Hey there, science explorers! Ever wonder about the tiny, invisible world all around us? It’s like a secret universe packed with things so small you can’t even see them. Today, we’re going to dive into a fun little puzzle from that world. We’ll be playing detective with some special stuff called N₂O₅.
Now, N₂O₅ might sound a bit like a secret agent name, right? It's a molecule, which is basically a tiny building block of matter. Think of it like a LEGO brick, but unimaginably smaller! And we’re talking about a very specific amount of these bricks: 0.400 moles of them.
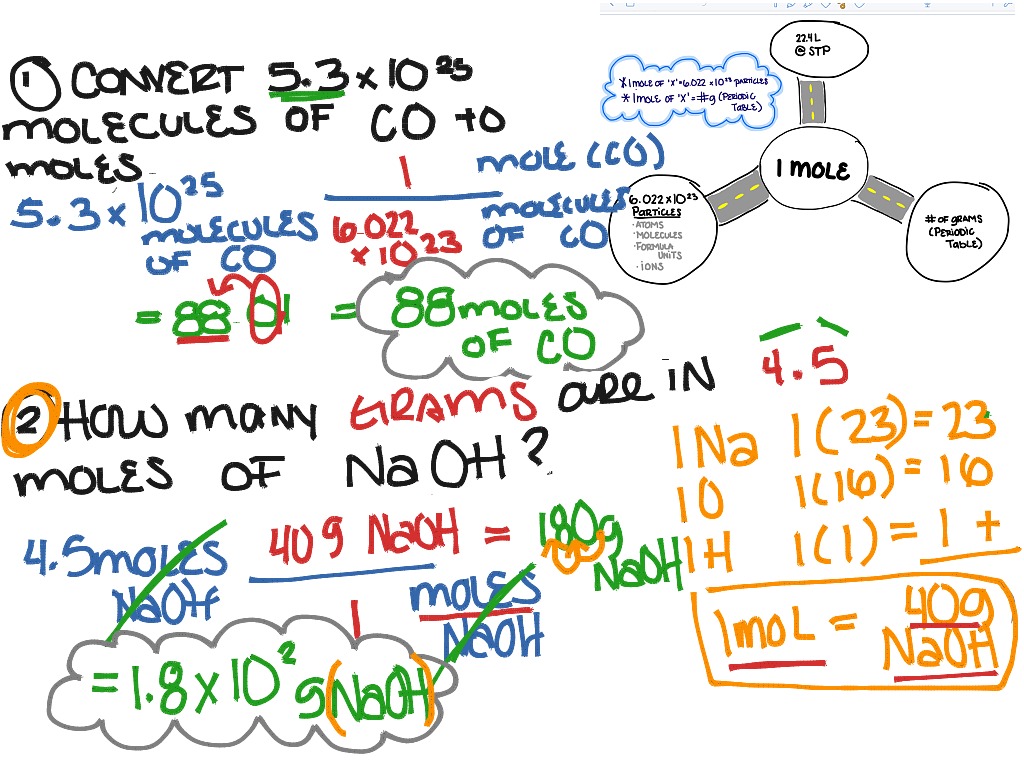
So, what’s a "mole" anyway? It’s not the little guy who digs tunnels! In chemistry, a mole is just a special way of counting. It’s like saying "a dozen" for eggs. A mole is a HUGE number of particles. It’s 6.022 x 10²³ particles, to be exact! That's a 6 followed by 23 zeros! Mind-boggling, isn't it?
Imagine trying to count that many things! It would take forever, even if you had a super-fast counting machine. But that’s the magic of the mole. It gives scientists a handy way to talk about really, really big numbers of molecules without getting lost in a sea of zeros.
Our mission, should we choose to accept it, is to figure out how many individual N₂O₅ molecules are hiding in those 0.400 moles. It's like asking: if you have 0.400 dozen eggs, how many actual eggs do you have? You'd multiply the number of dozens by 12, right? This is kind of the same idea, but with a much, much bigger multiplier!
This is where the real fun begins. We get to put on our math hats and do some super-cool calculations. It’s not scary math, promise! It's more like a fun brain teaser. We're unlocking a secret number.
So, we have our amount of N₂O₅, which is 0.400 moles. And we know that one mole is equal to 6.022 x 10²³ particles. These particles, in our case, are the N₂O₅ molecules.
To find the total number of molecules, we simply multiply the number of moles we have by the number of particles in one mole. Easy peasy, lemon squeezy!
Let’s break it down. We take our 0.400 moles of N₂O₅. Then, we multiply that by the magic number, 6.022 x 10²³ molecules/mole. The "mole" units cancel out, leaving us with just molecules!
It’s like a cancellation party for units! Poof! They disappear, and we’re left with the answer we're looking for.
The calculation looks like this: 0.400 x 6.022 x 10²³.
Now, let’s do a little mental multiplication. Think of 0.4 as just a bit less than half. So, we're expecting an answer that's a bit less than half of 6.022 x 10²³. That's still a massive number, though!
When we crunch the numbers, we get something truly spectacular. It’s approximately 2.409 x 10²³ molecules of N₂O₅.
Can you even wrap your head around that number? 2.409 followed by 23 zeros! That’s more than all the grains of sand on all the beaches in the world. It’s more stars than you can count in the night sky. It’s an almost incomprehensible amount of tiny, invisible things.
What makes this so special? It’s the sheer scale of it. We're talking about a handful of N₂O₅ (well, 0.400 moles is a reasonable amount) and it contains an astronomical number of individual molecules. It’s a testament to how small things can be and how many of them can exist.
Think about it: each and every one of those 2.409 x 10²³ molecules is identical to the others. They all have the same structure, the same atoms bonded together in the same way. It’s a perfect, tiny army of N₂O₅.

This isn't just a random fact; it's the foundation of chemistry. Understanding these numbers helps scientists predict how substances will react, how much of something they need for an experiment, and how much product they'll get. It's like having the secret code to how the universe works at its most fundamental level.
N₂O₅, or dinitrogen pentoxide, is a fascinating molecule. It's a colorless solid that readily decomposes. It plays a role in atmospheric chemistry, particularly in the stratosphere where it can influence ozone depletion. So, it's not just a theoretical concept; it's a real-world player!
The fact that we can take a measurement like "moles" and translate it into an exact count of individual particles is pure scientific brilliance. It bridges the gap between the macroscopic world we experience and the microscopic world we can only imagine.
It’s like being able to say, "I have 0.400 bags of marbles, and in each bag, there are 6.022 x 10²³ marbles." Then, you can calculate the total number of marbles you have. But instead of marbles, it’s these incredibly tiny molecules.
This concept helps us appreciate the complexity and the sheer abundance of matter around us. Every breath you take, every sip of water, every glance at a flower – it’s all made up of an unbelievable number of molecules. Our calculation for N₂O₅ is just one tiny glimpse into that vast, molecular universe.
So, the next time you hear about moles in chemistry, don’t just think of it as a weird unit. Think of it as a gateway to understanding the incredible number of building blocks that make up everything. It's a number so big it feels almost magical.
And for 0.400 moles of N₂O₅, we've calculated there are a mind-boggling 2.409 x 10²³ molecules. Isn't science amazing? It's a journey into the incredibly small and the incredibly numerous, all wrapped up in one fascinating number.
The next time you're curious about what's in a specific amount of something, remember this puzzle. You have the power to unlock the secrets of the molecular world, one calculation at a time!
It’s this kind of insight that makes chemistry so much more than just formulas and reactions. It’s about exploring the unseen, quantifying the immeasurable, and marveling at the sheer scale of existence. So, go forth and explore the wonderful world of molecules!
And remember, for 0.400 moles of N₂O₅, the answer is a colossal 2.409 x 10²³ molecules. Pretty neat, huh?