
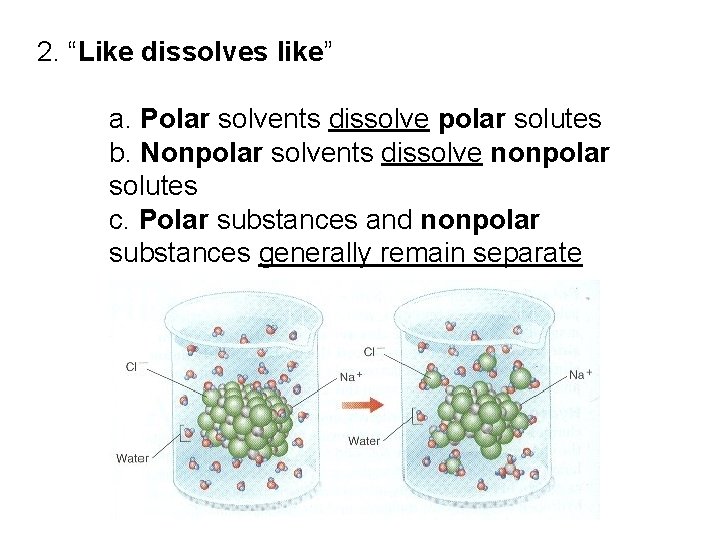
Have you ever wondered about the secret lives of solvents? These unsung heroes of the science world are like tiny molecular matchmakers. They help other substances get together, dissolve, and generally have a grand old time. Today, we're going to chat about one particular solvent that's got a bit of a reputation. It's called Hexane, and it's a real gem in the nonpolar solvent club.
Now, "nonpolar" might sound a little technical, but think of it like this: some molecules are like tiny magnets, with a positive end and a negative end. They love to stick to other things that are also a bit magnetic, like water. These are called polar molecules. But Hexane? Hexane is like a chill, go-with-the-flow kind of molecule. It doesn't have those strong positive or negative ends. It's balanced, it's neutral, and it's super happy hanging out with other molecules that are just as chill.
So, what kind of party does Hexane crash? Because it's a nonpolar solvent, it absolutely adores dissolving nonpolar substances. It's like a rule of thumb in the solvent world: "like dissolves like." If you're a nonpolar molecule, Hexane is your best buddy, your ultimate party starter, your wingman for dissolving.
Let's talk about some of the things Hexane absolutely devours. Think about all those oily, greasy, waxy things in the world. They're usually nonpolar. Have you ever tried to wash grease off your hands with just water? It's a struggle, right? Water is polar, and grease is nonpolar. They just don't mix. But bring in Hexane? Oh boy, does it make magic happen!
One of the most famous things Hexane is known for dissolving is oils. Not just any oils, but the kind you find in plants. Think of those delicious vegetable oils you use for cooking, or the essential oils that make your aromatherapy diffuser smell so lovely. Hexane is a star player when it comes to extracting these precious liquids from seeds and fruits. Imagine a sunflower seed, packed with goodness. Hexane can gently coax out all those wonderful, nourishing oils, leaving behind the rest of the seed. It's a process used in industry to get us those everyday cooking oils we love. How cool is that?

But it's not just food oils. Hexane also has a field day with fats. These are the building blocks of many things we encounter, from the natural fats in our food to the waxes that protect leaves on plants. If it's got that fatty, nonpolar structure, Hexane is ready to mingle and dissolve it.
Then there are waxes. Think of the wax on apples to keep them fresh, or the beeswax used in candles and cosmetics. These are often complex mixtures of long hydrocarbon chains, and guess what? They are decidedly nonpolar. So, Hexane comes along, waltzes with the wax molecules, and helps to break them down and spread them out. This makes Hexane incredibly useful in industries that deal with waxes, like in producing polishes or removing unwanted wax residues.
What about plastics? Many plastics are also nonpolar. Hexane can be used to dissolve certain types of polymers, which is essential in recycling processes or in manufacturing where plastics need to be melted and reformed. It's like Hexane has a special key that unlocks the gooey potential of many plastics.
And let's not forget about those essential pigments found in nature. Think of the vibrant colors in leaves during autumn or the rich hues in flower petals. Many of these natural color compounds are nonpolar. Hexane is often employed to extract these pigments, allowing us to study them or even use them in dyes and paints. It's like Hexane is a treasure hunter for color!
It's quite fascinating, isn't it? This simple, nonpolar molecule, Hexane, has this incredible ability to interact with and dissolve a whole world of nonpolar substances. It's the reason we can enjoy so many wonderful products, from cooking oils to the pigments that color our world.

The beauty of Hexane lies in its straightforward nature. It's not trying to be something it's not. It's a nonpolar solvent, and it excels at dissolving nonpolar things. This predictability makes it a valuable tool. It's like having a specialized wrench in your toolbox; you know exactly when and how to use it to get the job done.
So, the next time you hear about Hexane, don't be intimidated by the sciencey name. Just remember it's the ultimate dissolved-with-nonpolar-things expert. It's the friend who knows exactly which crowd to hang out with and how to make them all feel at home. It's a silent but powerful force, making our lives a little bit easier and a lot more colorful, one dissolved nonpolar molecule at a time.

Hexane is your go-to for anything oily, fatty, waxy, or made of certain plastics and natural pigments. It's the master of nonpolar dissolution!
It’s a bit like a microscopic party planner, ensuring that all the nonpolar guests get to mingle and dissolve into the fun. The way it can selectively pull out these substances is truly remarkable. It’s not just about dissolving; it’s about precision and making connections. It’s why this seemingly simple chemical compound plays such a big role in so many different industries.
Think about it: without solvents like Hexane, extracting those essential oils for your favorite perfume or getting the oils out of seeds to make your salad dressing would be a much, much harder task. It’s a reminder that even the most "basic" chemicals have extraordinary capabilities.
So, while it might just sound like another chemical name, Hexane is actually a bit of a celebrity in its own right, a star performer in the world of chemistry, celebrated for its unparalleled ability to dissolve its nonpolar companions. It’s a quiet achiever, a behind-the-scenes superstar that makes a tangible difference in our everyday lives. Isn't that something to be curious about?

.PNG)

