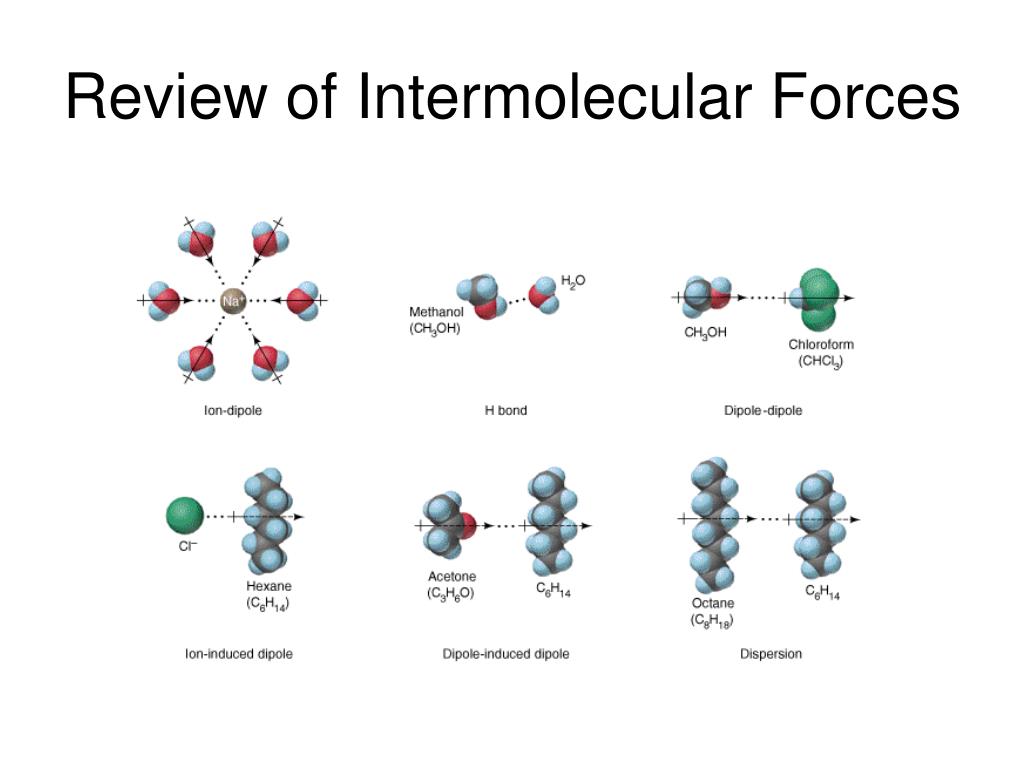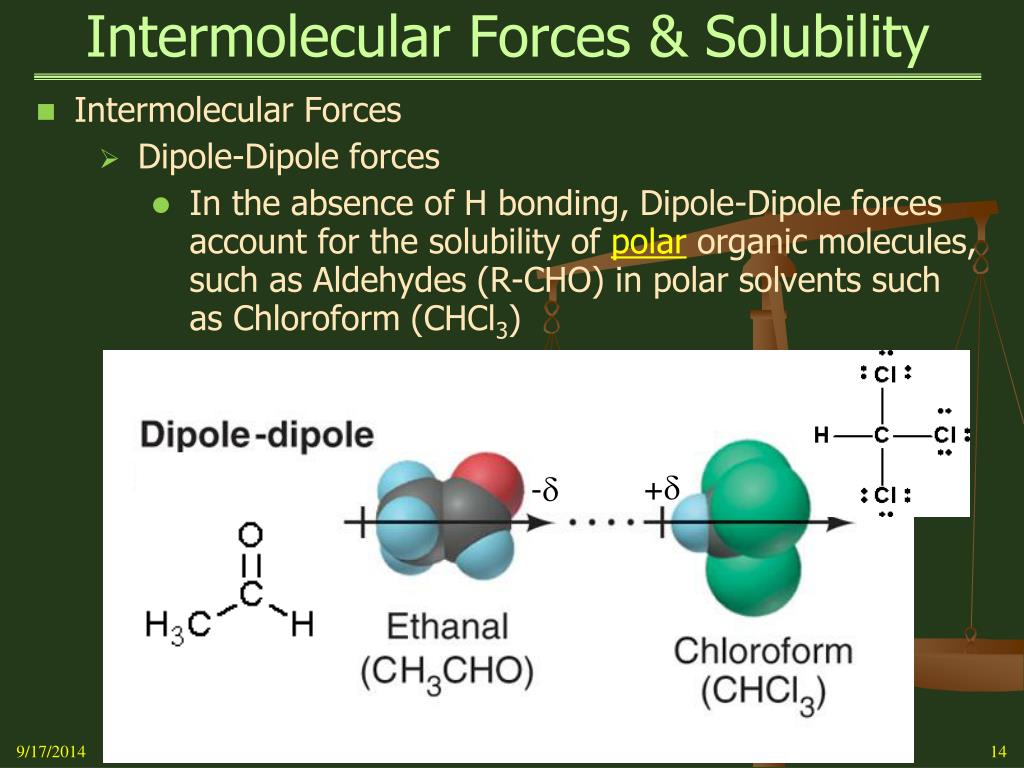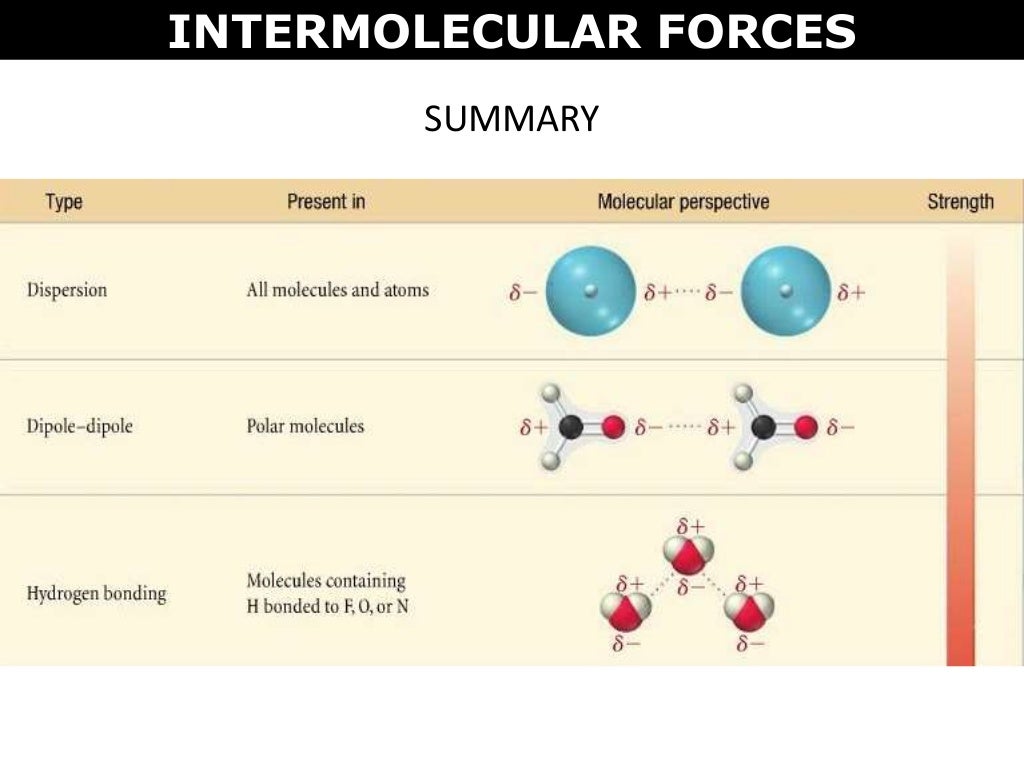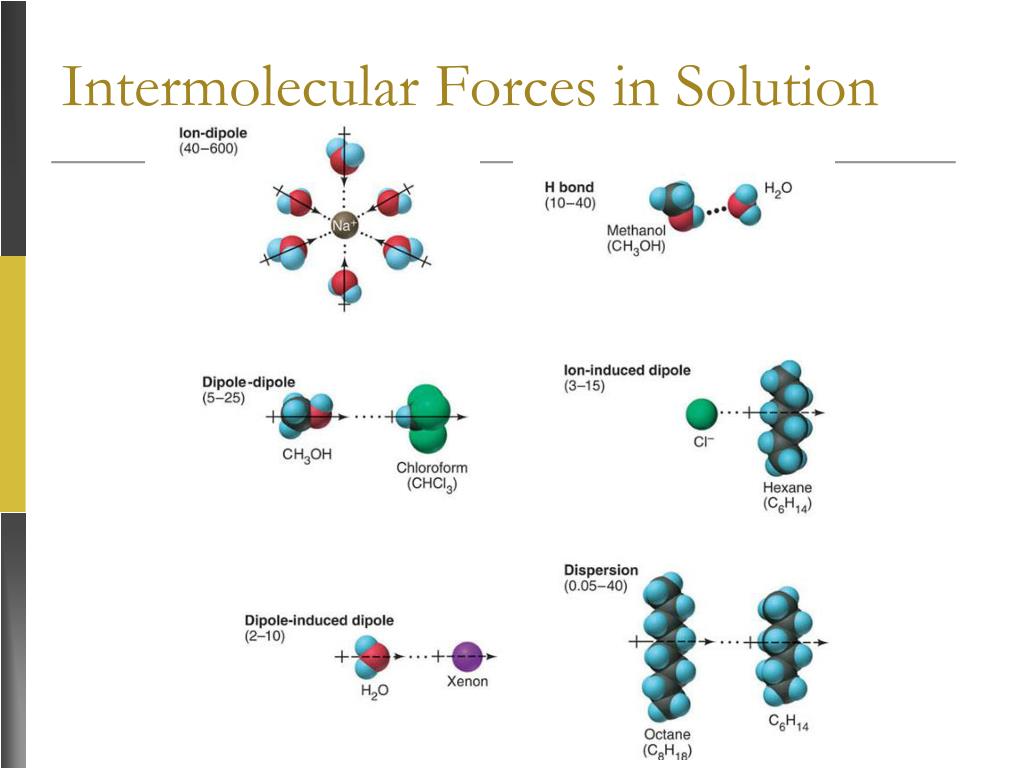
Ever wonder why oil and water just don't play nice? It's all about the invisible dance of tiny forces! These forces, called intermolecular forces, are like the secret handshakes between molecules. They're what make some things dissolve into others, and sometimes, they just refuse to mix.
Think of it like a party. Some molecules are super outgoing and love to mingle with everyone. Others are a bit more reserved and only want to hang out with their own kind. The "dissolving" process, or solvation, is basically these molecules deciding who they want to party with.
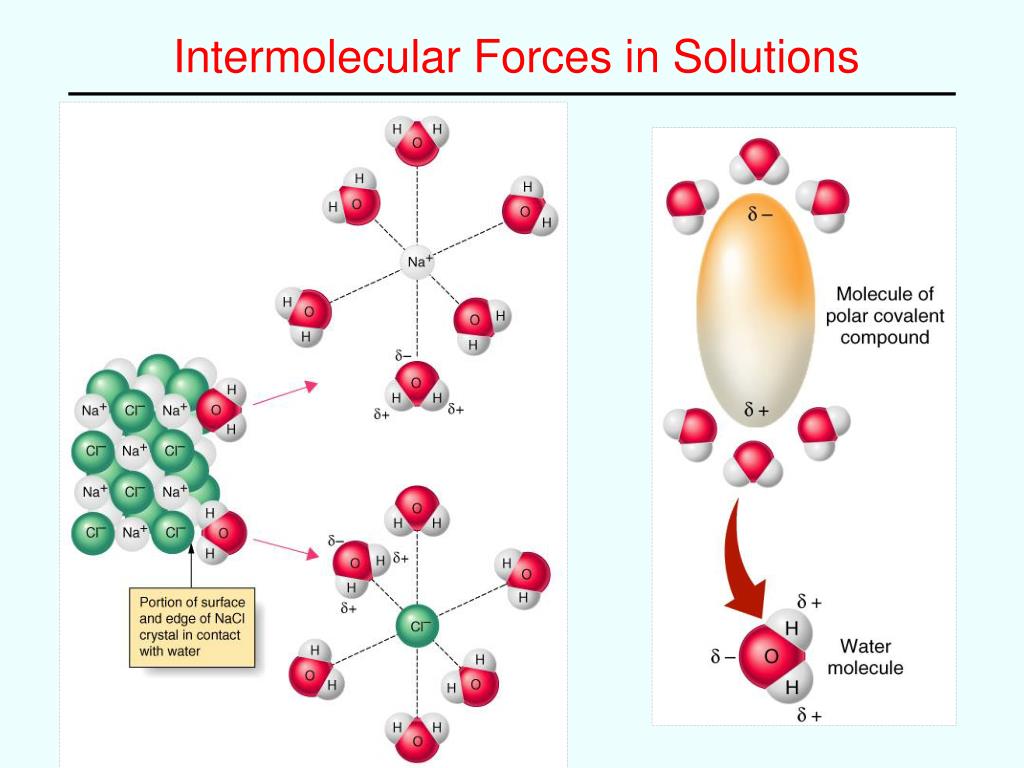
When you toss some salt into water, it’s a match made in heaven! The water molecules, which are like cheerful, chatty people, surround the salt ions, which are a bit more like lone stars. These interactions are so strong and welcoming that the salt just breaks apart and disappears. It's a beautiful example of how "like dissolves like."
So, what are these "likes" we're talking about? It’s all about the polarity of molecules. Imagine a molecule having a slight positive end and a slight negative end, like a tiny, gentle magnet. Water is a classic example of a polar molecule. It's got these little positive and negative poles that just love to attract other charged or slightly charged bits.
Now, take something like oil. Oil molecules are pretty much neutral. They don't have those distinct positive and negative ends. They’re more like a whole bunch of friends who all have the same vibe. They prefer to stick with each other because they don't have anything special to attract them to the water molecules.
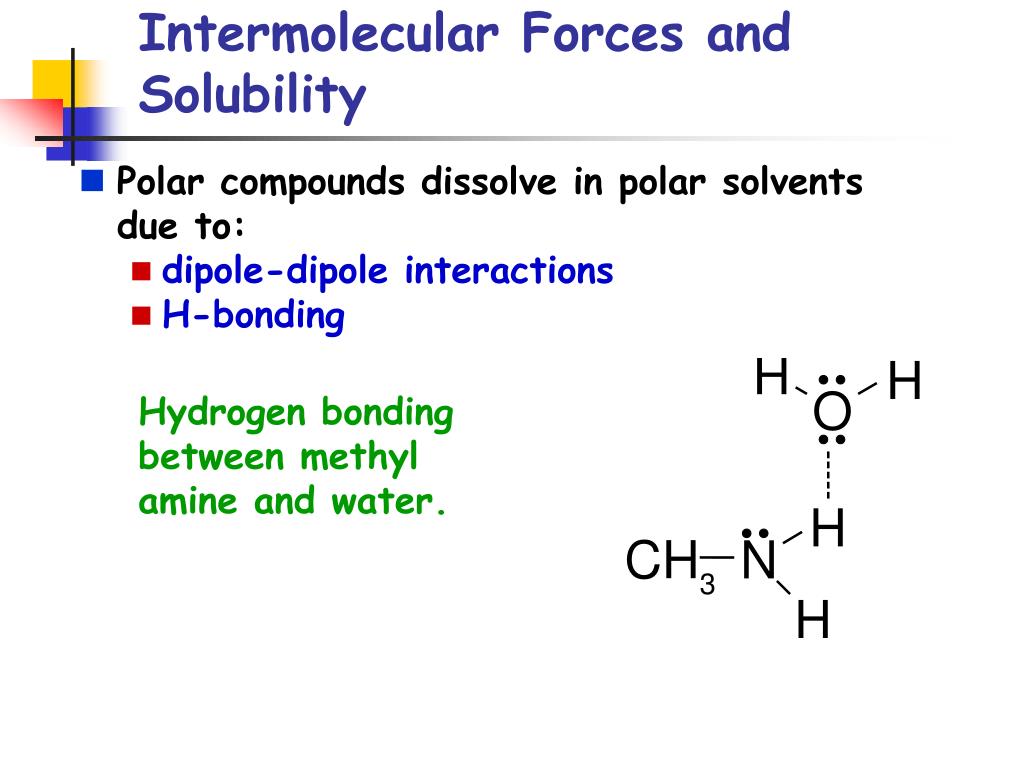
This is where those intermolecular forces really shine. Water molecules have a particularly strong type of intermolecular force called hydrogen bonding. It's like a super-duper, extra-strong hug they give to each other. They also have a force called dipole-dipole interactions, which are like smaller, friendly nudges between their charged ends.
When a polar substance, like salt (which breaks into charged ions) or sugar (which is also polar), meets water, these strong forces jump into action. The water molecules, with their positive and negative poles, are attracted to the oppositely charged parts of the salt or the slightly charged parts of the sugar. They pull them away from their buddies, creating a happy, dissolved mixture.
Oil, on the other hand, has weaker intermolecular forces. The main ones are called London dispersion forces. These are like fleeting, temporary attractions that happen when electrons in a molecule all bunch up on one side for a split second, creating a temporary negative charge. It's a very weak handshake that doesn't last long.
Because oil molecules have these weak forces, they don't have enough oomph to break the strong hydrogen bonds between water molecules. And the water molecules don't have anything strong enough to attract the oil molecules away from their own kind. So, they just sit there, side-by-side, like two groups of people at a party who haven't been introduced and have nothing in common.
It's fascinating to think about how these tiny, invisible forces dictate so much of what we see. Why does your salad dressing separate? Intermolecular forces! Why does soap work to clean greasy dishes? You guessed it, intermolecular forces! Soap molecules are clever; they have a polar end that likes water and a non-polar end that likes grease. They act as a bridge, allowing the grease to be carried away by the water.
The strength of these forces can vary a lot. Some molecules have really strong attractions, leading to substances that are very soluble. Others have weaker attractions, making them less soluble or even insoluble. It's like a spectrum of how well molecules get along.
Consider alcohol. It has a polar part (thanks to an oxygen atom) and a non-polar part (the carbon chain). This dual nature means it can interact with both polar things (like water) and non-polar things (like some oils). That's why alcohol is often used to dissolve things that water can't handle alone.
The process of solvation isn't just about dissolving. It also involves energy. When new interactions form between the solute (the stuff being dissolved) and the solvent (the stuff doing the dissolving), energy is released. When existing interactions in the solute and solvent need to be broken, energy is absorbed.
If the energy released from forming new solute-solvent interactions is greater than the energy absorbed to break old interactions, the process happens easily. This is a super happy, energy-releasing situation, and the solute dissolves wonderfully. It's like everyone at the party is so excited to meet new people that they have tons of energy to spare!
If the opposite is true, and more energy is needed to break apart the original molecules than is released when they mix, then solvation won't happen readily. The molecules decide it’s just too much effort to break up and make new friends. They’d rather stay with their existing pals.
The types of intermolecular forces involved also influence the rate of solvation. Stronger forces might mean a slower initial mixing, as it takes more effort to pull the molecules apart. However, once they start interacting, the strong attractions can lead to a complete and stable solution.

It's like a delicate balance. You have to overcome the existing "friendships" (intermolecular forces) within the pure solute and pure solvent to create new "friendships" between them. If the new friendships are attractive enough, and the energy cost isn't too high, then you get that magical disappearing act!
Think about it in terms of what you see every day. When you're making a cup of tea, the sugar dissolves because its polar molecules are attracted to the polar water molecules. The intermolecular forces between sugar and water are strong enough to overcome the forces holding the sugar crystals together and the forces holding the water molecules together.
But if you try to dissolve a greasy stain with plain water, it's a no-go. The non-polar grease molecules only have weak London dispersion forces. They can't compete with the strong hydrogen bonds of water. The water molecules are much happier holding hands with each other than trying to pull apart the greasy molecules.
This understanding of intermolecular forces is actually super important in lots of fields. In medicine, it helps us understand how drugs dissolve in the body. In cooking, it explains why some ingredients blend well and others don't. It's even crucial in environmental science, like understanding how pollutants dissolve in water.
So, the next time you see something dissolve, or not dissolve, take a moment to appreciate the silent, unseen world of intermolecular forces. They’re the unsung heroes (or villains, if you're trying to mix oil and water!) of chemistry, making our world a fascinating place to explore.

It's like a tiny, microscopic drama playing out all the time. Who will mix? Who won't? It all comes down to the invisible strings that tie molecules together. And understanding those strings can unlock a whole new appreciation for the everyday magic happening around us.
So, don't just think of it as dissolving; think of it as a molecular social event. And the intermolecular forces? They are the ultimate matchmakers, determining who gets to join the party and who has to sit on the sidelines.
The simplicity of the concept, "like dissolves like," is actually rooted in these complex interactions. It’s a beautiful reminder that even the most common phenomena have deep, fascinating explanations waiting to be discovered. It’s this hidden world of molecular behavior that makes chemistry so endlessly captivating.
So, go ahead, pour some water on that oil spill (it won't help much, sadly!), or marvel at how quickly sugar vanishes in your coffee. You're witnessing the captivating power of intermolecular forces at play, orchestrating the grand dance of solvation.
The universe is made of stories, not of atoms. —Muriel Rukeyser
And in the world of molecules, those stories are told through the strength and nature of their intermolecular attractions. It's a constant narrative of connection, repulsion, and the ultimate quest for stability. It’s a story that’s both incredibly simple and profoundly complex, making it an endlessly entertaining spectacle for anyone curious enough to look.