Ever wondered what makes aluminum foil cling to your leftovers or why aluminum cans are so good at holding fizzy drinks? It all comes down to the tiny, zippy particles inside every atom: electrons! And when it comes to aluminum, there's a surprisingly simple answer to how many electrons it likes to play with. It's a bit like figuring out how many cookies your dog wants to steal – there's a common behavior you can usually predict!
Understanding how aluminum interacts with electrons isn't just for super-smart scientists; it's actually quite useful and can be a lot of fun for anyone curious about the world around them. For beginners, it’s a fantastic starting point for grasping basic chemistry concepts. Think of it as learning the alphabet before you can read a book. For families exploring science together, it’s a great way to spark conversations about matter and how things work. Imagine explaining to your kids why aluminum is used in airplanes – it’s all about its lightweight strength, which is linked to its electron behavior!
Hobbyists, whether you’re into making your own batteries, experimenting with metal etching, or even just understanding how your cookware performs, knowing about aluminum's electron tendencies can give you an edge. For example, when you see aluminum reacting with something, it’s usually because it’s either trying to gain or lose those pesky electrons to achieve a more stable state. And aluminum, bless its heart, is a bit of a generous soul in the electron world.
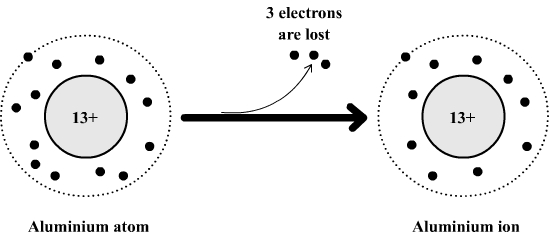
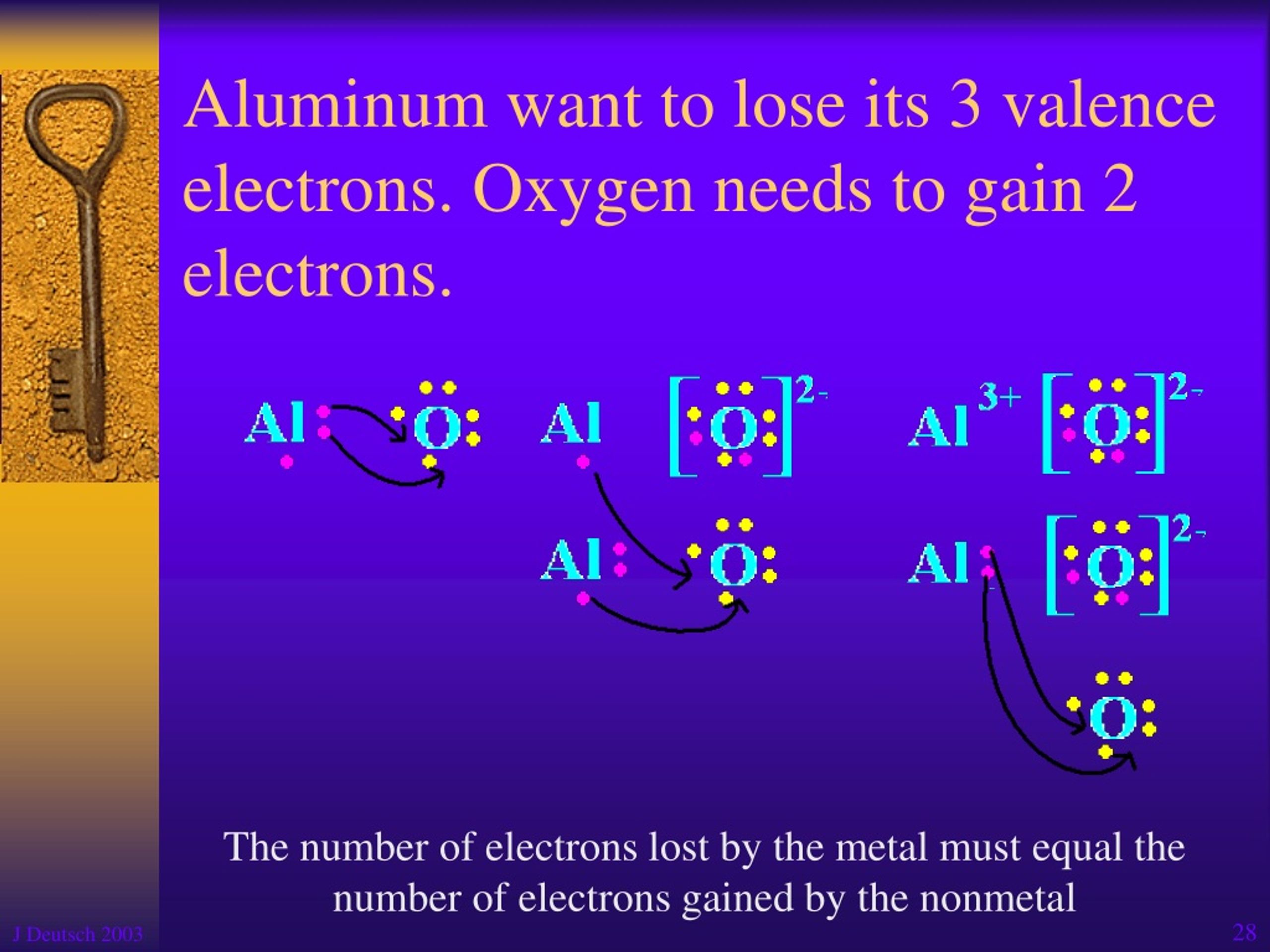
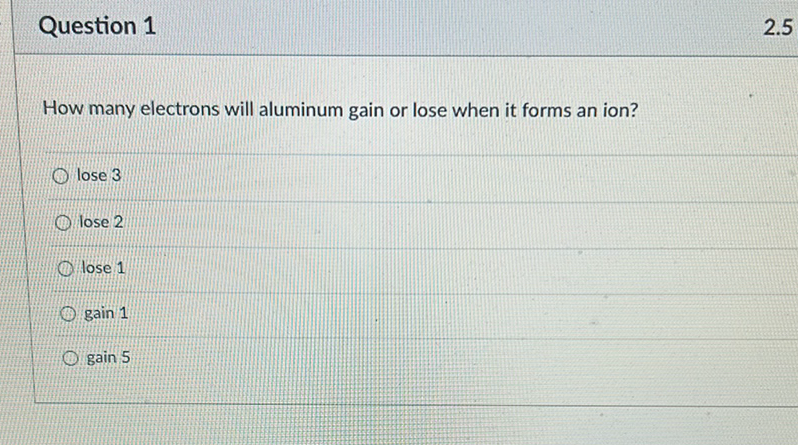
So, how many electrons does aluminum typically gain or lose? The answer is remarkably consistent: aluminum tends to lose 3 electrons. This is because aluminum atoms have three electrons in their outermost shell that are relatively easy to let go of. By losing these three, the aluminum atom becomes an ion with a positive charge. This tendency to lose electrons is what makes aluminum a reactive metal, and it's crucial for its ability to form strong bonds with other elements, like in the strong, lightweight alloys used in everything from cars to phones.
Think about it this way: imagine aluminum as a person with three extra coins they don't really need and are a bit awkward to carry around. It’s much easier and more stable for them to just give those coins away to someone who needs them. Once it's given those coins (electrons) away, it feels more balanced and content. This makes it readily available to form compounds with other elements that are looking to *gain electrons.
For those eager to dive a little deeper, you might encounter variations when aluminum is part of a complex chemical compound, but its fundamental nature is to be electron-deficient. The most common and stable form it takes when reacting is as a positive ion, specifically Al3+, meaning it has lost those three electrons.

Getting started with this concept is super simple! You don't need a fancy lab. Next time you’re using aluminum foil, think about those three electrons. Or, if you have an old aluminum can, consider how its structure is a result of atoms that happily shed those outer electrons. You can even find simple online diagrams showing atomic structure to visualize this electron behavior. It’s all about observation and a little bit of imagination!
Ultimately, understanding how many electrons aluminum gains or loses – spoiler alert: it loses 3 – is a gateway to appreciating the fascinating world of chemistry. It’s a small piece of knowledge that unlocks bigger understandings about the materials we use every single day, making the ordinary a little more extraordinary and a whole lot more fun!