud their "directions" are, we can deduce their proportions. It’s a testament to how even the tiniest, seemingly insignificant differences at the molecular level can have enormous consequences.
So next time you hear about enantiomeric excess or NMR, don't picture intimidating equations. Picture a baker carefully checking their cookie batches, or a scientist listening to the unique "songs" of molecules, all to ensure we get the best, most effective, and safest "treats" possible. It's a little bit of chemistry magic, powered by everyday logic and a whole lot of careful observation.
This ability to distinguish between these molecular twins is not just a laboratory trick; it's a crucial step in creating medicines that work, flavors that taste just right, and materials with specific properties. It’s about precision, about understanding the subtle nuances that make one molecule a superhero and its mirror image a mere bystander, or worse. And all thanks to a clever way of listening to what molecules have to say when they're tickled by a magnet!



.jpg)
