Hey there, sunshine! Ever find yourself staring out the window, sipping your artisanal coffee, and a random science question pops into your head? You know, the kind that’s a little bit more exciting than what’s for dinner? Today, we’re diving into the wonderfully weird world of atoms, specifically, the curious case of selenium. Now, before you picture lab coats and bubbling beakers, let’s keep this as chill as a perfectly chilled cucumber salad. We’re talking about its valence electrons, and trust me, it’s more fascinating than a documentary about sloths.
So, what exactly are these elusive things called valence electrons? Think of them as the atom’s outermost crew, the party animals on the edge of the atomic club. They’re the ones who get to mingle, bond, and generally cause all the chemical ruckus. Without them, atoms would be pretty lonely and wouldn’t form the dazzling molecules that make up, well, everything. From your favorite comfy sweater to the very air you breathe, it’s all thanks to these energetic little guys.
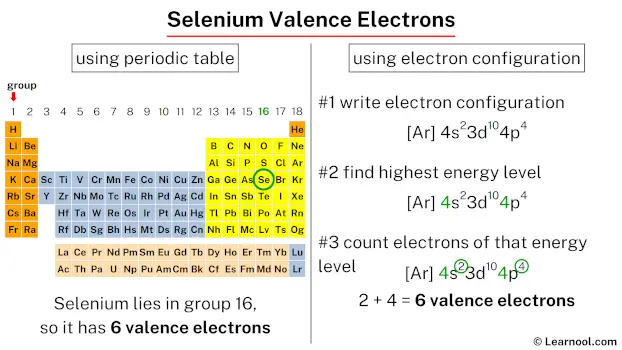
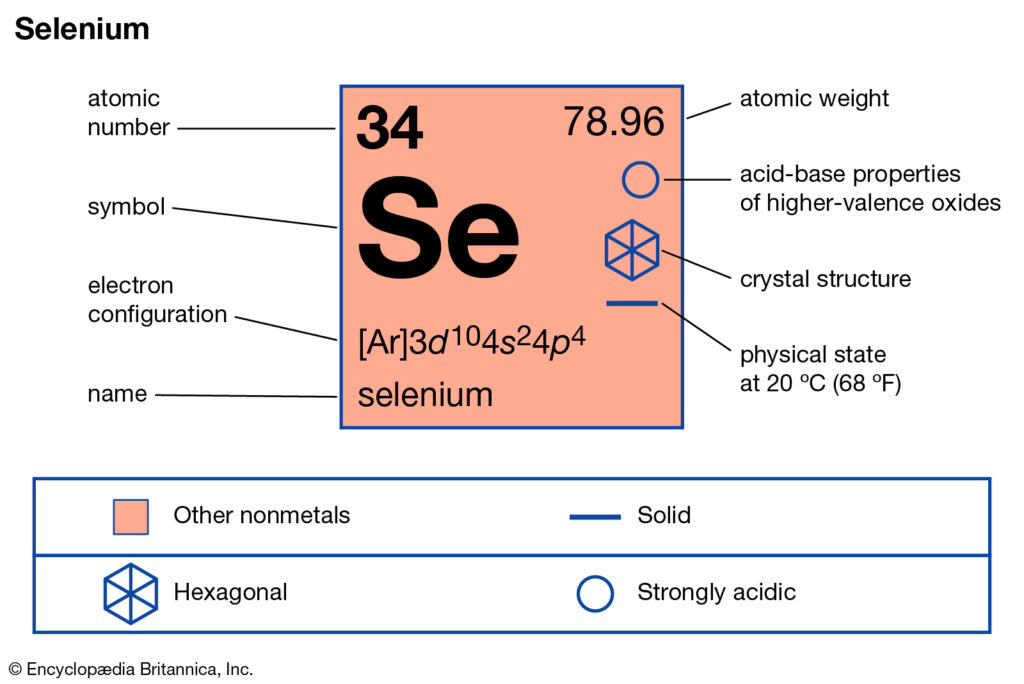
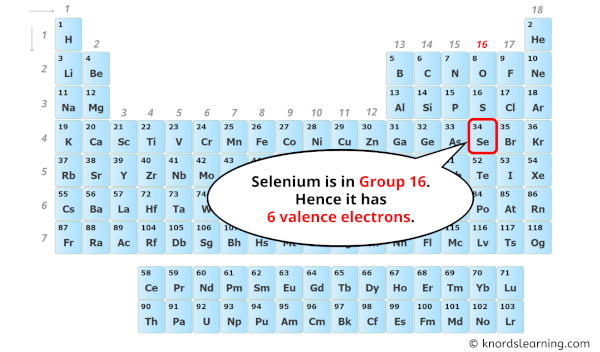
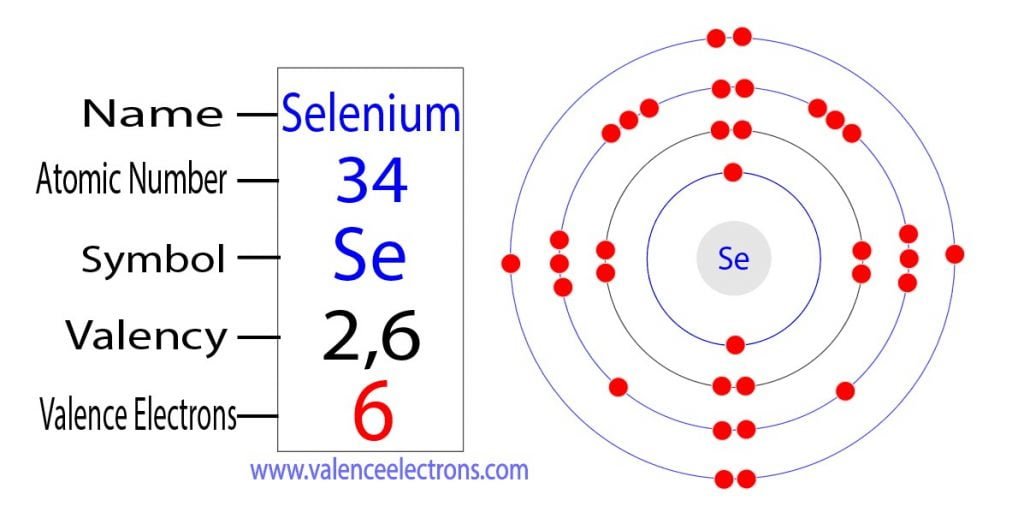
Now, let’s bring on the star of our show: Selenium. Ever heard of it? If you’re a fan of superfoods, you might have! It’s that trace mineral often touted for its antioxidant powers, showing up in Brazil nuts (seriously, just one or two can pack a punch!) and even some seafood. It’s element number 34 on the periodic table, which means it’s got a bit of a story to tell. And like any good story, it all starts with its electron configuration. Don't worry, we’re not going to get bogged down in quantum mechanics. Think of it more like assigning roles in a superhero team.
Unpacking the Electron Shells
Atoms have layers, or shells, where their electrons hang out. The inner shells are like the VIP section, pretty stable and not easily disturbed. The valence electrons, however, are chilling in the outermost shell, the place where the action really happens. They’re the ones that decide how an atom behaves, whether it’s going to be a social butterfly, happily sharing electrons, or a bit more reserved, preferring to keep to itself.
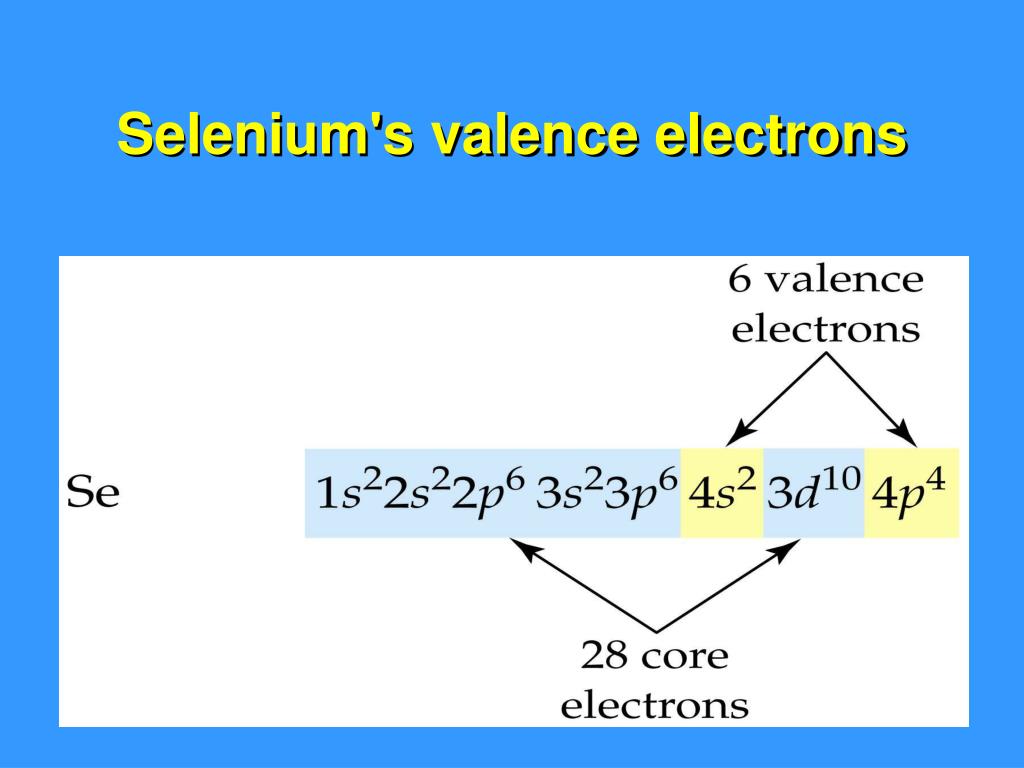
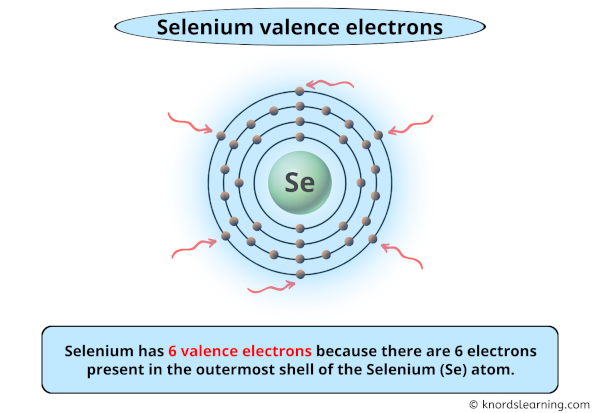
For selenium, which sits pretty in Group 16 of the periodic table (alongside oxygen and sulfur, its chemical cousins), its electron configuration is a bit of a giveaway. When you look at its full electron setup, you’ll see it has a certain number of electrons in its outer shell. And that number, my friends, is a beautiful, clean six. Yes, that’s right. Selenium boasts a magnificent six valence electrons.
Think of it like this: if the periodic table were a bustling city, selenium would live in the "Six-Shooter" district. Every element in that district is known for having six characters in its outermost electron posse. This little detail is super important because it dictates how selenium likes to play the chemical game. It's like knowing your friend always brings the best snacks to a party – you know what to expect!
Why Six Valence Electrons Matters (More Than You Think!)
So, why is this number six so significant? Well, atoms generally strive for a full outer shell, a state of perfect electron harmony, often referred to as the octet rule. It’s the atomic equivalent of finding your favorite playlist – everything just feels right. For most atoms, this means having eight electrons in their outer shell. This is where selenium gets interesting.
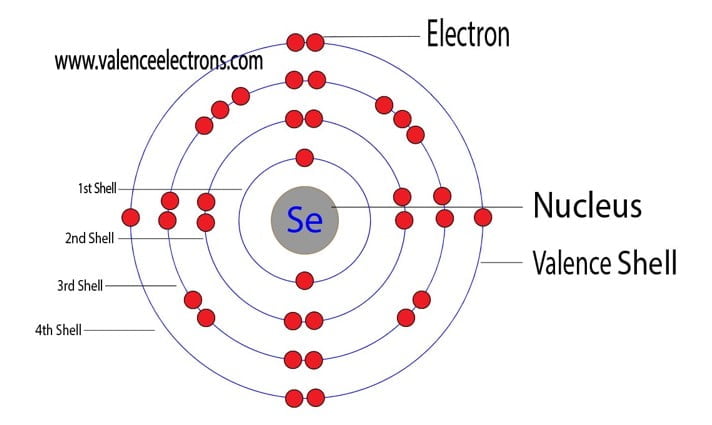
With its six valence electrons, selenium is almost there. It’s like being at the final level of a video game and you’re just one power-up away from victory. This means selenium is quite keen on finding two more electrons to complete its octet. It’s not as desperate as, say, an element with only one valence electron, but it’s definitely not kicking back and relaxing either. It’s in a state of chemical readiness!
This tendency to want two more electrons makes selenium a versatile player in chemical reactions. It can readily form bonds with other atoms, either by accepting electrons or by sharing them in a way that helps everyone achieve a more stable configuration. This is why you find selenium in so many important compounds, playing a crucial role in biological processes and industrial applications alike.
Selenium in the Wild (and Your Diet!)
When we talk about selenium in the context of health, these six valence electrons are working their magic. As an antioxidant, selenium helps protect your cells from damage caused by unstable molecules called free radicals. It’s like having a tiny, incredibly efficient bodyguard for your cells, thanks to those readily available electrons ready to neutralize the bad guys.
Think about the role of antioxidants in your favorite smoothie recipes. Berries, leafy greens – they’re all packed with compounds that help combat oxidative stress. Selenium is in a similar league, contributing to your body’s defense system. It’s found in enzymes like glutathione peroxidase, which are vital for detoxification and protecting your DNA.
%2C+sulfur+(S)+and+selenium+(Se)+have+6+valence+electrons!.jpg)
This is where those fun facts come in! Did you know that the amount of selenium in plants varies greatly depending on the soil they grow in? If the soil is rich in selenium, the plants will be too. This is why consuming a variety of foods and considering your geographic location can be important for ensuring adequate intake. It’s like a little treasure hunt for nutrients!
Culturally, the appreciation for selenium-rich foods often pops up in discussions about healthy eating. Brazil nuts, as mentioned, are selenium powerhouses. Just a couple of these can satisfy your daily requirement! It's a delicious reminder that nature often provides the simplest solutions. Imagine enjoying a delicious snack that's also a tiny chemical marvel.
Beyond Biology: Selenium in Technology
But wait, there's more! The chemical properties of selenium, driven by its six valence electrons, also make it useful in a surprising array of technologies. Its ability to conduct electricity is influenced by light, a property known as photoconductivity. This has led to its use in early photocopiers and light-sensitive devices.
Think about the humble photocopier – a marvel of mid-century ingenuity. Back in the day, selenium drums were at the heart of its operation. When light hit the drum, it changed its electrical conductivity, allowing toner to stick to the right places, thus creating copies. It’s a testament to how fundamental atomic structure can lead to widespread practical applications.
It's also found its way into glassmaking, giving glass a pinkish tint and helping to decolorize glass that has a greenish hue caused by iron impurities. So, the next time you admire a beautifully clear wine glass, you might be indirectly appreciating the work of selenium and its electron shenanigans!
The Periodic Table: A Cosmic Family Reunion
Let’s zoom out for a second and appreciate the periodic table itself. It’s not just a chart; it’s a beautiful representation of how elements are related. Elements in the same group, like selenium and its neighbors oxygen and sulfur, share similar chemical characteristics because they have the same number of valence electrons. It’s like a cosmic family reunion where everyone has a similar personality trait.
Oxygen, with its six valence electrons, is notoriously reactive, forming water and being essential for respiration. Sulfur, also with six, is a key component in proteins and has that distinct smell in volcanic gases. Selenium fits right in this energetic family, contributing its unique brand of chemical charm.
Understanding these relationships helps us predict how elements will behave and interact. It’s like knowing that if you like pizza, you’ll probably enjoy pasta – there’s a common thread! This understanding is the bedrock of chemistry, from designing new medicines to creating advanced materials.
Fun Fact Fiesta!
Here’s a little something to chew on: while essential in small amounts, selenium can be toxic in larger doses. It’s a classic case of “too much of a good thing.” This delicate balance is common in nature and highlights the importance of understanding the precise roles and quantities of different elements. It’s a bit like moderation in all things, even in our atomic interactions!
Another fun tidbit: the word "selenium" comes from the Greek word "selene," meaning "moon." This is because selenium was discovered shortly after tellurium, which was named after "tellus," the Latin word for Earth. The discoverer, Jöns Jacob Berzelius, saw them as a pair, a celestial duo. How poetic is that? Atoms with lunar connections!
And finally, did you know that selenium is used in some anti-dandruff shampoos? Those six valence electrons are apparently quite the problem-solvers, even for itchy scalps! It’s a reminder that chemistry is all around us, in the most unexpected places.
Bringing It Back to Our Daily Grind
So, we’ve journeyed from the cosmic dance of electrons to the practical applications of a humble element. At its core, the fact that selenium has six valence electrons is a simple number, but it’s a number that unlocks a universe of chemical possibilities. It influences its role in our bodies, its use in technology, and its place in the grand tapestry of the periodic table.
Think about it the next time you’re enjoying a Brazil nut, scrolling through your phone, or even just breathing in the air. That invisible world of atoms and electrons is constantly at work, shaping our reality. And the seemingly small details, like the number of valence electrons an atom possesses, are the fundamental building blocks of it all.
It’s a reminder that even in the most complex systems, whether it’s our own biology or the technology we rely on, the underlying principles are often elegantly simple. And sometimes, the most profound understanding comes from appreciating those fundamental truths, like the fact that selenium, our moon-named friend, is perfectly content with its six energetic valence electrons, ready to make its mark on the world, one chemical bond at a time.