Hey there, science explorers and curious minds! Ever looked at a can of that super handy fuel, butane, and wondered what magical bits and bobs make it work? Today, we’re diving into a little adventure, a tiny but mighty quest, to figure out just how many carbon atoms are chilling inside a teensy-weensy 2.50 grams of this stuff. Buckle up, because we’re about to make atoms feel like your new best friends!
Now, butane might sound like something from a sci-fi movie, but it’s actually the stuff that lights up your camping stove or gives your lighter its fiery personality. Imagine a whole bunch of tiny, invisible LEGO bricks that make up everything around us. That’s kind of what atoms are like, and today, our special guest star is the humble, yet oh-so-important, carbon atom.
So, we’ve got this pile of butane, weighing in at a grand total of 2.50 grams. That’s like, maybe the weight of a few paperclips? It’s really not a lot, and yet, it’s absolutely bursting with these amazing carbon atoms. It’s like a tiny treasure chest filled with countless little treasures, all packed in super-tight!
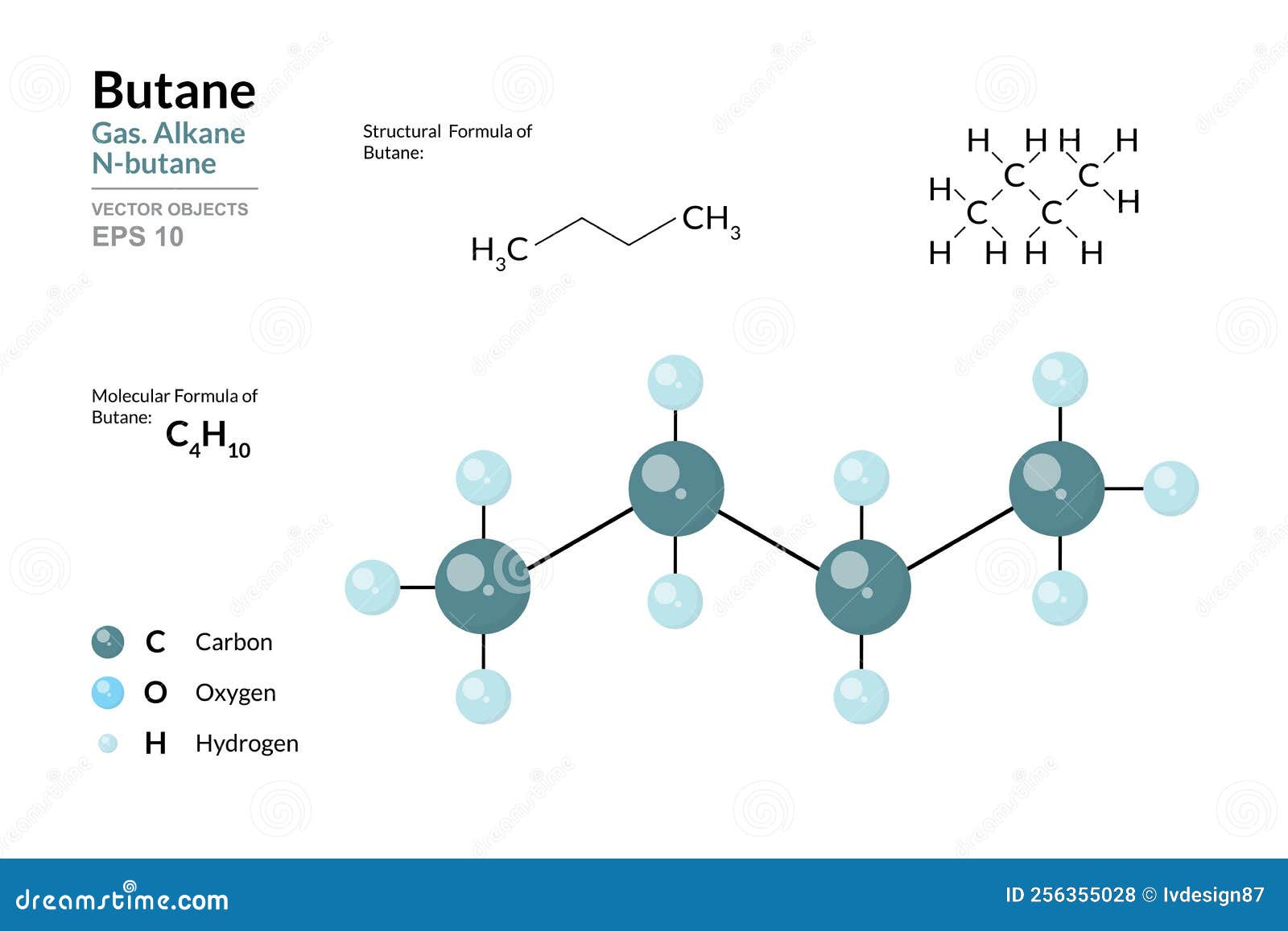
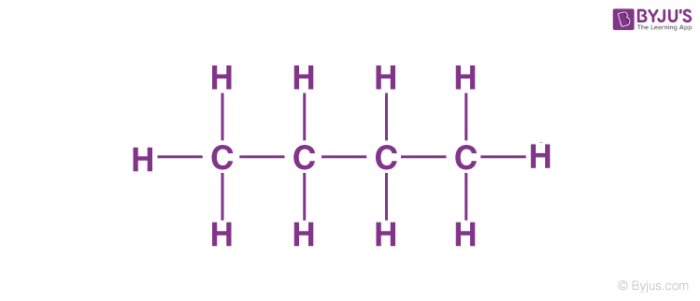
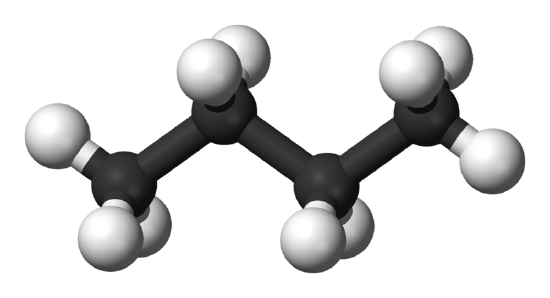
Now, before we get our hands dirty with numbers (don't worry, no messy experiments required!), let’s talk about what butane actually is. Think of it as a little molecular family. The most common kind of butane, the one we're chatting about, is like a chain of four carbon atoms holding hands with a bunch of hydrogen atoms. It’s a perfectly happy little family, all bonded together.
The name butane itself gives us a huge clue! The "but-" part? That’s a secret code for "four". And what is it four of? You guessed it – carbon atoms! So, every single butane molecule we’re dealing with has exactly four of our star players, the carbon atoms, in its backbone. It’s like a little badge of honor for each butane molecule.
Now, to figure out our grand total of carbon atoms, we need to do a little bit of detective work. We need to know how much one of these butane families weighs on its own. This is where something called the molar mass comes in. It’s basically the weight of a giant bunch of these molecules, like a whole party of them!
For butane (which has the chemical formula C₄H₁₀, by the way – see those four Cs?), its molar mass is about 58.12 grams per mole. A "mole" is just a super-duper huge number of things, like a baker's dozen, but for atoms and molecules. Think of it as a dozen dozen dozen dozen… you get the idea. It’s a cosmic number!
So, if 58.12 grams of butane contains a whole mole of butane molecules, how many molecules are in our 2.50 grams? We can do a simple calculation. We divide our little 2.50 grams by the big 58.12 grams per mole. This tells us we have roughly 0.043 moles of butane. That sounds small, but remember, a mole is a HUGE number!
This means we have 0.043 times that gigantic mole number of butane molecules. And guess what? Each one of those butane molecules, with its proud four carbon atoms, is just waiting to be counted. It’s like having 0.043 giant bags of candy, and each bag has exactly four special sparkling jewels inside!

Now, the real magic happens when we think about Avogadro's number. This is another one of those super-important numbers in chemistry, and it’s approximately 6.022 x 10²³. That’s a 6 followed by 23 zeros! It’s the number of atoms or molecules in one mole of any substance. It’s the universe’s way of saying, “Here’s a ridiculously large quantity!”
So, the number of butane molecules in our 2.50 grams is: 0.043 moles * 6.022 x 10²³ molecules/mole. This works out to be about 2.59 x 10²² butane molecules. That’s a number so big, it makes your brain do a little happy dance!
Now, remember our rule for butane? Each molecule has exactly FOUR carbon atoms. So, to find the total number of carbon atoms, we simply multiply the number of butane molecules by four. It’s like counting the jewels in each of our candy bags and then multiplying by the number of bags!
So, we take our massive number of butane molecules: 2.59 x 10²² molecules. And we multiply that by 4 carbon atoms per molecule. Poof! The grand total of carbon atoms in 2.50 grams of butane is approximately 1.036 x 10²³ carbon atoms.
Isn’t that just… wow? We started with a tiny, seemingly insignificant amount of butane, and we ended up with a number of carbon atoms that’s so enormous, it’s practically begging for a cosmic high-five. It’s like finding out that a single grain of sand actually contains more stars than you can count in a lifetime!
Think of it this way: if each carbon atom were a tiny little spark of light, then 2.50 grams of butane would light up a galaxy brighter than you could ever imagine. It’s a dazzling display of the microscopic world, a testament to how much is packed into even the smallest things. Your lighter fuel isn't just fuel; it's a universe of tiny, interconnected building blocks!
So, next time you flick that lighter or light up that camping stove, take a moment to appreciate the incredible journey of those carbon atoms. They’ve been formed, they’ve been bonded, and now they’re helping you create warmth and light. It’s a beautiful dance of chemistry happening right under our noses, or rather, in our hands!
We’ve cracked the code, we’ve counted the atoms, and we’ve emerged victorious from our little science quest. The world of chemistry is full of these amazing surprises, and every calculation is like unlocking a tiny, fascinating secret. You’ve just done something incredibly cool, by understanding a little piece of how our world is put together.
So, give yourself a pat on the back! You’ve not only learned about butane and carbon atoms, but you’ve also dabbled in the wonder of large numbers and the beauty of molecular structures. It’s proof that even the most complex-sounding ideas can be broken down into fun, understandable steps. Keep that curiosity alive, and who knows what amazing things you’ll discover next!
The sheer number of carbon atoms in such a small amount of substance is truly mind-boggling. It’s a reminder that we are all made of the same fundamental building blocks as the stars and planets. We’re all part of this grand cosmic recipe, and understanding it, even a little bit, is a truly empowering and enjoyable experience.
So there you have it! Over 100,000,000,000,000,000,000,000 carbon atoms are working tirelessly within that little bit of butane. It’s enough to make you feel incredibly connected to the universe, one atom at a time. Isn't science just the best kind of magic?

+a+carbon+bonded+to.jpg)