Hey there, welcome! Ever find yourself wondering about the little building blocks that make up everything around us? You know, the stuff that fuels our cars, lights our homes, and even makes up the air we breathe (well, some of it!). Today, we're going to take a peek at something super simple, yet surprisingly fascinating: propane. And more specifically, we're going to tackle a question that might sound a tad technical, but trust me, it's as easy-going as a Sunday morning stroll. We're asking: How many structural isomers does propane have?
Now, before you picture complex chemistry textbooks or late-night study sessions (no thanks!), let's break down what "structural isomers" even means in a way that's more like chatting over coffee than a science lecture. Think of it like this: imagine you have a set of LEGO bricks. You can build a whole bunch of different things with the same set of bricks, right? You could build a car, a house, or even a wobbly-looking spaceship. These are all made from the same bricks, but they're put together in different ways, giving them different shapes and functions. That, my friends, is the essence of structural isomers!
In chemistry, these "LEGO bricks" are atoms – like carbon and hydrogen. When we talk about structural isomers, we're talking about molecules that have the exact same number of atoms of each type, but those atoms are connected in a different arrangement. It's like having the same ingredients for a recipe, but instead of making a cake, you whip up some cookies. Same stuff, different outcome!
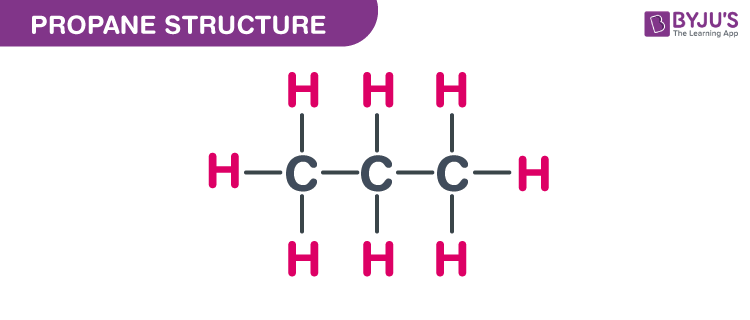
So, let's get down to propane. What's propane, you ask? Well, if you've ever used a grill for a summer barbecue, or seen those big tanks on the back of food trucks, that's probably propane! It's a gas that's super handy for cooking and heating. Chemically speaking, propane is a pretty straightforward molecule. It has three carbon atoms and eight hydrogen atoms. That's our basic recipe: 3 carbons and 8 hydrogens. Simple enough, right?
Now, here's where the fun begins. With those 3 carbon atoms and 8 hydrogen atoms, how many different ways can we actually connect them? Could we arrange them in a straight line? Could we bend them around? Could we make them do a little dance with each other? This is what we're trying to figure out with our "how many structural isomers" question.
Imagine you have three little friends, and you're trying to arrange them in a line for a photo. You can put Friend A first, then B, then C. Or you could put B first, then A, then C. And so on. There are different ways to arrange them. But with atoms, it's a bit more about who's holding hands with whom. Carbon atoms, in particular, are like social butterflies. They love to bond with other atoms, especially hydrogen.
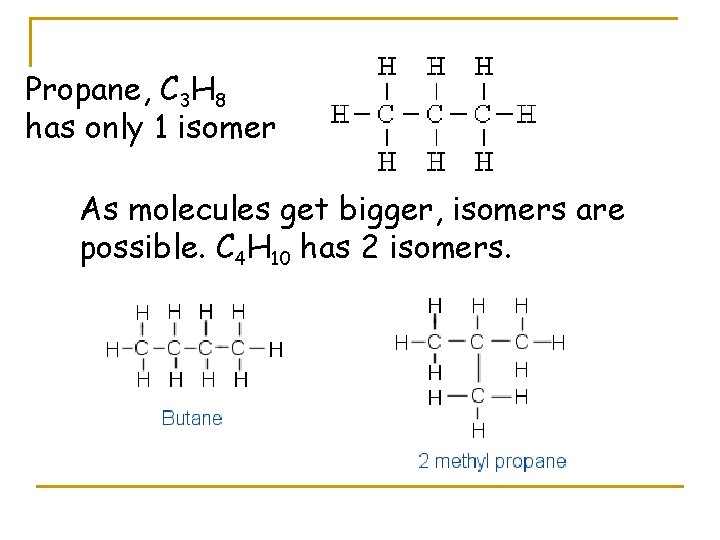
So, let's try to build our propane molecule. We have our three carbon atoms. The most straightforward way to connect them is in a nice, long, straight chain. Picture a little train with three carriages. We've got carbon-carbon-carbon. Now, we need to add our eight hydrogen atoms to make everyone happy. Each carbon atom likes to have a total of four "connections" (or bonds). In our straight chain, the carbon at either end will be connected to one other carbon and three hydrogens. The carbon in the middle will be connected to two other carbons and two hydrogens. Let's count them up: 3 hydrogens + 3 hydrogens + 2 hydrogens = 8 hydrogens. Perfect! This is one way to build propane, and it's the most common one you'll encounter. We often call this n-propane, where "n" stands for "normal," meaning it's the straight-up, no-nonsense version.
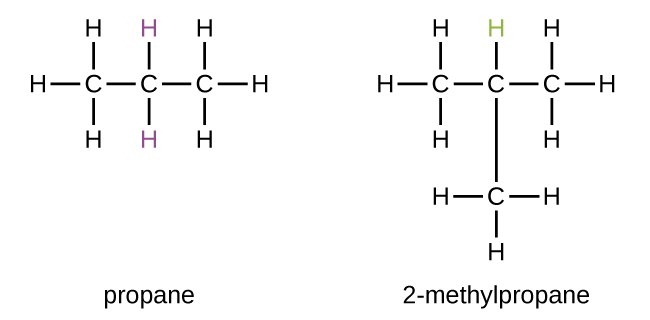
This straight chain arrangement is like a perfectly aligned row of dominoes, all set to fall in a predictable way. It's stable, it's efficient, and it's the way most propane you use is put together. It's the reliable old friend of the molecule world.
Now, the big question: can we rearrange those same 3 carbon atoms and 8 hydrogen atoms to make something different? Something with the same ingredients but a unique structure? Let's try to get creative. What if we don't make a straight line? What if we try to make a little cluster?
We have three carbon atoms. If we try to make a branched structure, we need one carbon atom to be the "center" and the other two carbons to be "branches" coming off it. So, imagine one carbon in the middle, and then two other carbons attached to that middle one. Like a tiny little "Y" shape, or a mini tripod, but with carbons. The middle carbon would be connected to two other carbons. That's two of its four connections used up. Now, each of those two "branch" carbons is only connected to that middle carbon. They each need two more connections to reach their quota of four. So, we'd attach two hydrogens to each of those branch carbons. That uses up 2 hydrogens + 2 hydrogens = 4 hydrogens. We've also used up our three carbon atoms (the middle one and the two branches).
But wait a minute! We've only accounted for 4 hydrogen atoms. We need 8! Where can the other 4 hydrogens go? Remember that middle carbon? It's only made two connections so far (to the two branch carbons). It has two more connections available. So, we can add two hydrogens to the middle carbon. Now it has 2 connections to carbons + 2 connections to hydrogens = 4 connections. Great! So far, our middle carbon has 2 hydrogens, and each branch carbon has 2 hydrogens. That’s 2 + 2 + 2 = 6 hydrogens. Still not 8!
Let's rethink. We have 3 carbons. We want to branch. The only way to branch with 3 carbons is if one carbon is connected to the other two. Think of it like a short stick with another short stick stuck in the middle. If we try to have one carbon connected to two other carbons, that middle carbon already has two bonds. If we then try to attach the third carbon to one of those first two, we're getting into a much bigger molecule or a ring, which we can't do with just three carbons. The only way to branch with three carbons is to have one carbon in the middle, and the other two attached to it. But we found that only uses 6 hydrogens.
Let's reconsider our "LEGO bricks." We have 3 carbon "bricks" and 8 hydrogen "bricks." The straight chain: C-C-C. The end carbons get 3 H's each. The middle gets 2 H's. 3+2+3 = 8. This works beautifully. This is our n-propane. It’s like a neat row of three houses on a street.
Now, for a branched structure with 3 carbons. This means one carbon must be attached to more than one other carbon. With only three carbons, the only way one carbon can be attached to more than one other carbon is if it's attached to two other carbons. This is like a central hub with two spokes. So, C connected to C, and that central C also connected to another C. This forms a chain of three carbons, but with the middle one acting as a connection point. This IS the straight chain! There's no other way to connect 3 carbons where one carbon is attached to two others without it becoming part of a longer chain.
Let's try to visualize the possibilities again. We have three carbon atoms. Possibility 1: Connect them in a line. C-C-C. We've seen this works perfectly for propane, giving us n-propane. This is our straight-chain isomer. It's like three friends standing shoulder-to-shoulder.

Possibility 2: Can we arrange them differently? What if we tried to make a triangle? A ring? We only have 3 carbons, so a ring would be C-C-C with the ends connected. That would be cyclopropane, which is a different molecule altogether (it has a different chemical formula). So, that's not an isomer of propane. For isomers, we need the exact same number of atoms.
What about branching? To branch, one carbon has to be connected to more than one other carbon. With only three carbons, if one carbon is connected to two others, it naturally forms a straight chain. Imagine trying to make a tiny, cramped cluster. You have your three carbons. If you try to attach two carbons to a central one, you've used up all your carbons, and that middle carbon only has two bonds to other carbons. It needs two more bonds to reach its total of four. So, you'd add two hydrogens to the middle one. The two "outer" carbons each only have one bond to the central carbon, so they each need three more bonds. That would be 3 + 1 + 3 = 7 hydrogens. That's not propane! And we still only have three carbons.
This is where the "aha!" moment happens. It turns out, with just three carbon atoms, there is only one way to arrange them and attach the correct number of hydrogen atoms to satisfy their bonding needs. That one way is the straight chain.
So, to answer our question directly: How many structural isomers does propane have? The answer is a beautiful, simple, and resounding one.

That one structural isomer is n-propane. There are no other arrangements of 3 carbon atoms and 8 hydrogen atoms that create a different molecule with the same formula (C3H8). It’s like having a recipe for a simple chocolate chip cookie, and no matter how you try to mix those specific ingredients (flour, sugar, butter, chocolate chips – with the right proportions), you can only end up with a cookie. You can't magically turn it into a pizza or a salad with the same set of core ingredients!
Why should we care about this seemingly small detail? Well, it's a fantastic introduction to the vast world of organic chemistry! It shows us that even with the simplest molecules, there can be rules governing how they're built. Understanding isomers is super important because molecules that have the same chemical formula can behave very differently. Think of it like identical twins. They look the same, have the same genes (same formula), but one might be a brilliant artist and the other a math whiz (different properties and reactions!).
For bigger molecules, the number of isomers can explode! For example, molecules with just 6 carbon atoms can have hundreds of different structural isomers! This is why there are so many different plastics, medicines, fuels, and flavors in the world. Each unique arrangement of atoms leads to a unique substance with its own characteristics. So, while propane only has one isomer, this concept is the gateway to understanding the incredible diversity of life and materials around us.
It's a gentle reminder that even the most fundamental things have their own elegant order. Propane, in its simplicity, teaches us a big lesson about how atoms connect and the uniqueness that arises from arrangement. So next time you fire up that grill, you can give a little nod to the straightforward, single-isomer beauty of propane! Isn't that neat?