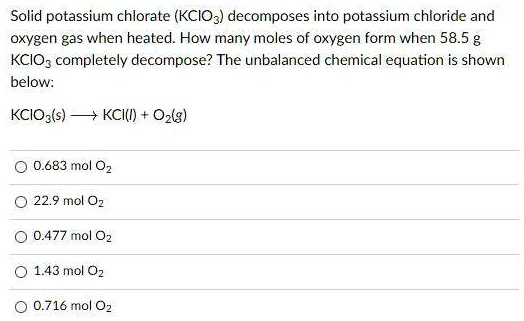
Hey there, science superstars and curious cats! Ever find yourself staring at a pile of Potassium Chlorate, or as we scientists affectionately call it, KClO₃, and wondering, "Just how much of this fizzy-whizzy stuff do I need to make, say, a spectacular bang or a dramatic puff of smoke?" Well, buckle up, buttercups, because we're about to dive into the wonderfully weird world of chemical quantities, and it’s going to be a blast – no pun intended… okay, maybe a little pun intended!
You see, in the grand ol' kitchen of the universe, when atoms and molecules decide to get together and have a party – that’s a chemical reaction, folks! – they don't just grab random amounts of each other like you might grab a handful of jellybeans. Nope, nope, nope! They have very specific rules, like a super-organized potluck. And one of the most important rules is about how much of each ingredient you need to make sure the reaction goes off without a hitch, or in our case, with just the right amount of oomph.
This is where our marvelous friend, the mole, swoops in like a caped crusader! Now, the word "mole" might make you think of a little furry critter digging tunnels, and while it is a unit of counting, it’s a giant unit of counting. We’re talking about counting atoms and molecules, those tiny invisible building blocks of everything. Imagine trying to count every single grain of sand on a beach – that’s kind of the scale we’re dealing with! A mole is simply a way to bundle up a ridiculously huge number of things, specifically 6.022 x 10²³ of them. Yeah, that’s a 6 followed by 23 zeros. Even our most ambitious squirrels couldn't hoard that many nuts!
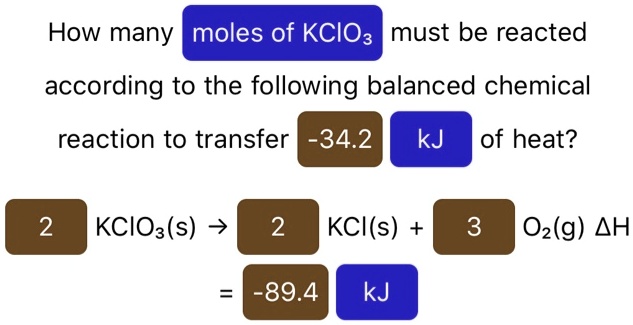
So, when we ask, "How many moles of KClO₃ must be reacted?", we're not just asking about a handful. We're asking about a specific, astronomical number of Potassium Chlorate molecules needed to achieve a particular chemical outcome. It’s like asking, "How many jellybeans do I need to make this entire giant jellybean mosaic of the Mona Lisa?" The answer is going to be… a lot!
Let’s imagine our KClO₃ is the star ingredient for a super-duper firework. We want a magnificent burst of light and color, not a pathetic fizzle or a catastrophic explosion that rearranges the furniture in your neighbor's living room. To get that perfect burst, we need to know exactly how many KClO₃ molecules need to dance with their chemical partners. And that’s where the mole comes in, acting as our universal measuring cup for microscopic ingredients.

Think about baking a cake. If a recipe says you need 2 cups of flour, you don't just eyeball it, right? You measure it out precisely. In chemistry, the mole is our super-precise measuring tool for ingredients that are too small to see. When we're talking about KClO₃, it’s not just about plopping some powder in a dish. We need to know the mole count to ensure the reaction proceeds as planned, creating the desired effect, whether that’s a gentle decomposition into oxygen and Potassium Chloride (KCl), or a more enthusiastic performance.
Sometimes, our KClO₃ is part of a grander chemical symphony. It might be reacting with something else, like sugar, to create a classic, fun reaction (don't try this at home without expert supervision, folks!). The amount of KClO₃ we need is directly tied to the amount of its dance partner. If you have too much sugar and not enough KClO₃, your sparkly show might be a bit dull. If you have way too much KClO₃ and not enough sugar, well, let’s just say things could get… unnecessarily exciting.

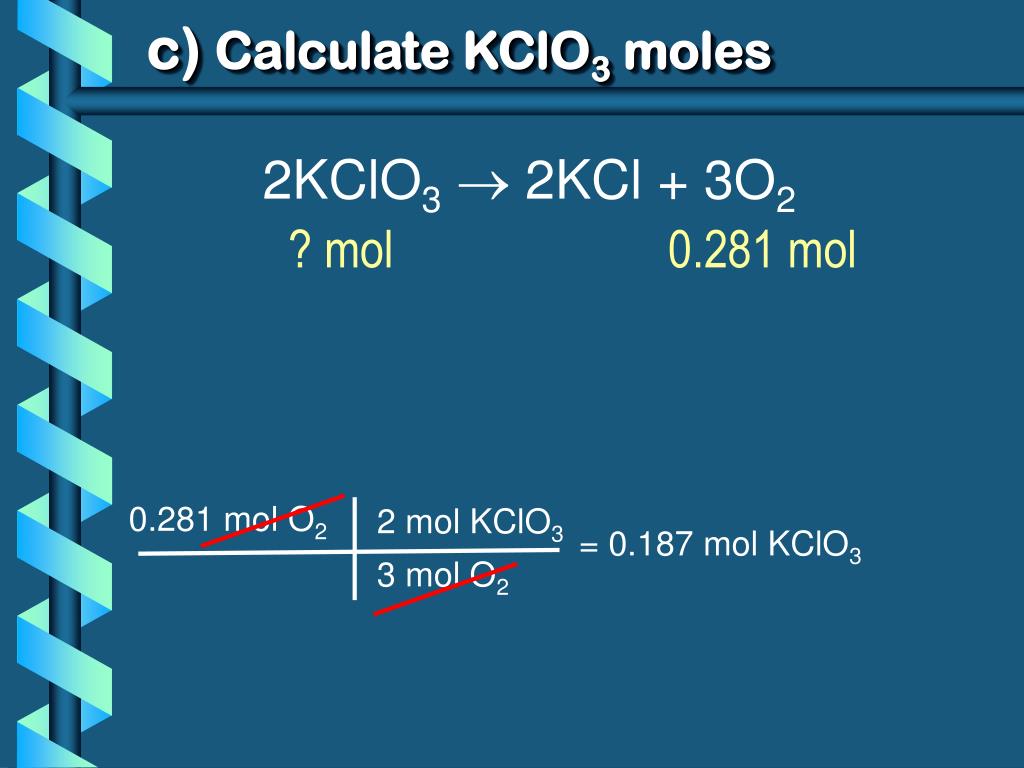
The beauty of the mole is that it allows us to scale things up or down with absolute confidence. If we know that, for instance, 2 moles of KClO₃ will give us a fantastic puff of smoke, and we only want a tiny whisper of smoke, we can simply use half a mole. It’s all about proportion, like scaling a recipe for one person versus a whole birthday party. The underlying ratio stays the same, just the total quantity changes.
So, the next time you hear someone ask about the number of moles of KClO₃ that need to be reacted, don't picture a scientist with a tiny squirrel. Picture a meticulously organized chemist, armed with the ultimate measuring stick for the invisible world, ensuring that every chemical reaction, from the most subtle to the most spectacular, performs exactly as intended. It’s all about precision, a dash of calculation, and a whole lot of chemical fun!