Okay, let's talk about atoms. Not in that super-boring, textbook way, but in a fun, kitchen-table sort of way. We're diving into the weird and wonderful world of electron shells. Think of atoms like tiny houses, and the electrons are the residents. The outermost residents are the ones that really get to mingle and make things happen. These are our valence electrons. They're the life of the party, the ones that decide if our atom is going to be a social butterfly or a grumpy hermit.
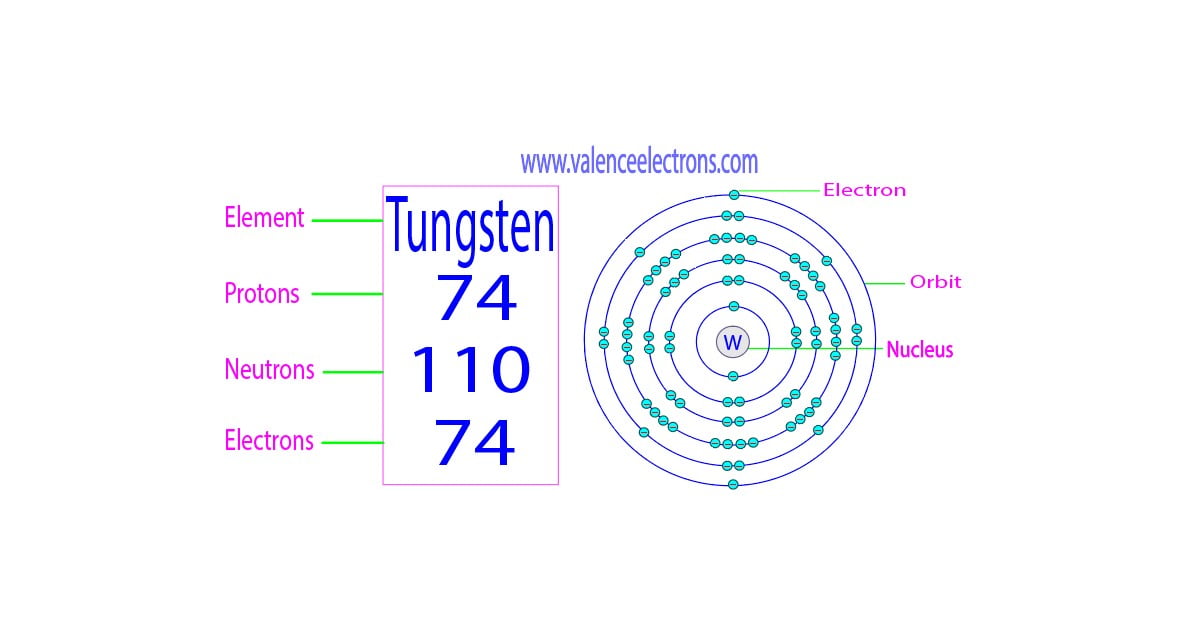
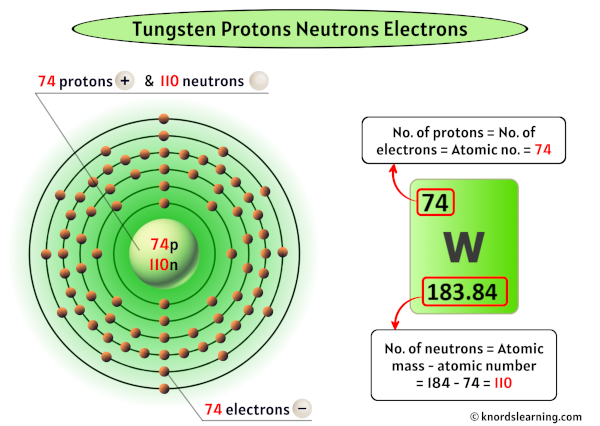
Now, there's this element, Tungsten. You might know it from your lightbulbs. Yes, that little glowing wire inside is made of tungsten! Pretty cool, right? It's a super strong metal, and it can handle a lot of heat without melting. It’s a bit of a superhero in the element world.
So, the big question, the one that keeps some scientists up at night (probably not really, but let’s pretend!), is: How many valence electrons does tungsten have? Get ready for a little bit of a curveball, because the answer isn't as straightforward as you might expect. It’s a bit like trying to count the jellybeans in a jar where some have rolled under the couch. You think you know, but then you find one you missed.
My personal, slightly rebellious, and very unofficial opinion? I think tungsten is playing a little bit of a trick on us. It looks like it should have a certain number, based on where it sits on that giant periodic table chart. You know, that colorful poster that’s probably hanging in a science classroom somewhere, looking all important.
If you were to just glance at its position, you might say, "Aha! It's in this group, so it must have X valence electrons!" And that's a perfectly reasonable guess. It's like seeing a friend wearing a red shirt and assuming they’re going to a picnic because picnics often involve red. But maybe they're just wearing the red shirt because it’s clean.
The thing about tungsten is that it’s a bit of a rebel. It doesn't always play by the simplest rules. It’s a transition metal, and those guys are known for being a little… complicated. They’re like the teenagers of the periodic table, capable of a lot of interesting stuff but also sometimes surprising you with their choices.
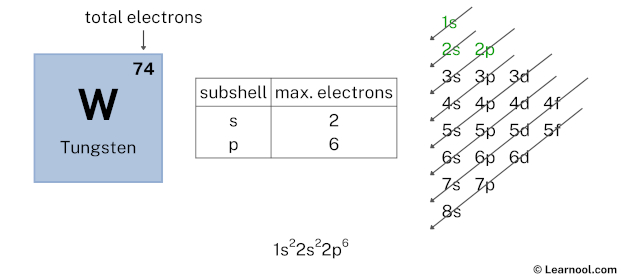
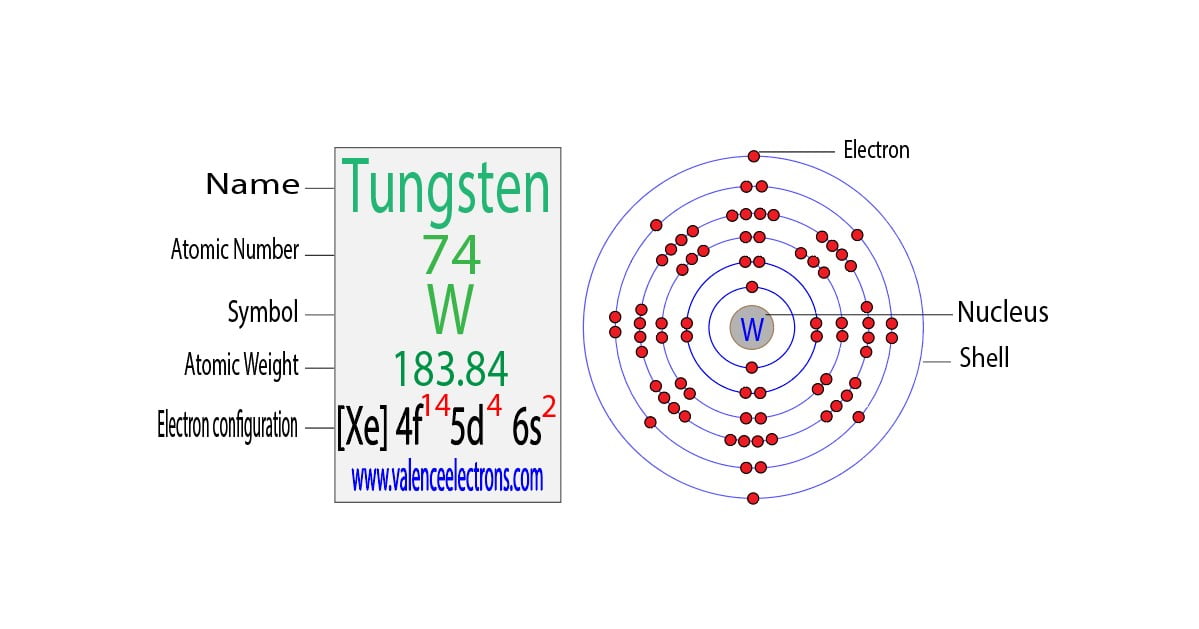
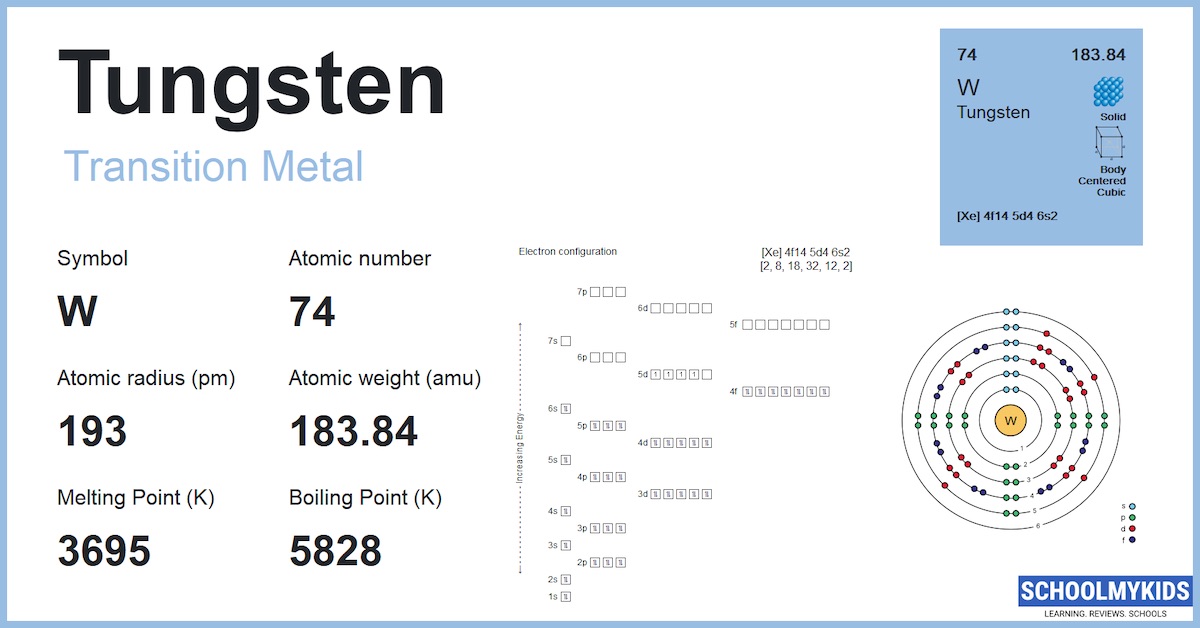
So, let's talk numbers, but let’s do it with a wink. Officially, the electron configuration of tungsten is a bit of a mouthful. It’s something like [Xe] 4f¹⁴ 5d⁴ 6s². Now, don't let that scare you. Think of "[Xe]" as the inner circle of friends who don't really come out to play. The real action is in the outer shells, the 4f, 5d, and 6s.
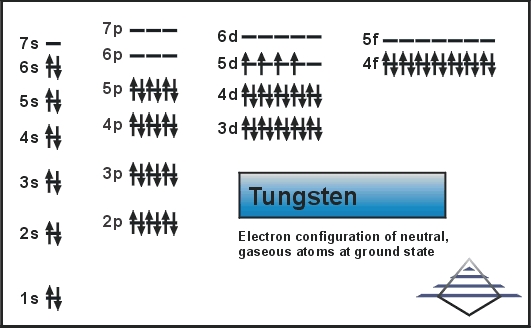
For valence electrons, we're typically looking at the electrons in the outermost energy shells. In the case of tungsten, the 6s shell has 2 electrons. That seems pretty straightforward, right? That’s two valence electrons right there. Easy peasy.
But here’s where it gets interesting, and why I think tungsten is a little mischievous. The 5d shell, which is also pretty close to the outside, has 4 electrons. Are those considered valence electrons? Sometimes, yes! Depending on what kind of chemical party tungsten is attending, those 5d electrons can get involved. They can be shared, swapped, or generally thrown into the mix. So, suddenly, instead of just 2, you might have 2 + 4 = 6 valence electrons!

Now, some strict chemists might say, "No, no, no! Valence electrons are only the outermost ones!" And that’s fine for them. That’s their opinion. But I’m here to tell you that tungsten is more complex. It’s not a one-trick pony. It’s got hidden talents.
So, if you ask me, "How many valence electrons does tungsten have?" my answer is: it depends on the day, the mood, and what it’s trying to achieve. It has at least 2, but it can also easily act like it has 6. It's like a chameleon, blending in and adapting.

It’s the element equivalent of saying, "Well, I could just have this one cookie, but I might have the whole sleeve if I feel like it."
This flexibility is why tungsten is so useful. It can form all sorts of different chemical bonds. It’s not just stuck being one way. It’s got options!
So, next time you flick on a light switch and see that beautiful glow, give a little nod to tungsten. And maybe, just maybe, wink back at it, because you know its little secret about those valence electrons. It’s not just one number; it’s a whole spectrum of possibilities. And I, for one, find that delightfully complex and a little bit hilarious. It’s the element that keeps you guessing, and I’m perfectly okay with that. It’s an unpopular opinion, I know, but sometimes the most interesting things are the ones that defy simple answers. Tungsten, you magnificent, electron-bending mystery, we salute you!