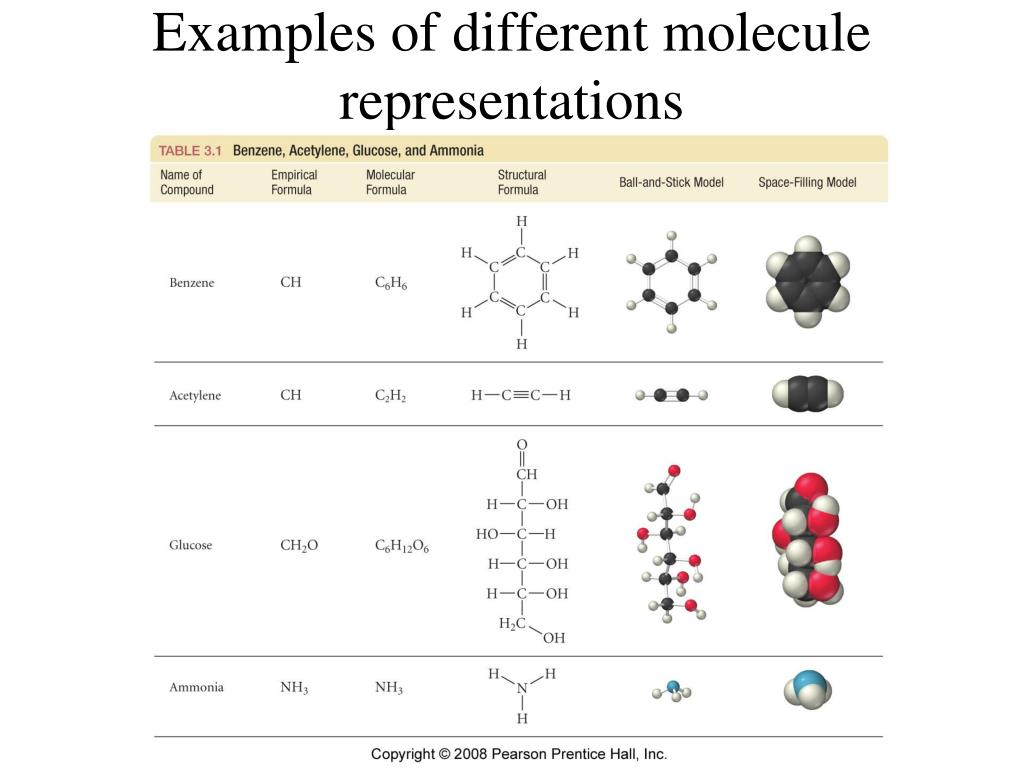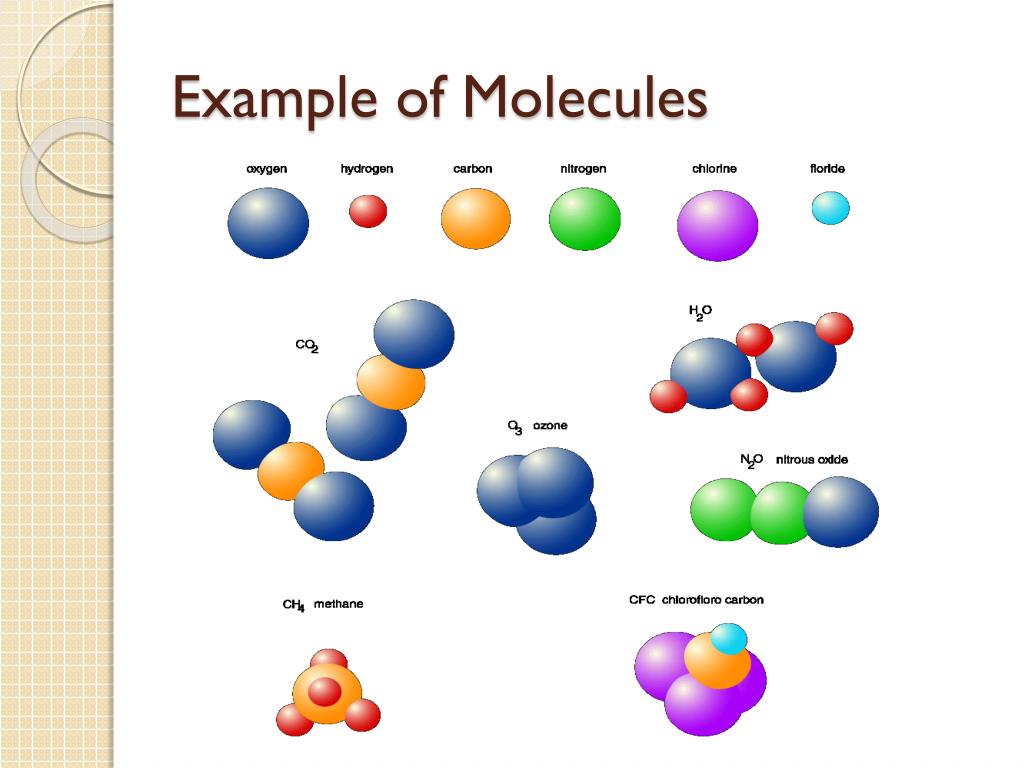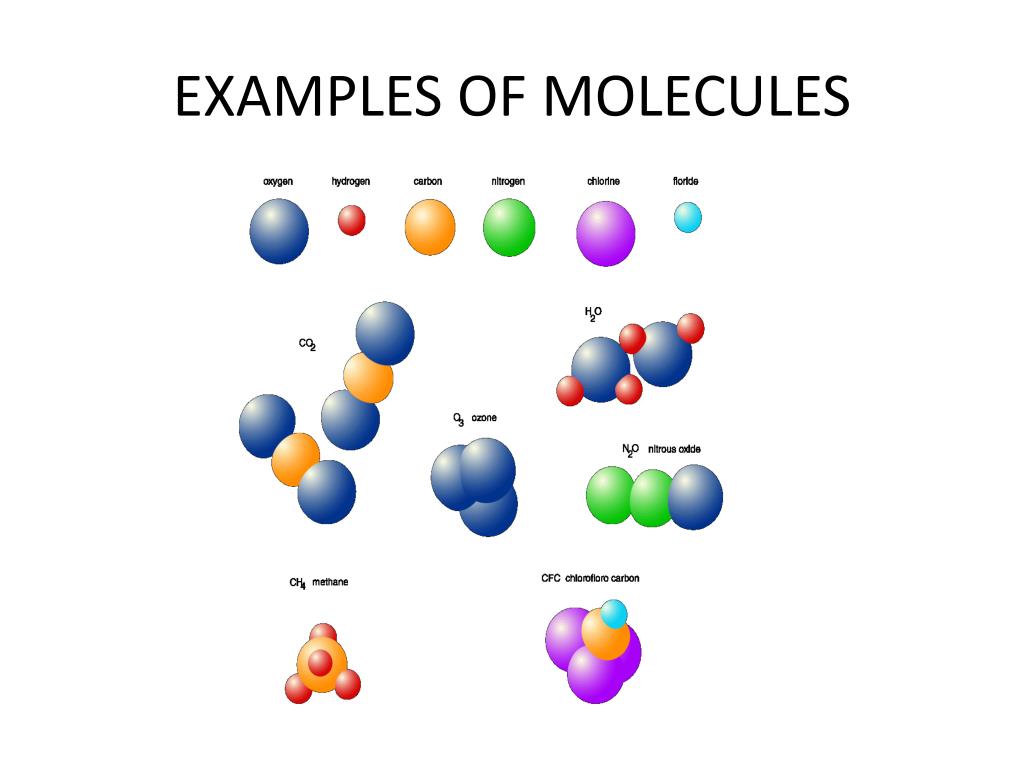
Ever wondered about the hidden world within the things you touch, see, and even breathe? It’s a realm packed with unimaginably tiny building blocks called molecules. While we can’t see them with our own eyes, these invisible particles are the fundamental ingredients of everything around us, from the water we drink to the air we inhale. And the truly mind-boggling part? The sheer number of them in even the smallest pinch of substance is astronomical! This isn't just a topic for dusty textbooks; understanding how many molecules are in a sample is surprisingly fun, incredibly useful, and has sparked a lot of popular curiosity.
Think about it: when you taste a sweet drop of honey, you're experiencing the collective action of billions upon billions of sugar molecules. When you feel the coolness of a glass of water, it's the constant dance of water molecules. The very concept of "how many" in this microscopic world is a gateway to appreciating the complexity and interconnectedness of our universe. It’s like having a secret decoder ring for reality, revealing the incredible scale of the matter we interact with every single moment.
The Astonishing Count: Unveiling the Molecular Universe
So, how do scientists even begin to figure out these colossal numbers? It’s not like they have tiny molecule counters! The key lies in a concept called the mole. Don't let the name fool you; it’s not referring to a tiny burrowing animal! In chemistry, a mole is simply a convenient way to count a specific, enormous number of particles. This magical number is known as Avogadro's number, which is approximately 6.022 x 1023. To put that into perspective, that's a 6 followed by 23 zeros! Imagine having that many grains of sand on all the beaches in the world – it's even more than that!
The beauty of the mole is that it connects the microscopic world of molecules to the macroscopic world we can measure. For example, scientists know the molar mass of a substance, which is the mass of one mole of that substance in grams. By weighing a sample and knowing its molar mass, they can then calculate how many moles are present. And from there? It's a straightforward multiplication by Avogadro's number to arrive at the mind-blowing total number of molecules.
Let’s take water (H2O) as a prime example. A single drop of water might seem insignificant, but it contains an incredible number of water molecules. If we consider just 18 grams of water – which is roughly a tablespoon – that amount is precisely one mole. This means that single tablespoon of water holds approximately 6.022 x 1023 water molecules! It’s a staggering thought, isn't it? Every time you take a sip of water, you're ingesting an unfathomable number of these tiny H2O units.
The implications of this go far beyond mere curiosity. Understanding the number of molecules in a sample is crucial for countless scientific and practical applications. In medicine, for instance, dosages of drugs are carefully calculated based on the number of active molecules needed to achieve a therapeutic effect. Too few molecules, and the drug might not work; too many, and it could be toxic. This precision relies on the principles of moles and Avogadro’s number.
Imagine a single grain of salt. It's so small, yet it contains approximately 1.5 x 1020 sodium chloride (NaCl) molecules. That's 150 quintillion molecules in that tiny crystal!
SOLVED: How many molecules are present in each sample? 6.85 mol SO2 0.
In the food industry, understanding molecular counts helps in controlling flavors, textures, and nutritional content. When you see "low sodium" on a food label, it’s a reflection of controlled amounts of sodium-containing molecules. In environmental science, tracking pollutants often involves counting the molecules of specific contaminants in air or water samples to assess their impact and develop cleanup strategies.
Even in everyday activities, this concept plays a role. When you bake a cake, the precise measurements of ingredients ensure the right chemical reactions occur, driven by the specific number of molecules of each component interacting. The science behind a fluffy cake or a perfectly risen loaf of bread is, at its core, about orchestrating the behavior of vast numbers of molecules.
The concept of counting molecules isn't just for chemists in labs. It's a foundational idea that helps us appreciate the scale of the universe and the intricate workings of the substances we rely on. It transforms our perspective, making the ordinary extraordinary and revealing the hidden, bustling molecular world that makes up everything we know. So, the next time you look at a glass of water or a pinch of salt, remember the unbelievable multitude of molecules present, each playing its part in the grand tapestry of existence.