Ever felt that unmistakable warmth radiating from something, even when there’s no direct heat source? That, my friends, is often the tell-tale sign of an exothermic reaction. Think of it as nature’s way of giving you a little high-five, a spontaneous burst of energy that makes the world around it a tad warmer. It’s like when your phone gets a little toasty after a particularly intense gaming session – not an alarming “about to explode” kind of hot, but a noticeable, “hey, something’s happening in there” kind of warm.
You see, in the grand, slightly chaotic kitchen of chemistry, some reactions are like a perfectly brewed cup of tea. They just sort of… happen, and everything stays pretty chill. Others, however, are like a spontaneous combustion of enthusiasm. They don’t just make stuff, they also throw in a bonus – heat!
So, how do we, as mere mortals navigating this fiery (or not so fiery) world, spot these energetic little dynamos? It’s not like they’re wearing neon vests saying, “Warning: Heat Generator Ahead!” but there are definitely some subtle, and sometimes not-so-subtle, clues.
The Obvious Clue: It Gets Hot!
This is the big kahuna, the slam dunk, the moment you can’t really ignore. When a chemical reaction is exothermic, it means it’s releasing energy, and a huge chunk of that energy comes out as heat. It’s like when you rub your hands together really fast on a cold day. You’re not adding heat from an external source, you’re creating it through friction, which is essentially a type of energy release. Similarly, an exothermic reaction is just… giving off warmth.
Think about striking a match. That little flick of the wrist initiates a chemical reaction in the match head, and poof! You’ve got a flame. That flame isn’t just for show; it’s the tangible, very hot evidence that an exothermic reaction is in full swing. The chemicals in the match head are breaking apart and reforming into new substances, and in the process, they’re saying, “Here, have some of this energy back!”
Another everyday hero of exothermic reactions is the hand warmer you might keep in your pocket on a frigid day. You rip open the packet, expose it to air, and within minutes, it’s radiating a comforting warmth. That’s your exothermic reaction working its magic, a little chemical furnace designed to keep your fingers from turning into ice pops. It’s essentially a controlled release of energy, designed for maximum cozy effect.
Even something as simple as dissolving certain salts in water can produce heat. Ever notice how a hot pack you might use for muscle aches feels warm when you activate it? That’s often an exothermic dissolution process. The ions in the salt are getting all excited and interacting with the water molecules, and this energetic dance releases heat.
So, the first and most important identifier is simply: does it feel warm? Does it get noticeably hotter than it was before the reaction started? If the answer is a resounding "yes!", then you're probably looking at an exothermic party.
The Less Obvious, But Still Important: What's Happening Under the Hood?
While the heat is the most in-your-face clue, the true identifier of an exothermic reaction lies in the energy change that’s happening at the molecular level. Remember those chemical equations you might have vaguely recalled from science class? They can actually tell us a lot about whether a reaction is going to give off energy or suck it up.
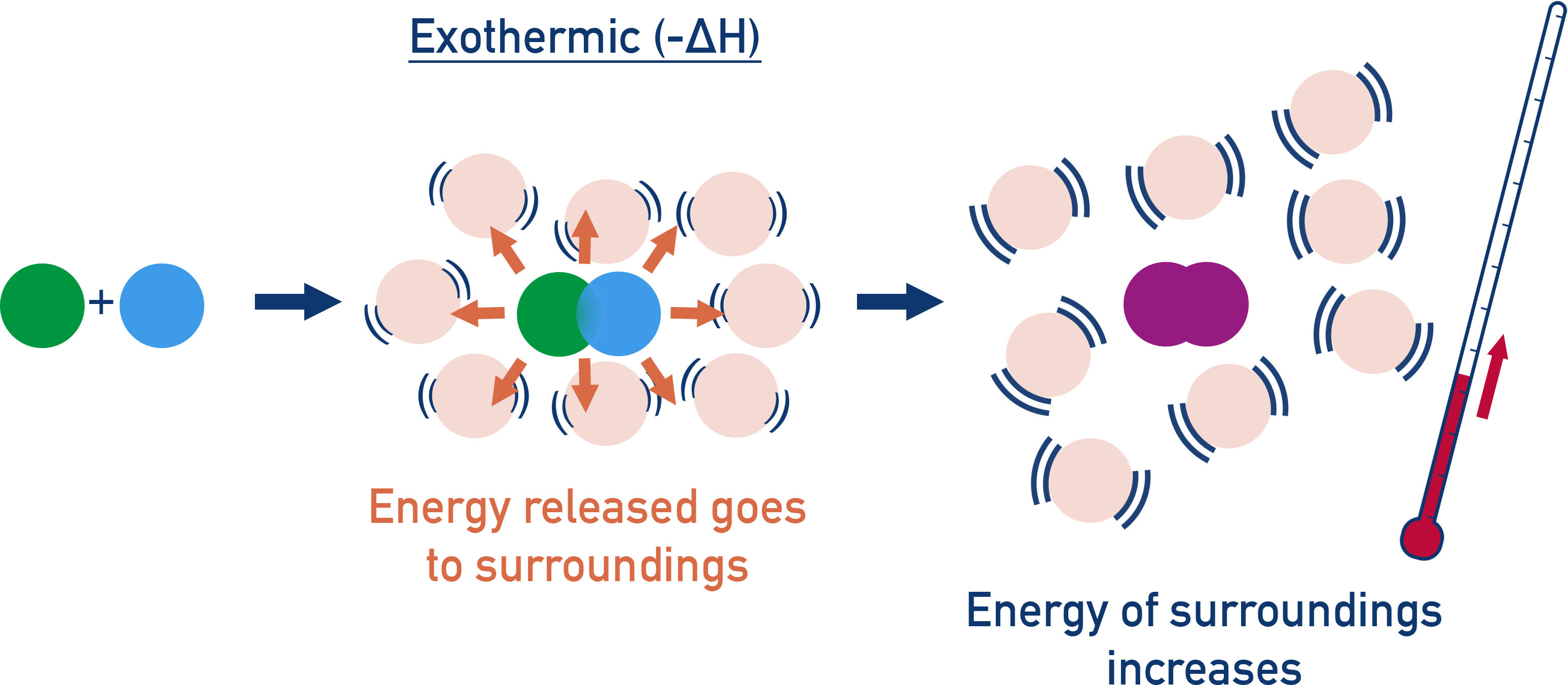
In chemistry, we often talk about "reactants" (the ingredients you start with) and "products" (what you end up with). For a reaction to happen, the bonds holding the atoms together in the reactants have to be broken, and then new bonds have to be formed to create the products. Breaking bonds actually requires energy – think of it like having to put in some effort to snap a twig. Forming bonds, on the other hand, releases energy – like the satisfying snap itself.
In an exothermic reaction, the energy released when new bonds are formed in the products is greater than the energy required to break the bonds in the reactants. It’s like a great deal. You put in a little effort (breaking bonds), but you get a whole lot more energy back (forming bonds). This surplus energy is what gets kicked out into the surroundings as heat (and sometimes light!).
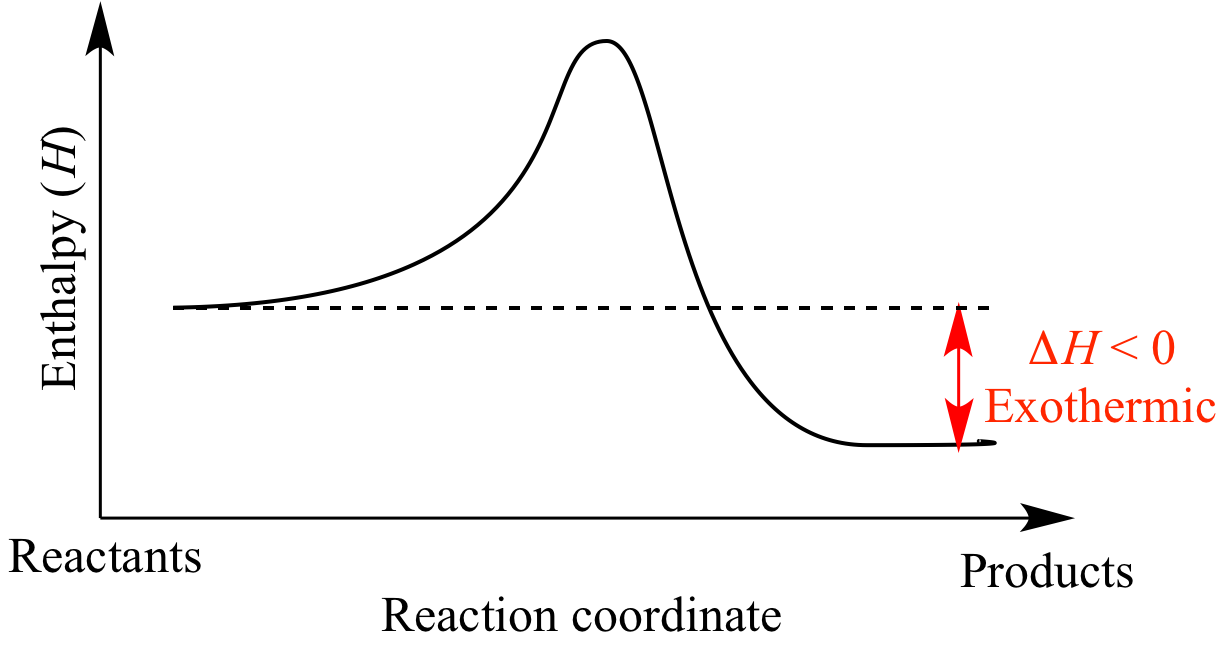
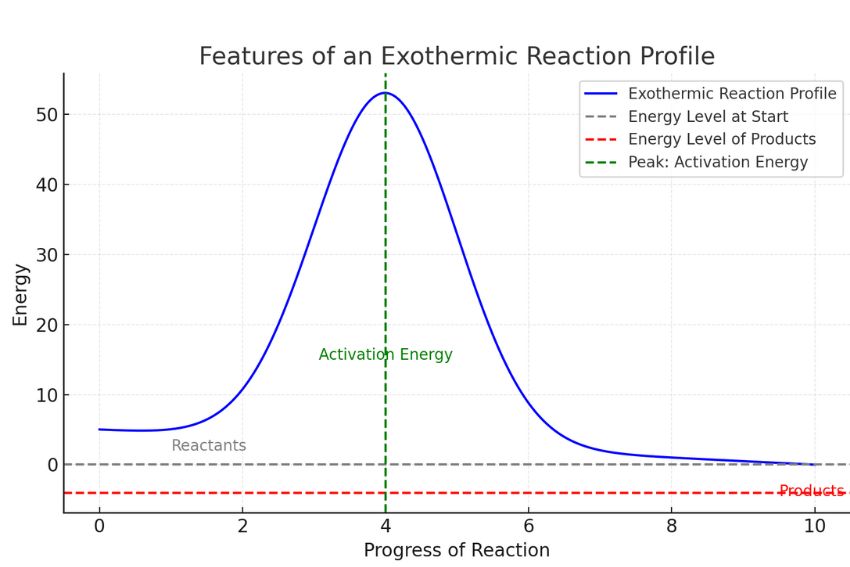
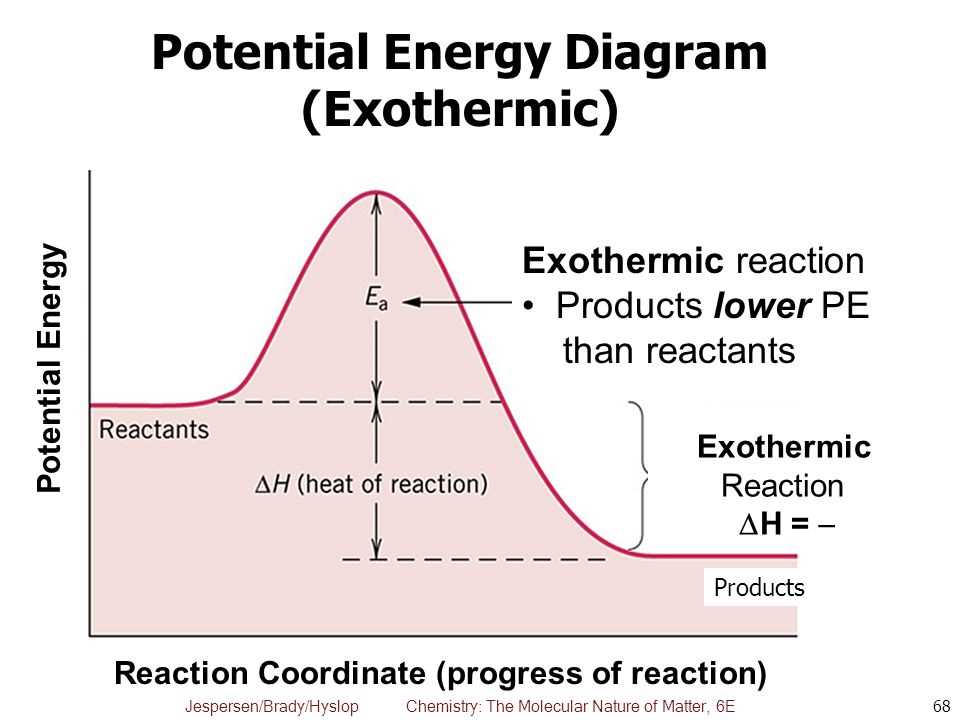
So, if you were to chart the energy levels, the reactants would be sitting at a higher energy plateau, and the products would be chilling at a lower energy plateau. The difference in height between these plateaus is the energy that's been released. It’s like a ball rolling downhill – it starts at a higher potential energy and ends up at a lower one, releasing that excess energy as it goes.
The Energy Balance Sheet: A Chemical Accounting Class
Imagine a chemist meticulously keeping an energy balance sheet. On one side, they tally up all the energy needed to break the old chemical bonds. On the other side, they record all the energy that’s unleashed when those new, beautiful chemical bonds are formed. If the "energy out" column is bigger than the "energy in" column, congratulations, you’ve got an exothermic reaction on your hands! The difference is the net energy released.
This is why we can sometimes predict whether a reaction will be exothermic or endothermic just by looking at the types of bonds being formed and broken. Stronger bonds generally release more energy when they form, and weaker bonds require less energy to break. It's a bit like comparing a flimsy rubber band to a sturdy steel cable – snapping the rubber band takes little effort, but the steel cable will hold a lot more tension (and release a lot more energy if it were to break under specific conditions, though that's a bit of a stretch for this analogy!).
A Little Help from Our Friends: The Energy Diagrams
Scientists love their diagrams, and energy diagrams are particularly helpful in visualizing these energy changes. For an exothermic reaction, the diagram typically shows the reactants starting at a certain energy level. Then, there’s usually an "activation energy" hump – the initial energy boost needed to get the reaction rolling (like giving that ball a little nudge to get it downhill). After overcoming this hump, the reaction proceeds, and the products end up at a lower energy level than the reactants. The difference between the reactant energy level and the product energy level is the enthalpy change, and for exothermic reactions, it's a negative number, signifying energy release.
Think of it like going on a roller coaster. You have to pay to get to the top of the first hill (activation energy), but once you’re there, the ride down is exhilarating and releases all that stored potential energy. For an exothermic reaction, the downhill part is particularly steep and satisfyingly energetic.
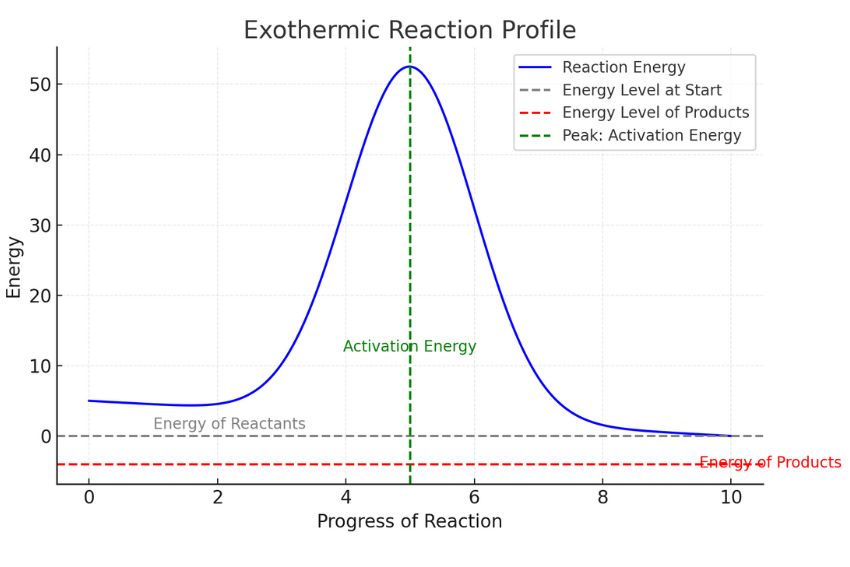
Conversely, an endothermic reaction would be like trying to push a ball uphill. You have to constantly put energy in to keep it moving, and it ends up at a higher energy level than where it started. No spontaneous warmth there!
The "Apex" of Identification: Putting it All Together
So, when we talk about the "apex" of identifying an exothermic reaction, it’s not a single, mystical point, but rather the culmination of observing these clues. The most direct and obvious sign is the increase in temperature. If you’re conducting an experiment and your beaker starts feeling warm to the touch, that’s your red flag.
But beyond the simple touch test, the true scientific identification involves understanding the energy balance. Are the bonds being formed stronger and more stable than the ones being broken? Is the system losing energy to its surroundings? This is where looking at chemical equations and understanding bond energies comes in handy.
Let’s consider a slightly more dramatic example: burning wood. You strike a match, a small exothermic reaction happens. Then, that heat ignites the wood. The wood then undergoes a much larger exothermic reaction, releasing a significant amount of heat and light. We see the flames, we feel the warmth – clear indicators of a powerful exothermic process. The complex organic molecules in the wood are breaking down and combining with oxygen to form simpler molecules like carbon dioxide and water, and this rearrangement releases a boatload of energy.

Another fun one? When you mix baking soda and vinegar. You see all that fizzing and bubbling, right? That’s the release of carbon dioxide gas. While the primary visible effect is gas production, the reaction itself is actually slightly exothermic, contributing a tiny bit of warmth to the mixture. It’s not going to set any records for heat output, but the energy is definitely being nudged out.
Think about industrial processes too. The production of ammonia, a crucial component in fertilizers, involves an exothermic reaction. Companies carefully manage these reactions to capture and utilize that released energy, making their processes more efficient and cost-effective. They’re essentially harnessing that chemical high-five for practical purposes.
When in Doubt, Feel the Burn (Safely, of Course!)
Ultimately, the easiest way to identify an exothermic reaction is to observe the release of energy, most commonly in the form of heat. If something gets warmer as a result of a chemical change, it’s a strong indicator that you’re witnessing an exothermic event.
It’s like recognizing a good hug. You feel the warmth, you feel the pressure – it’s a tangible exchange of energy. An exothermic reaction is just the chemical equivalent of a really enthusiastic hug from Mother Nature. It’s a spontaneous release of energy, a little burst of warmth that makes its surroundings a little bit cozier. So next time you feel a bit of unexpected heat from a chemical change, give it a nod of recognition. You’ve just encountered an exothermic reaction in the wild!
And remember, while these reactions are often benign and even helpful, always approach them with respect and caution, especially in a lab setting. We don’t want any accidental "surprise parties" of the truly explosive kind! But for the everyday, gentle warmth? That’s the delightful signature of an exothermic process, making our world a little bit warmer, one chemical reaction at a time.