Hey there, science explorers! Ever found yourself staring at a chemistry worksheet, specifically one labeled "Gas Stoichiometry Chem Worksheet 14 5 Answers," and wondered, "What in the world is this all about?" No worries, you're not alone. Think of this as a friendly chat about a topic that's actually pretty neat once you get the hang of it. We're diving into the world of gas stoichiometry, and let me tell you, it’s not as scary as it sounds. It’s like unlocking a secret code to how gases behave when they get together and react. Pretty cool, right?
So, what is stoichiometry, anyway? At its core, stoichiometry is all about the quantitative relationships between reactants and products in a chemical reaction. Imagine you're baking a cake. Stoichiometry is like the recipe – it tells you exactly how much flour, sugar, and eggs you need to make a perfect cake. In chemistry, instead of flour and eggs, we’re talking about molecules and atoms. And when we throw gases into the mix, things get a little… airy!
Gas stoichiometry is just stoichiometry, but with a focus on gases. Now, why are gases so special? Well, gases are wonderfully unpredictable and yet predictable at the same time. They spread out to fill any container, their particles are zipping around like tiny, hyperactive bumper cars, and their behavior can be described by some pretty elegant laws. Think of them as the rock stars of the chemistry world – a bit wild, but with a lot of underlying order.
When we're dealing with reactions involving gases, we need to know how much of a gas is reacting or being produced. This is where the "Chem Worksheet 14 5 Answers" probably comes in. These worksheets are usually designed to help you practice these calculations. They're like the training grounds for becoming a chemistry detective, piecing together clues to figure out the missing amounts.
The Magic of the Mole Ratio
The absolute heart and soul of any stoichiometry problem, gas or not, is the mole ratio. This comes directly from the balanced chemical equation. Think of the balanced equation as the official rulebook for the reaction. If you see 2 molecules of hydrogen reacting with 1 molecule of oxygen to make 2 molecules of water, the mole ratio tells you that for every 2 moles of hydrogen, you’ll use 1 mole of oxygen, and you’ll get 2 moles of water. Simple, right? It's like saying for every two red bricks, you need one blue brick to build a specific wall.

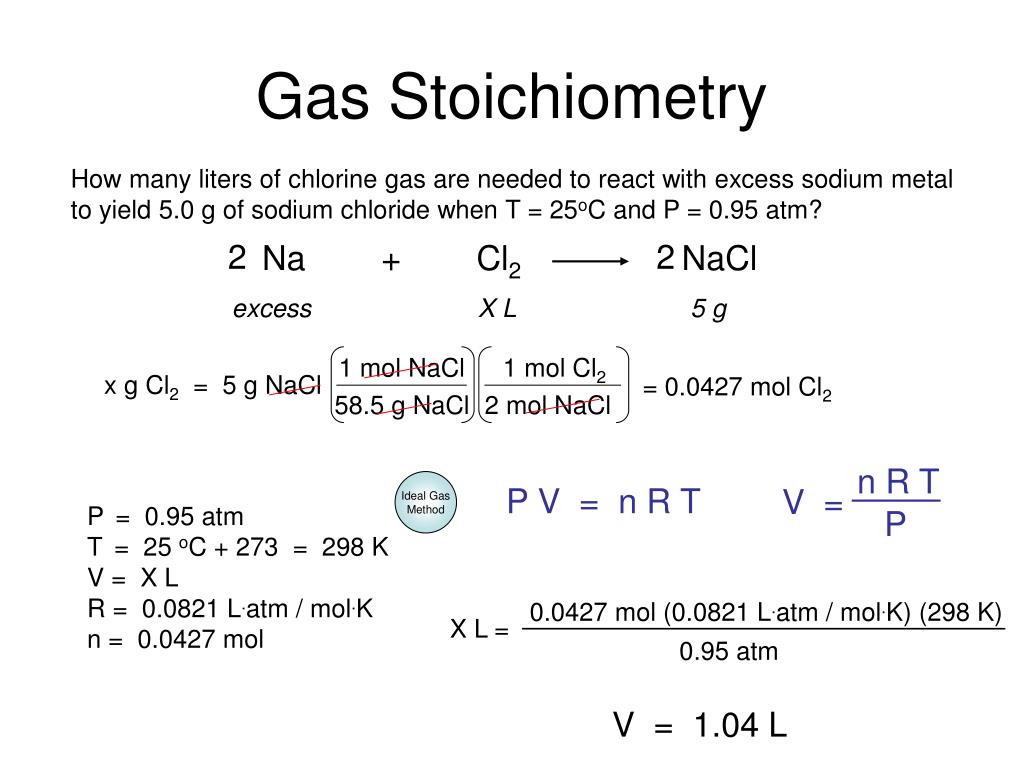
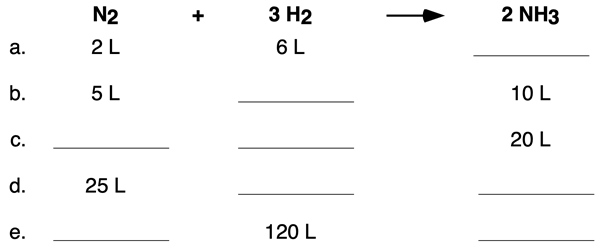
In gas stoichiometry, this mole ratio is still king. However, we often deal with gases in terms of their volume, especially at certain temperatures and pressures. And that’s where things get extra interesting. Have you ever wondered why a balloon expands when you blow into it? It’s all about the volume of the gas inside! In reactions, the volumes of gases that react and are produced can also be related by simple ratios, thanks to some brilliant scientists.
Avogadro's Little Secret
Here’s where Avogadro’s Law swoops in like a superhero. This law basically says that, at the same temperature and pressure, equal volumes of gases contain the same number of molecules (or moles). Mind. Blown. This means that the mole ratios we get from a balanced equation can also be applied directly to the volumes of gases involved in a reaction, provided the temperature and pressure are constant. It’s like being able to use your cake recipe not just for how many eggs, but also for how many bowls of batter you’ll get!

So, if your balanced equation says 2 moles of gas A react with 1 mole of gas B to form 2 moles of gas C, and you're doing this all in a sealed container at the same temperature and pressure, then 2 liters of gas A will react with 1 liter of gas B to produce 2 liters of gas C. How neat is that? It simplifies things a whole bunch, doesn't it?
This is often the core concept you'll be practicing on those "Gas Stoichiometry Chem Worksheet 14 5 Answers" sheets. You'll be given a reaction, maybe some information about one of the gases (like its volume, or mass which you then convert to moles), and you'll have to figure out the volume or mass of another gas involved. It’s like being a chemist on a mission!
Beyond the Simple Ratios: The Ideal Gas Law
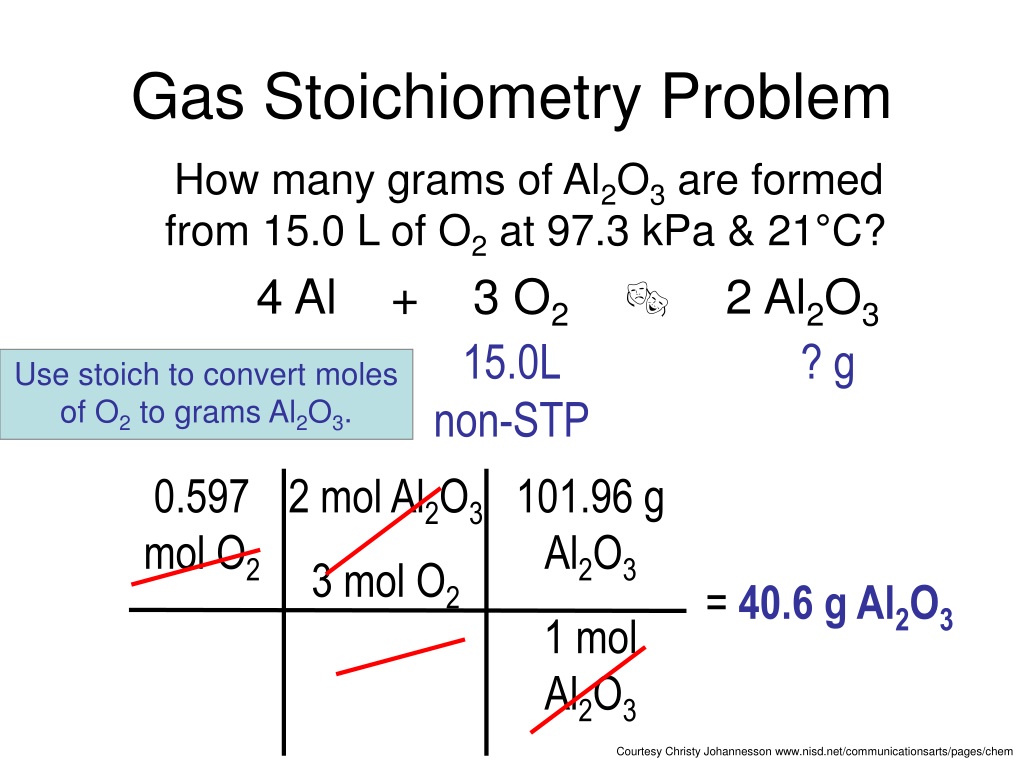
But what happens when the temperature or pressure isn't the same? Ah, that’s where things get a bit more complex, and the Ideal Gas Law (PV = nRT) becomes our best friend. This equation is like the ultimate weather report for gases. P stands for pressure, V for volume, n for the number of moles, R for the ideal gas constant (a fancy number), and T for temperature.

When you can't assume constant temperature and pressure, you use the Ideal Gas Law to figure out the number of moles (n) of a gas if you know its pressure, volume, and temperature. Once you have the moles, you can then use your trusty mole ratios from the balanced equation to find the moles of another substance. If you need the volume of that other gas, you can then plug its moles, the given temperature, and pressure back into the Ideal Gas Law to solve for its volume. It’s a bit of a multi-step dance, but totally doable!
These worksheets often test your ability to switch between volume and moles using this law. It’s like having a translator that converts between different units of measurement, but for gases and their properties. You might be given the volume of a gas at one set of conditions and asked to find the volume of another gas produced under different conditions. It’s a good way to really get a feel for how interconnected these gas properties are.

Putting It All Together: The "Answers" Part
Now, about those "Chem Worksheet 14 5 Answers." These are your guides. After you've wrestled with a problem, trying to figure out the mole ratios, maybe converting grams to moles, or using the Ideal Gas Law, you get to check your work. It’s a crucial part of learning. You wouldn’t learn to ride a bike without falling a few times, right? Checking the answers helps you see where you might have gone off track and reinforces what you did correctly.
Think of those answers as the solutions to little puzzles. Each problem is a case to solve, and the worksheet provides the evidence and the final verdict. Did you correctly convert grams to moles? Did you use the right mole ratio? Did you plug the correct numbers into the Ideal Gas Law? Seeing the correct answer helps solidify the process in your mind. It's like getting a gold star for mastering a new skill!
So, when you see "Gas Stoichiometry Chem Worksheet 14 5 Answers," don't let it intimidate you. Instead, think of it as an opportunity to understand the fascinating world of how gases behave in chemical reactions. It's about using balanced equations, mole ratios, and sometimes the powerful Ideal Gas Law to predict and quantify chemical changes. It’s a bit like being a chemist detective, using clues and logic to solve the mysteries of the invisible world of gases. And honestly, that’s pretty awesome.