Hey there, curious minds! Ever found yourself staring up at the night sky, or maybe just at the intricate design of a snowflake, and thought, "Wow, there's so much going on under the hood!"? Well, guess what? The same applies to the teeny-tiny building blocks of everything – atoms! And today, we're diving into a particularly snazzy corner of atomic real estate: the 3d subshell. Sounds a bit technical, right? But stick with me, because understanding how many electrons can party in this particular space is actually a surprisingly delightful little puzzle that can add a little sparkle to your day.
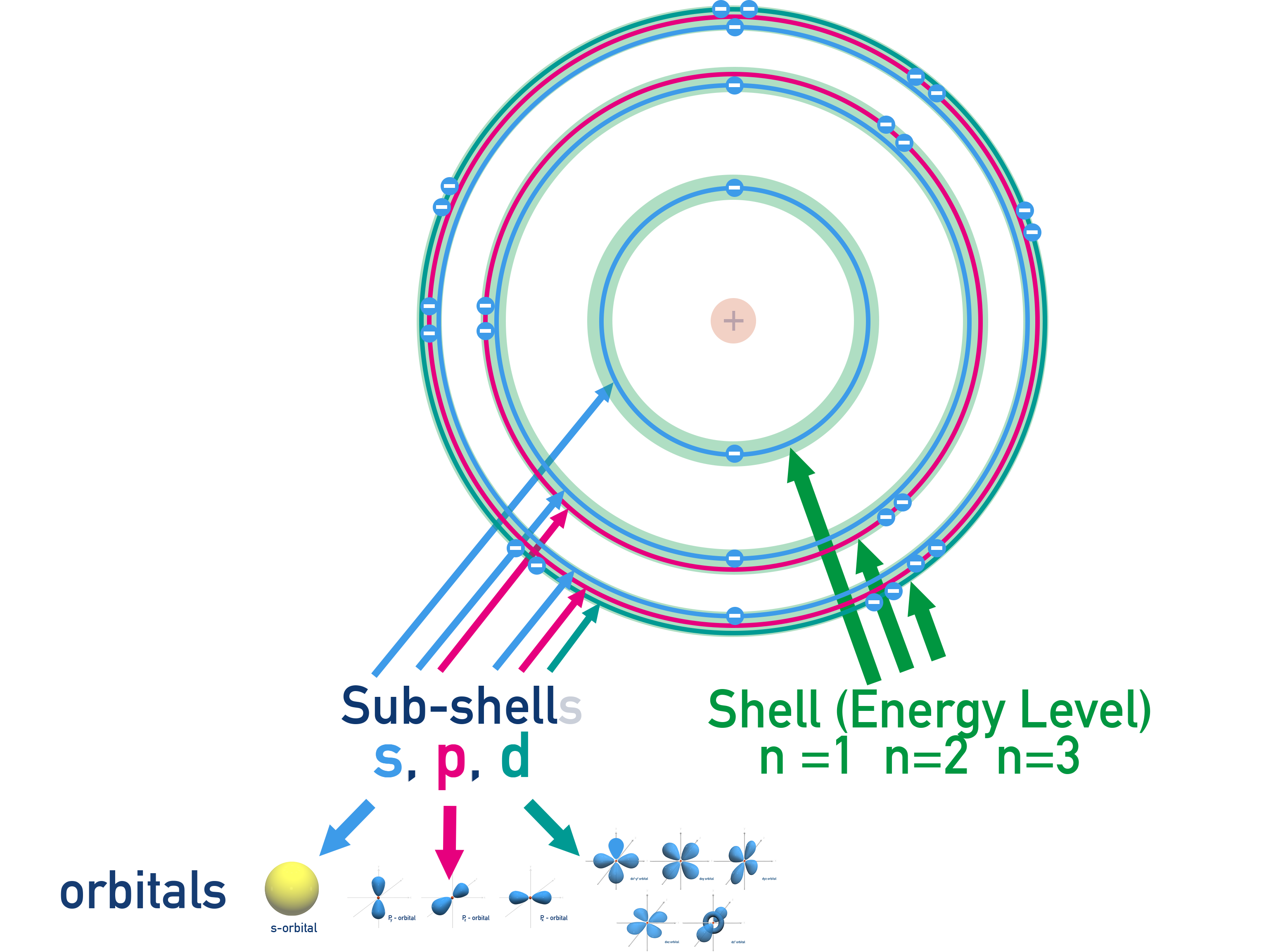
So, let's talk about electrons. Think of them as the energetic little dancers of the atomic world. They're not just randomly bouncing around; oh no, they have their favorite spots, their preferred dance floors, if you will. These spots are organized into shells and, within those shells, even smaller areas called subshells. It's like a cosmic apartment building with different neighborhoods!
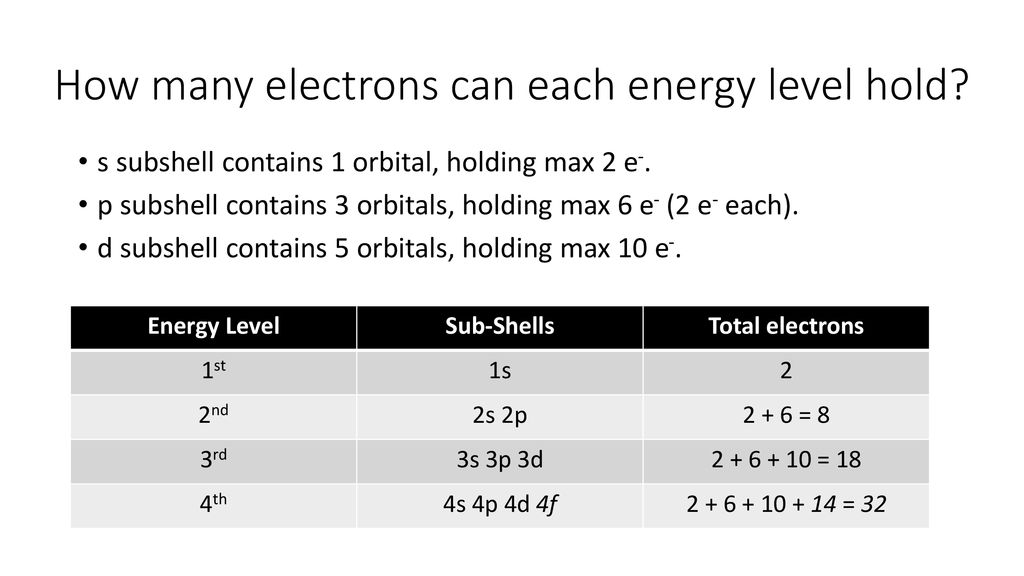
We've got the K shell (that's the first one, super close to the nucleus), then the L shell, and so on. Each shell can hold a certain number of electrons, and within each shell, we have different types of subshells, labeled with letters: s, p, d, and f. Today's star player is the 'd' subshell, and we're specifically looking at the one in the third shell – hence, the 3d subshell. Easy peasy so far, right?
The Mystery of the 3d Subshell: A Capacity Question
Now, the big question of the day: how many of our electron dancers can fit into the 3d subshell? Drumroll, please... The answer is a charming ten! Yep, ten electrons can comfortably groove and move within the 3d subshell. Isn't that neat? It’s like finding out your favorite coffee shop has exactly ten seats at the counter – a perfect little capacity.
But why ten? This is where things get a smidge more intricate, but trust me, it's a fun kind of intricate. It all comes down to the shape and orientation of these subshells. Think of the 's' subshell as a single, cozy spherical room. It can hold a maximum of two electrons. Then we have the 'p' subshell, which is like having three dumbbell-shaped rooms, all oriented in different directions. Each of those rooms can hold two electrons, so that's six in total. Getting the picture?

The 'd' subshell is where things get a bit more elaborate. It's like having not just one or two, but five distinct regions, each capable of holding two electrons. These five regions, called orbitals, have more complex shapes – think cloverleafs and a sort of donut with a dumbbell! Each of these five orbitals can house a pair of electrons, and when you multiply those five orbitals by two electrons each, what do you get? You guessed it – ten electrons!
The Magic of Orbitals: Where Electrons Find Their Place
These orbitals are like tiny, personal spaces for electrons. And here’s a super important rule, called the Pauli Exclusion Principle: each orbital can hold a maximum of two electrons, but they have to have opposite "spins." Imagine them as two little dancers in a small room, one spinning clockwise and the other counter-clockwise. They can share the space, but they have to be doing their own unique spin. This is a fundamental principle that dictates how atoms are built, and it’s pretty darn cool when you think about it!
So, the 3d subshell, with its five orbitals, can accommodate these ten electron pairs. This capacity for ten electrons in the 3d subshell is actually a big deal in chemistry. It's the reason why we have so many different elements with fascinating properties. Think about transition metals – elements like iron, copper, and gold. Their characteristic behaviors are often due to how their electrons fill up these d orbitals!

The filling of the d subshell is a bit like a game of musical chairs. As you add more electrons to an atom, they fill up these orbitals in a specific order, influenced by energy levels and repulsion. Sometimes, an orbital might be half-full (five electrons), which is a particularly stable arrangement, like a team that's perfectly balanced. Other times, it’s full (ten electrons), also a state of good stability. It’s this dance of filling and emptying that gives elements their unique personalities!
Seriously, isn't that just marvelous? We're talking about the fundamental architecture of reality, and it's governed by rules that are both elegant and surprisingly… predictable. The fact that a specific region in an atom, the 3d subshell, has this exact capacity of ten electrons, isn't just a random number. It's a key to understanding the diversity and complexity of the universe around us. From the vibrant colors of a peacock's feather to the conductivity of a metal wire, so much of it is tied back to these electron arrangements!
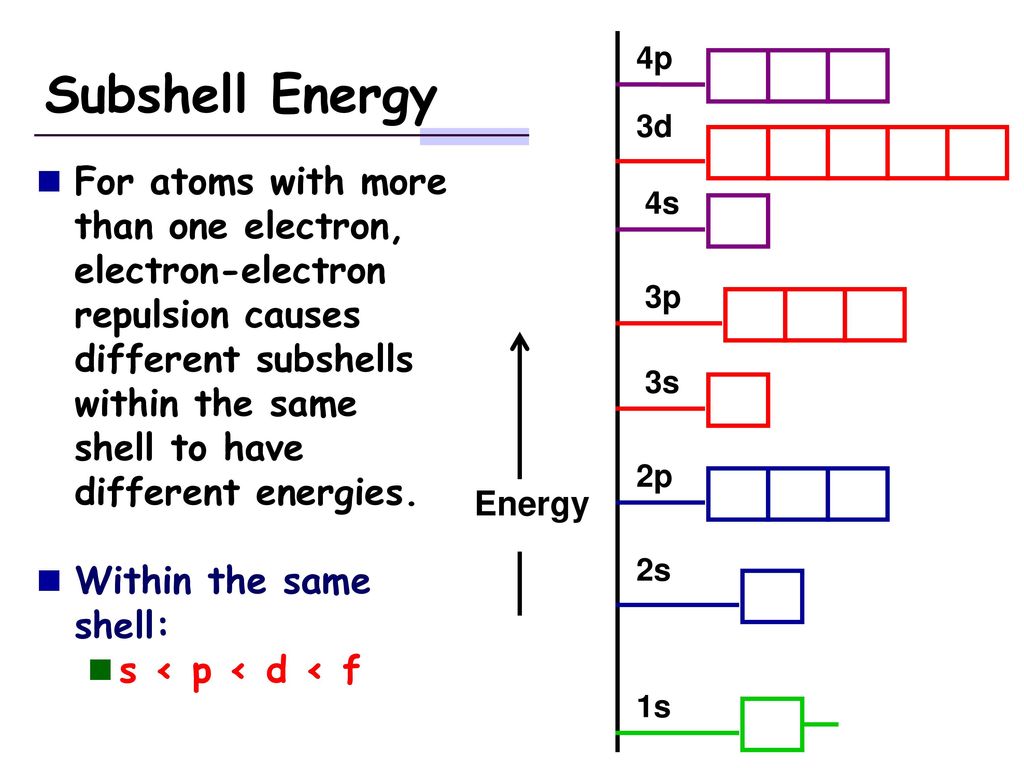
It’s like having a secret code. Once you know that the 3d subshell holds ten, you can start to see it everywhere. You can look at the periodic table and understand why certain elements behave the way they do. It’s not just a collection of boxes; it’s a map of atomic potential! It’s a reminder that even in the smallest, most invisible parts of our world, there’s a remarkable order and a beautiful logic at play.
So, the next time you’re feeling a bit uninspired, remember the 3d subshell. Remember that even in the realm of atoms, there’s a capacity for ten, a structure that allows for complexity and wonder. It’s a tiny glimpse into the incredible engineering of nature, and it’s a story that continues to unfold the more we explore.
This little tidbit about electron capacity is just the tip of the iceberg. The world of atomic structure is brimming with fascinating concepts, each one a stepping stone to a deeper understanding of everything. So, why not let this spark your curiosity? Dive a little deeper, ask a few more questions. You might just find that the universe, in all its electron-filled glory, is even more fun and inspiring than you ever imagined!